Creative Biolabs offers a full package of immunogenicity analysis services to support preclinical drug discovery for small molecule, biologic drugs, and immunotherapy development programs. Particularly, we are happy to help global customers analyze the potential immunogenicity of multiple immunomodulatory agents for cancer therapies, which including but not limited to antibodies targeting immune checkpoints, bispecific antibodies, and chimeric antigen receptor.
In the past few years, numerous immune checkpoint inhibitors, as well as immuno-oncology mAbs with different structures/composition and immunomodulatory activities, have revolutionized treatment regimens for several malignancies. Meanwhile, the increasing use of immunomodulatory agents arises the risk of unwanted immune responses on the safety and efficacy of treatment compared with non-immunomodulatory agents. For example, immunomodulatory agents may potentiate immune responses through direct immunomodulation or inhibit immune responses as in the case of proapoptotic/depleting antibodies that target immune cells. In addition, in some cases, unwanted immune responses may result in the induction of anti-drug antibodies (ADA), which can induce infusion-related reactions or alter the pharmacokinetics (PK) of an agent.
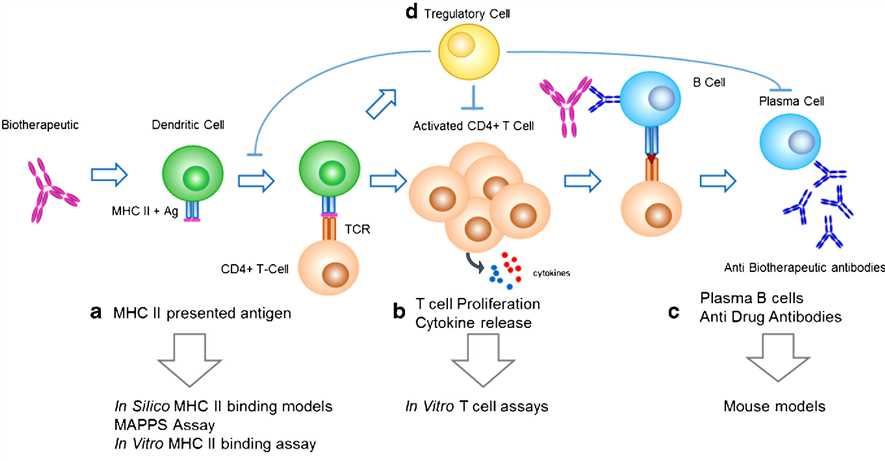 Fig.1 Key processes involved in the development of a humoral, MHC class II-mediated anti-biotherapeutic IgG response and corresponding predictive immunogenicity tools. (Gokemeijer, 2017)
Fig.1 Key processes involved in the development of a humoral, MHC class II-mediated anti-biotherapeutic IgG response and corresponding predictive immunogenicity tools. (Gokemeijer, 2017)
It has been reported that numerous factors such as product-related impurities, excipients, dosing regimen, type and stage of the disease, as well as prior and concomitant treatments, may affect the immunogenicity of a mAb. During preclinical development, both in silico algorithms and in vitro assays have been used to help select molecules with lower immunogenicity risk. Commonly, in silico algorithms are used to predict potential T cell epitopes. And in vitro binding assays are used to confirm the predicted sequences binding to MHC molecules. What’s more, the immunogenicity signals of a protein product can be assessed with dendritic cells (DC)-activation assay.
Working in the field of immunogenicity analysis for many years, Creative Biolabs has successfully established a top SIAT® immunogenicity platform to offer a full package of analysis services for global customers. Our experienced experts are able to provide high-quality immunogenicity evaluation services and reliable results analysis in a short period of time. Our comprehensive range of immunogenicity evaluation tests including:
For more details about our immunogenicity analysis services of tumors, please feel free to contact us or directly send us a quote.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.