Immunophenotyping based Prostate Cancer Recognition Service
Background Service Workflow Advantages FAQs Related Sections Contact Us
To address challenges in biomarker identification and patient stratification for complex clinical trials in oncology, Creative Biolabs offers an immunophenotyping based prostate cancer recognition service that helps you accelerate drug discovery and streamline clinical trial processes through advanced high-dimensional data analysis and innovative protein engineering techniques. Our solution provides a non-invasive, objective method for disease detection and risk assessment, laying the groundwork for targeted therapies and personalized medicine.
Immunophenotyping: A New Era in Prostate Cancer Recognition
Current prostate cancer (PCa) diagnostic methods, such as the prostate-specific antigen (PSA) test, often lead to a high number of false positives, resulting in unnecessary and invasive prostate biopsies. This presents a significant challenge in patient care and clinical research. The immune system is intricately involved in the development and progression of tumors, offering a powerful avenue for early detection. As demonstrated in published literature, advanced computational approaches applied to high-dimensional immunophenotyping data can provide a more accurate and objective assessment of disease status and risk, a crucial necessity for modern oncology.
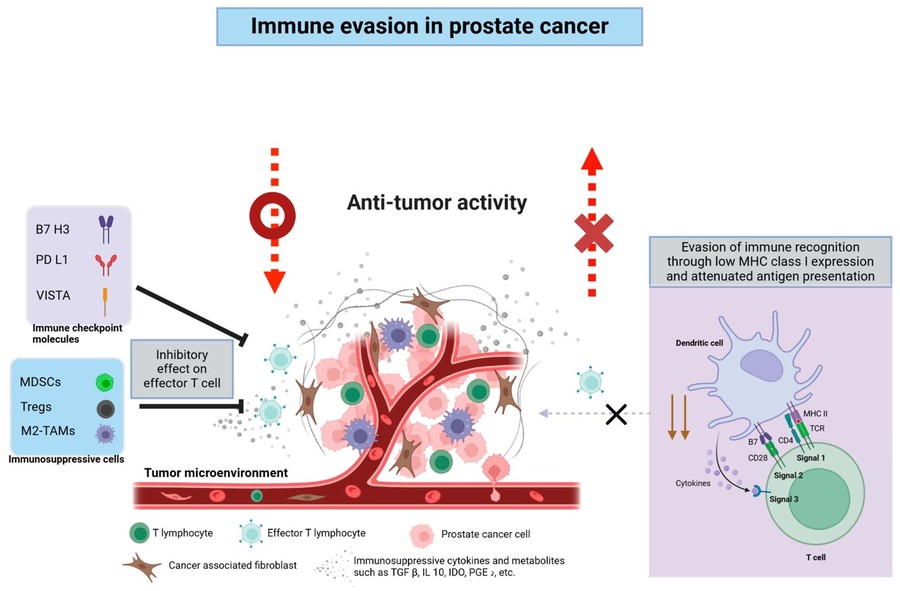 Fig.1 Immune evasion in prostate cancer.1
Fig.1 Immune evasion in prostate cancer.1
Immunophenotyping based Prostate Cancer Recognition Service at Creative Biolabs
Creative Biolabs' immunophenotyping based prostate cancer recognition service provides a sophisticated and data-driven solution that moves beyond the limitations of traditional methods. We deliver clear, actionable insights that can be utilized to inform biomarker discovery, guide patient stratification for clinical trials, and develop personalized therapeutic strategies. Our service is designed to support your project from initial sample analysis to final strategic recommendations.
Required Starting Materials
Clients can initiate the service by providing:
-
Peripheral blood mononuclear cells (PBMCs)
-
Cryopreserved whole blood samples
-
Previously generated high-dimensional flow cytometry data files
Workflow
Step 1: Sample Preparation and Quality Control
Our team processes your samples to isolate and prepare the immune cell populations required for immunophenotyping. This stage includes stringent quality control checks to ensure data integrity and reproducibility.
Step 2: Multi-parameter Flow Cytometry
We perform high-dimensional immunophenotyping using a comprehensive panel of markers to analyze key leukocyte populations. This process captures extensive data on dozens of cellular parameters simultaneously from a single sample.
Step 3: Advanced Computational Analysis
The high-dimensional data is processed using sophisticated machine learning algorithms and neural networks, moving beyond subjective manual gating. This allows us to identify subtle, complex patterns in immune cell profiles that are highly indicative of prostate cancer.
Step 4: Biological Interpretation and Report Generation
Our expert scientists interpret the computational results, correlating the identified immune profiles with disease status, clinical risk, and potential biomarkers. We compile a detailed report summarizing all findings and analyses.
Step 5: Strategic Project Recommendations
Based on the results and your project goals, we provide strategic recommendations on how to best leverage the data, whether for validating therapeutic targets or optimizing clinical trial design.
Our Service Highlights
Non-invasive and Objective
Our service reduces patient discomfort and risks associated with biopsies. Advanced computational analysis provides objective results, reducing subjectivity.
High Sensitivity and Specificity
Our sophisticated approach minimizes false positives, providing a more reliable foundation for clinical decisions.
Accelerated Project Timelines
By delivering precise, data-driven insights early on, our service helps you accelerate biomarker discovery, patient stratification, and drug development timelines.
Robust Data for Research
We provide a reliable foundation of high-dimensional data for your research and clinical decision-making, enabling more confident conclusions and strategic planning.
FAQs
Q1: How does this service compare to a standard PSA test?
A1: Our service provides a more comprehensive analysis than a standard PSA test. While PSA can be non-specifically elevated, our immunophenotyping approach analyzes the entire immune cell landscape, offering a multi-parameter view that increases the accuracy and reduces the risk of false positives.
Q2: Can your service differentiate between different stages or grades of prostate cancer?
A2: Yes, our advanced computational analysis is designed to identify subtle immune profile variations that can correlate with different disease statuses, including distinguishing between low-risk and high-risk disease. This capability is a powerful tool for more precise patient management.
Q3: What type of data will I receive, and how can I use it in my research?
A3: You will receive a detailed report with a clear interpretation of the data, as well as the raw data files. This information can be used to validate new therapeutic targets, identify novel biomarkers, and refine patient stratification strategies for future clinical studies.
Q4: Is your service compatible with other research platforms or technologies?
A4: Our service is highly flexible and can be integrated with other platforms. The data we provide is compatible with various bioinformatics tools and can serve as a foundation for further analysis, such as gene expression profiling or functional assays.
Q5: What is the turnaround time for the service?
A5: The typical project duration is between 4 and 8 weeks, but this can vary depending on the specific requirements. We recommend contacting us to discuss your project and receive a personalized timeline and quote.
Related Sections
Why Choose Creative Biolabs
Creative Biolabs is a leading partner in the biopharmaceutical industry, with deep scientific expertise and a commitment to innovative solutions. Our team of experienced biologists and computational scientists ensures precision and quality in every project. We are equipped with state-of-the-art technology to provide services that are both effective and reproducible. For more details about our services, please feel free to get in touch with us.
Reference
-
Kwon, Whi-An, and Jae Young Joung. "Immunotherapy in Prostate Cancer: From a "Cold" Tumor to a "Hot" Prospect." Cancers vol. 17,7 1064. 21 Mar. 2025, doi:10.3390/cancers17071064. Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only | Not For Clinical Use


 Fig.1 Immune evasion in prostate cancer.1
Fig.1 Immune evasion in prostate cancer.1

