Immunophenotyping based Ovarian Cancer Recognition Service
Background Service Workflow Advantages Customer Reviews FAQs Related Sections Contact Us
Facing challenges with long drug development cycles and unpredictable clinical outcomes for ovarian cancer? Creative Biolabs' immunophenotyping based ovarian cancer recognition service offers a robust solution, helping you streamline patient stratification and accelerate therapeutic research. By utilizing advanced flow cytometry and high-dimensional data analysis, we provide a deep understanding of the tumor's immune landscape, enabling you to design more effective, targeted treatment strategies.
The Crucial Role of Immunophenotyping in Ovarian Cancer Therapy
Despite advances, ovarian cancer remains the most lethal gynecological malignancy, often due to late-stage diagnosis and high rates of chemoresistance. The tumor microenvironment (TME) is a complex, dynamic system of immune cells, stromal cells, and molecules that can either fight the tumor or promote its growth and evasion from the immune system. A comprehensive understanding of this TME is crucial for developing effective immunotherapies. Traditional diagnostics often fail to capture this complexity, highlighting the need for advanced techniques like immunophenotyping to guide personalized treatment strategies. By systematically classifying ovarian cancer based on its immunophenotype, we can identify patient subgroups that are most likely to respond to specific immunotherapies, a necessity for the future of personalized medicine.
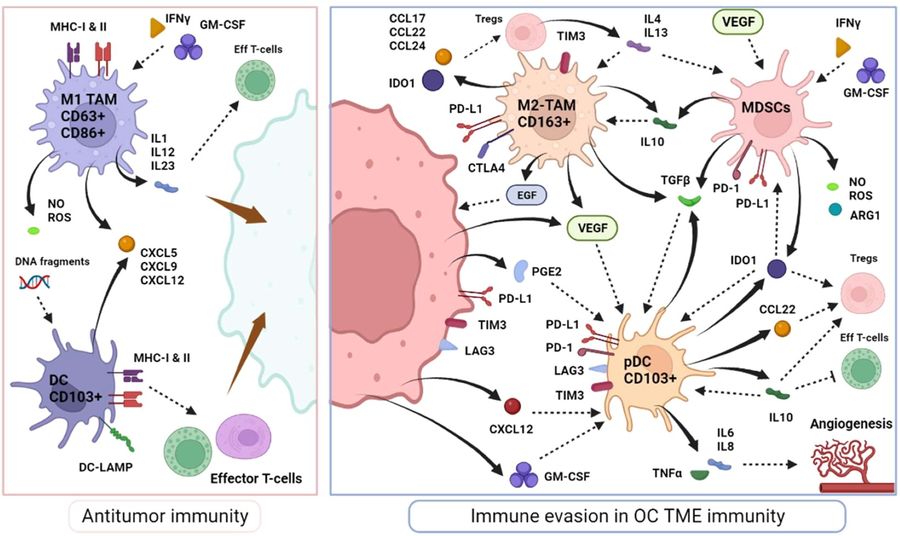 Fig.1 The immunological response of myeloid cells in the ovarian cancer microenvironment.1
Fig.1 The immunological response of myeloid cells in the ovarian cancer microenvironment.1
Immunophenotyping based Ovarian Cancer Recognition Service at Creative Biolabs
Our service provides a detailed, high-resolution view of the ovarian cancer immune landscape, offering critical insights to inform your research and clinical trials. We follow a systematic workflow from sample preparation to bioinformatic analysis, typically completed within 4-8 weeks, to provide actionable data. Our robust quality control measures, including rigorous antibody validation and standardized protocols, ensure the reliability and reproducibility of your results. This comprehensive approach delivers data on the composition, function, and spatial organization of immune cells within the tumor and its microenvironment, which is essential for biomarker discovery, patient stratification, and therapeutic target identification.
Workflow
Step 1: Sample Preparation and Processing
We begin by carefully disaggregating and preparing the provided samples to ensure a viable, individual cell suspension. This critical first step is optimized to maintain cell integrity and yield for subsequent analysis.
Step 2: Panel Design and Antibody Staining
Leveraging our expertise, we design a custom antibody panel with a multi-color flow cytometry approach. This panel is meticulously optimized to identify and quantify a broad range of immune cell subsets, including but not limited to T-cells, B-cells, NK cells, and various macrophage populations.
Step 3: Flow Cytometry Data Acquisition
Using state-of-the-art flow cytometry instruments, we acquire high-parameter data from the stained cell suspensions. This stage generates a large, comprehensive dataset that captures the unique immunophenotype of each sample.
Step 4: Bioinformatic Analysis and Interpretation
The raw data is then processed through advanced bioinformatic pipelines. We perform clustering analysis to identify distinct cell populations, compare immune profiles across different samples, and correlate findings with clinical data you provide, such as treatment response or survival outcomes.
Step 5: Comprehensive Report Generation
The final step involves compiling a detailed report that summarizes all findings. This report includes cell population frequencies, marker expression levels, graphical representations of the data, and expert interpretation to help you draw meaningful conclusions.
Our Service Highlights
High-Resolution Analysis
We provide a detailed, high-resolution view of the tumor immune microenvironment, going beyond traditional diagnostics to identify complex immune cell subsets and their interactions.
Target Identification
Our service serves as a critical tool for identifying novel therapeutic targets and biomarkers by providing a deep understanding of the immune landscape.
Predictive Insights
By analyzing the immunophenotype, we can help predict patient response to immunotherapies and guide patient stratification for clinical trials.
Personalized Medicine
Our data enables the development of truly personalized medicine strategies by linking specific immune profiles to treatment outcomes.
Informed Decision-Making
We empower you with high-quality, reproducible data and expert interpretation, allowing for more confident and strategic decisions in your therapeutic development pipeline.
Customer Reviews
-
Dr. A***n T 25/Apr/2025 «««««
Using Creative Biolabs' service in our research has significantly improved the precision of our patient stratification. The comprehensive data on tumor-infiltrating lymphocytes allowed us to better understand the immune landscape and more accurately select candidates for our clinical trial.
-
J***s R 11/Jun/2025 «««««
The immunophenotyping analysis from Creative Biolabs was instrumental in validating our in-house biomarker hypothesis. The high-parameter flow cytometry data provided concrete evidence of immune cell subtype variations, which was a crucial step in moving our project forward. Their expertise in panel design was a huge advantage.
FAQs
Q1: Can your service be used to guide patient selection for clinical trials?
A1: Yes. Our immunophenotyping service provides a detailed characterization of the tumor immune microenvironment, which is invaluable for patient stratification. By identifying specific immune profiles associated with treatment response, we help you select the most suitable patient cohort for your clinical trials, thereby increasing the likelihood of a successful outcome.
Q2: Is it possible to customize the analysis for my specific research needs?
A2: Absolutely. Our service is highly flexible and can be customized to your project. We work closely with our clients to design custom antibody panels that target specific markers of interest, ensuring that the analysis is perfectly aligned with your research questions.
Q3: What is the benefit of your service over a standard pathology report?
A3: While a pathology report offers a general overview of the tumor, our service provides a deep, quantitative, and multi-parameter analysis of the immune cells within the tumor and its microenvironment. We can identify subtle changes in immune cell populations and their activation states that are often missed by traditional histological methods, providing a more comprehensive understanding of the disease.
Q4: How can I begin a project with Creative Biolabs?
A4: Please simply click the inquiry prompt in any of the sections above, or contact us directly to discuss your project needs. Our team of experts will guide you through the process, from designing the experiment to providing you with a customized quote and a clear project timeline. We look forward to partnering with you.
Related Sections
Why Choose Creative Biolabs
Creative Biolabs brings over two decades of experience in immunobiology and cancer research to your project. Our commitment to quality, combined with our advanced technology platforms, ensures reliable and reproducible results. We offer a tailored approach, working closely with clients to design experiments that meet their specific research goals. If you would like more information about our services, please do not hesitate to contact us.
Reference
-
Blanc-Durand, Felix et al. "Targeting the immune microenvironment for ovarian cancer therapy." Frontiers in immunology vol. 14 1328651. 18 Dec. 2023, doi:10.3389/fimmu.2023.1328651. Distributed under Open Access License CC BY 4.0, without modification.


 Fig.1 The immunological response of myeloid cells in the ovarian cancer microenvironment.1
Fig.1 The immunological response of myeloid cells in the ovarian cancer microenvironment.1

