Creative Biolabs offers customers design and engineering services for synthetic binding proteins using protein scaffolds. It is a powerful method for isolation and exploitation of high-affinity binders in the fields of research, diagnosis and therapy.
Novel biomolecules with specified binding targets are particularly useful in many biotechnology applications, including diagnosis, therapy, and catalysis. Although natural proteins can be searched for such applications, engineering novel and useful biomolecules is attractive, and their availability can be potentially unlimited. Engineered non-antibody protein scaffolds have therefore been a hot topic in the last few years. They represent a new generation of universal binding frameworks, and can be used as future immune pharmaceuticals to complement the expanding repertoire of therapeutic monoclonal antibodies. Besides that, these protein scaffolds have the following advantages compared with other immune molecules: small sizes, good stability and easy production. They have become the invaluable tools for design of useful binding proteins in biotechnological and pharmaceutical applications.
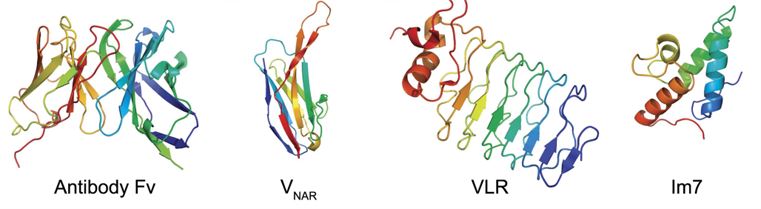
Figure 1. Cartoon illustrating diversity of protein folds utilised as binding-scaffolds. (Stewart D Nuttall et al., 2008)
Therefore, such scaffolds can be viewed as the building blocks for proteins with intended functions. The kind, number and spatial arrangement of the building blocks are expected to be crucial for protein function. Fibronectin is a very important player in the formation of the extracellular matrix and cell–cell interactions, while its type III domain (FN3) was used as a molecular scaffold. As a ubiquitous domain, FN3 occurs in approximate 2% of all animal proteins, and 15 of them are found in human fibronectin. FNfn10 is small (94 residues) and monomeric, and contains no disulfide bonds. High-level expression of correctly folded FNfn10 can be achieved in the bacteria system. The three-dimensional structure of FNfn10 has seven β-sandwich and three surface loops on each end potentially for multiple peptide segment presentation.
We can help you engineer proteins with various existing scaffolds, taking into consideration factors like disulfide formation and H-bonding for proper folding and stability. The synthesis and structural determination of engineering proteins are also available, whose specific properties can be further assessed by relevant biochemical and/or biophysical techniques.
Based on the formats of scaffold proteins, we can provide different customized protein design services, mainly including but not limited to:
If you are interested in our services, please contact us for more details.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.