Therapeutic antibodies contain a large number of post-translational modifications (PTMs), some of which affect the efficacy of the drug, the activity of the antibody and the binding of the antibody-antigen. In these modifications, cyclization of the antibody occurs mostly at the N-terminus and is generally caused by the cyclization of Glu or Gln. Recent studies have shown that glutaminase environmental enzymes promote the formation of N-terminal pGlu. Since most types of antibodies (Abs) contain glutamic acid and/or glutamine at their N-terminus, it is necessary to analyze the N-terminal cyclization to improve the modification of these antibodies and then improve their efficacy in the clinic. Creative Biolabs offers manufacturability assessment services by evaluating the N-terminal amino acid cyclization to achieve the maximum efficacy of potential drugs.
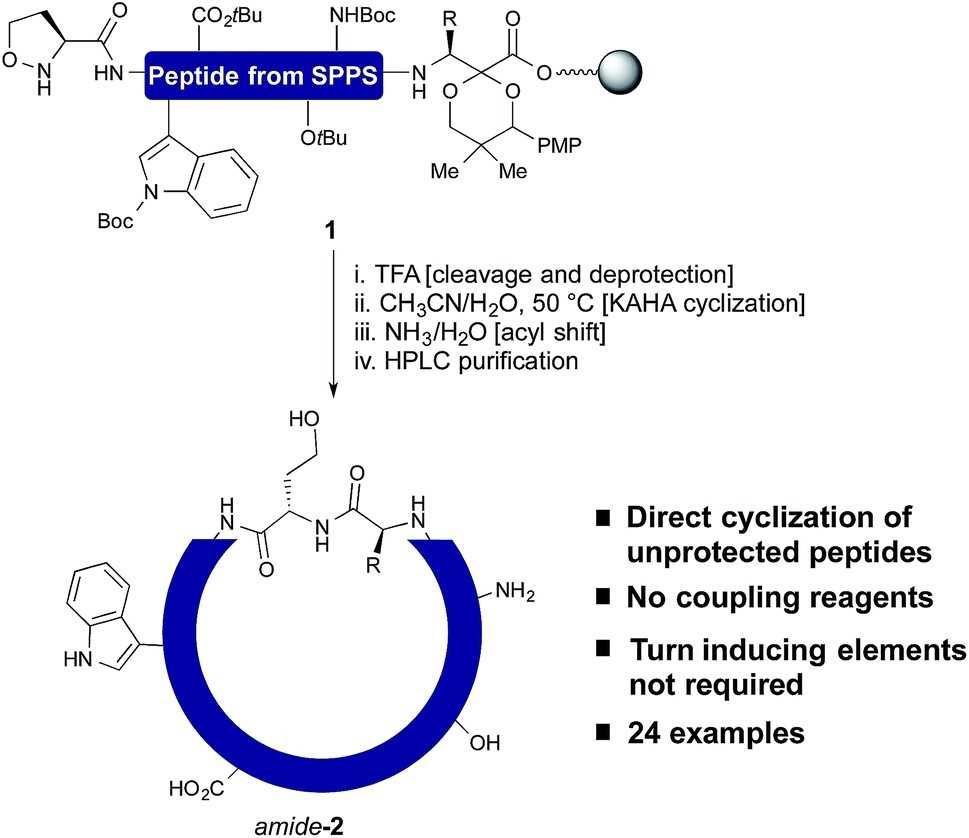 Fig.1 One step cleavage and cyclization of unprotected peptides with the KAHA ligation. (Rohrbacher, 2015)
Fig.1 One step cleavage and cyclization of unprotected peptides with the KAHA ligation. (Rohrbacher, 2015)
Cyclization of antibodies occurs mostly at the N-terminus, and Glu or Gln is generally converted to pyroGlu by the non-enzymatic reaction. After pyroGlu is formed, the terminal primary amine disappears, which increases the acidity of the antibody, but does not cause a change in charge.
If the biotechnological drug is N-terminally cyclized, there is no intermediate protein sequence, especially the end-closure of the antibody-drug due to pyroglutamic acid modification. Here we take monoclonal antibodies as an example to briefly introduce the steps of specific analysis and detection.
Monoclonal antibodies are double-stranded proteins that cannot be directly determined and must be resolved separately. The de-stranding method was SDS-PAGE reduction electrophoresis, and transferred to a PVDF membrane, followed by staining with Coomassie brilliant blue and decolorizing with methanol-water.
After protein transfer, it was washed several times with water while washing several times with pyroglutamate peptidase hydrolysis buffer.
The deblocked PVDF membrane was placed in a sample cell of a protein sequence analyzer, and the instrument was run to determine the N-terminal sequence.
The gene is inserted at an appropriate site of the antibody to improve the structure of the antibody and enhance the activity and affinity of the antibody.
Creative Biolabs owns advanced equipment and rich research experience in analyzing the N-terminal cyclization of your drug candidates. If you want to know more information, please contact us and we will happy to help you.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.