Immunophenotyping based Systemic Lupus Erythematosus Recognition Service
Background Service Workflow Advantages FAQs Related Sections Contact Us
Creative Biolabs provides a cost-effective immunophenotyping based systemic lupus erythematosus recognition service to address challenges in precise diagnosis, monitoring, and therapeutic stratification of Systemic Lupus Erythematosus (SLE) by accelerating disease understanding, providing high-resolution immune cell profiles, and developing highly specific insights into disease progression through advanced flow cytometry and bioinformatics analysis.
The Necessity of Immunophenotyping in Rheumatoid arthritis Therapy
Systemic Lupus Erythematosus (SLE) is a complicated autoimmune disease with many clinical symptoms and immunological profiles. Current diagnostic and monitoring approaches often lack the specificity and sensitivity required for early intervention and personalized treatment. Immunophenotyping provides a powerful tool to dissect the intricate cellular landscape of SLE, identifying distinct immune cell subsets and their activation states. This deep cellular profiling is crucial for understanding disease pathogenesis, identifying biomarkers for disease activity and treatment response, and ultimately developing more targeted and effective therapies. The development of advanced immunophenotyping services is therefore essential to overcome current limitations and advance lupus research and clinical management.
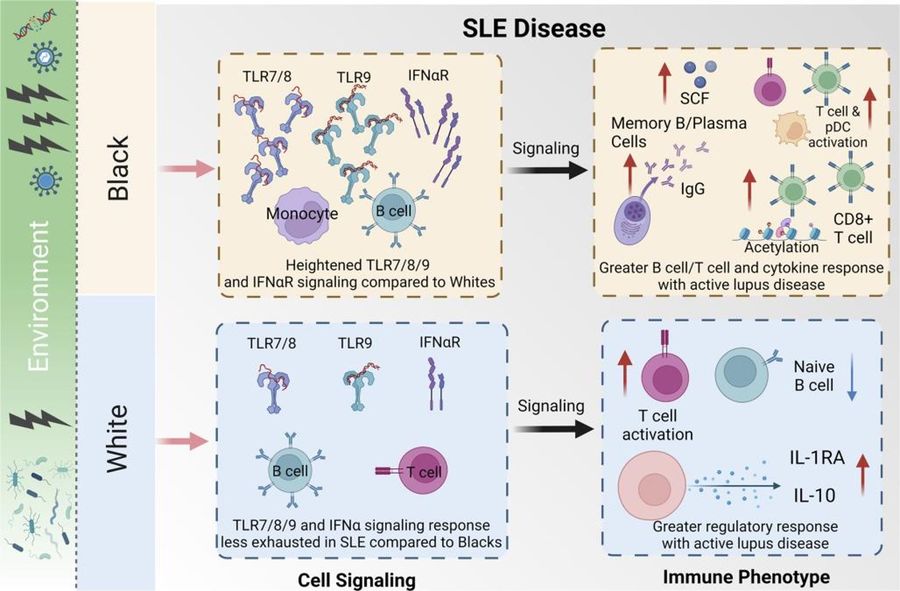 Fig.1 SLE disease in white women and black women.1
Fig.1 SLE disease in white women and black women.1
Immunophenotyping based Systemic Lupus Erythematosus Recognition Service at Creative Biolabs
Our service offers comprehensive, high-resolution analysis of immune cell populations, providing critical insights for your SLE research or clinical development projects. We perform this by utilizing advanced multi-parameter flow cytometry to meticulously characterize various immune cell subsets from your samples. This process typically takes 4 to 8 weeks, depending on project complexity, culminating in actionable data and detailed reports that support biomarker discovery, therapeutic target identification, and patient stratification. Our rigorous quality assurance protocols, including instrument calibration, data validation, and expert interpretation, ensure the reliability and accuracy of all results. We deliver precise cellular profiles, allowing you to gain a deeper understanding of disease mechanisms and therapeutic responses.
Required Starting Materials
-
Peripheral blood mononuclear cells (PBMCs), fresh or cryopreserved.
-
Whole blood samples (EDTA tubes recommended).
-
Clinical data (e.g., disease activity scores, medication history) for comprehensive correlation.
Workflow
Step 1: Sample Reception & Quality Control
Upon arrival, samples undergo rigorous quality assessment to ensure integrity and viability. This includes cell count, viability assessment, and basic morphological checks.
Step 2: Panel Design & Antibody Staining
Based on your project's specific objectives, we design a customized multi-parameter flow cytometry panel. Samples are then meticulously stained with a carefully validated cocktail of fluorescently conjugated antibodies targeting specific immune cell surface and intracellular markers relevant to SLE.
Step 3: Flow Cytometry Acquisition
Stained samples are acquired using state-of-the-art flow cytometers capable of analyzing up to 30+ parameters simultaneously. Our instruments are regularly calibrated to ensure consistent and accurate data collection.
Step 4: Data Pre-processing & Quality Assurance
Raw flow cytometry data (FCS files) undergo rigorous pre-processing, including compensation, doublet exclusion, and gating strategy validation. This procedure guarantees that the data is clean and reliable for further examination.
Step 5: Advanced Bioinformatics Analysis
We employ sophisticated bioinformatics tools and algorithms for unsupervised clustering and dimensionality reduction. This allows for the identification of novel cell populations, subtle shifts in immune cell subsets, and comprehensive visualization of complex datasets.
Step 6: Data Interpretation & Reporting
Our expert immunologists interpret the bioinformatics outputs, correlating cellular phenotypes with clinical data where provided. We provide detailed insights into the immune landscape, highlighting significant findings and their potential implications for SLE.
Our Service Highlights
-
Unparalleled Precision: We provide highly precise immune cell profiling, enabling deep insights into the heterogeneity of SLE.
-
Biomarker Identification: Our service facilitates the identification of novel biomarkers crucial for early diagnosis and monitoring disease activity.
-
Accelerated Research: The high-throughput nature of our approach significantly accelerates your research timelines.
-
Expertise-Driven Reliability: Our extensive expertise ensures robust and reliable data, critical for clinical translation.
-
Personalized Medicine Support: This comprehensive approach supports the development and implementation of personalized medicine strategies for SLE patients.
FAQs
Q1: What types of samples can I submit for the Immunophenotyping based Systemic Lupus Erythematosus Recognition Service?
A1: We accept fresh or cryopreserved peripheral blood mononuclear cells (PBMCs) as well as whole blood samples in EDTA tubes. For optimal results, please ensure proper sample collection and handling as per our guidelines.
Q2: How does this service compare to standard flow cytometry analysis for SLE?
A2: Our service goes beyond standard flow cytometry by offering highly customized multi-parameter panels, advanced bioinformatics analysis, and expert interpretation tailored specifically for the complexities of SLE. This allows for deeper insights into rare cell populations and subtle immune dysregulations not typically captured by basic analyses.
Q3: Can your service help identify specific cell subsets related to lupus nephritis or other SLE complications?
A3: Absolutely. By designing specific antibody panels and employing sophisticated analytical approaches, we can identify and quantify immune cell subsets implicated in various SLE manifestations, including lupus nephritis, providing valuable data for understanding disease heterogeneity.
Q4: What precautions should I consider when preparing and shipping samples for this service?
A4: To ensure sample integrity, it's crucial to follow our detailed sample preparation and shipping instructions, which will be provided upon project initiation. This typically includes maintaining a cold chain and using appropriate transport media.
Q5: Is it possible to integrate clinical data with the immunophenotyping results for a more comprehensive analysis?
A5: Yes, we strongly encourage submission of pertinent de-identified clinical data. Integrating clinical parameters with immune cell profiles allows for a more holistic understanding of disease progression and therapeutic response, enhancing the value of our analysis.
Related Sections
Beyond our SLE immunophenotyping service, Creative Biolabs provides a range of services designed to comprehensively support your autoimmune disease research and development endeavors:
Why Choose Creative Biolabs
Creative Biolabs' immunophenotyping-based systemic lupus erythematosus identification service is an effective and precise technique for deciphering the complexity of SLE. Leveraging advanced technology and deep expertise, we offer comprehensive immune cell profiling, delivering actionable insights to accelerate your research, enhance diagnostic capabilities, and pave the way for more effective therapeutic strategies. Please feel free to reach out to us with your meaningful projects.
Reference
-
Slight-Webb, Samantha et al. "Ancestry-based differences in the immune phenotype are associated with lupus activity." JCI insight vol. 8,16 e169584. 22 Aug. 2023, doi:10.1172/jci.insight.169584. Distributed under Open Access License CC BY 4.0, without modification.


 Fig.1 SLE disease in white women and black women.1
Fig.1 SLE disease in white women and black women.1
