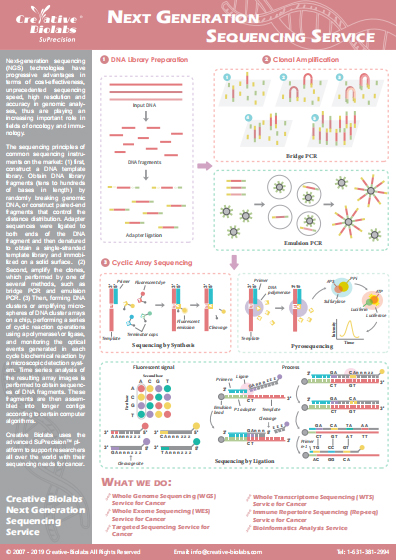
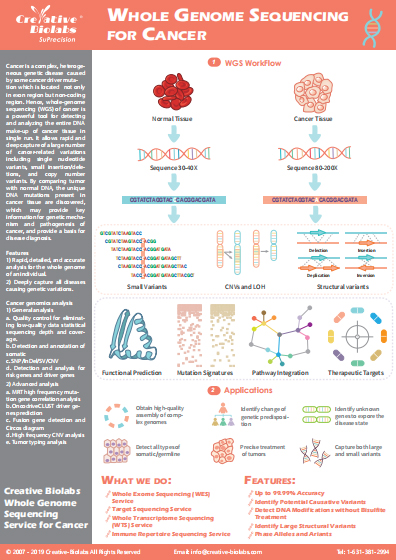
Whole Exome Sequencing (WES) based Tumor-Specific Neoantigen Discovery Service
With the development of cancer genomics, the identification of tumor-specific neoantigens is a crucial step for cancer immunotherapy. Equipped with world-leading technology platforms and professional scientific staff in next-generation sequencing (NGS), Creative Biolabs has accumulated extensive experiences in whole-exome sequencing (WES). Our seasoned scientists have developed an efficient pipeline for the identification of tumor-specific neoantigen. We are pleased to share our cutting-edge technology and extensive expertise in tumor-specific neoantigens identification service to assist our client’s related project development.
The Background of Tumor-Specific Neoantigens
Since tumor antigens are crucial biomarkers for identifying tumor cells and are potential targets for cancer therapy, they have been paid more and more attention in cancer diagnosis, prognosis and targeted therapy. Tumor antigens can be broadly classified into two categories based on their specificity: tumor-specific antigens and tumor-associated antigens. Tumor-specific antigens are only present in tumor cells. Tumor-associated antigens are aberrantly expressed in tumor cells and are also expressed in some normal cells. In addition to abnormal expression patterns, tumor cells also contain a range of cancer somatic mutations and mutations in protein-coding regions might produce tumor-specific mutant proteins. Tumor antigens derived from these tumor-specific mutant proteins are unparalleled tumor biomarkers, because they are only produced by tumor cells and are potential tumor-specific neoantigens. Several prediction tools, including INTEGRATE-neo and pVAC-seq, have been developed to predict tumor-specific neoantigens from cancer somatic mutations. These tools can predict neoantigens produced by non-synonymous somatic mutations and gene fusions, respectively. However, these tools only predict neoantigens presented by class I MHC molecules that can be recognized by T cells, they do not consider the mutations in the extracellular regions of membrane proteins that can be recognized by mutation-specific antibodies.

Neoantigen Discovery Service at Creative Biolabs
Creative Biolabs has designed a powerful pipeline for a comprehensive assessment of tumor-specific neoantigen. WES is performed on the tumor and normal tissue samples provided as peripheral blood. DNA is extracted from patients' tumors. Fragmented genomic DNA from tumor and normal samples extracted is used for library construction and exonic regions are captured by in-house method. Paired-end sequencing, resulting in 100 bases from each end of the fragments for the exome libraries is performed on our SuPrecision™ Platform. Neoantigen prediction is based on peptide sequence alignment. It is straightforward to detect a sequence fragment absence (deletion) or a newly added sequence fragment (insertion) in the given predicted protein isoform by comparing it to the matched reference protein. Then, a statistical analysis is conducted to derive a recommended anchor sequence size.
Key Advantages of Neoantigen Discovery Service at Creative Biolabs Include but Are Not Limited to:
- One-stop solution, from sample processing to candidate neoantigen identification.
- Comprehensive assessment
- Cutting-edge SuPrecision™ Platform
- Fast turnover time
Creative Biolabs is fully competent and dedicated to providing one-stop-solution service for tumor-specific neoantigens discovery. We are confident to provide top-quality service at the most competitive price. We offer turn-key or ala carte services customized to our client’s needs. Please contact us for more information and a detailed quote.
Reference
- Zhou, Z.; et al. TSNAD: an integrated software for cancer somatic mutation and tumour-specific neoantigen detection. R Soc Open Sci. 2017, 4(4):170050. Distributed under open access license CC BY 4.0, without modification.
Resources
Infographics
Podcast
- TMB Analysis for Checkpoint Immunotherapy Response
- WES based Variant Analysis
- WES based Structural Variant Detection
- WES based CNV Detection
- WES based Biomarker Discovery
- Personal Tumor-Specific Neoantigen Vaccine Development
- MRD Monitoring
- Microsatellite Instability Analysis
- Immune Repertoire Germline Gene & Allele Identification
- One-Stop Cancer 3D Modeling
- Circle-Seq based eccDNA Identification
- RNA-Seq based Tumor Microenvironment Analysis