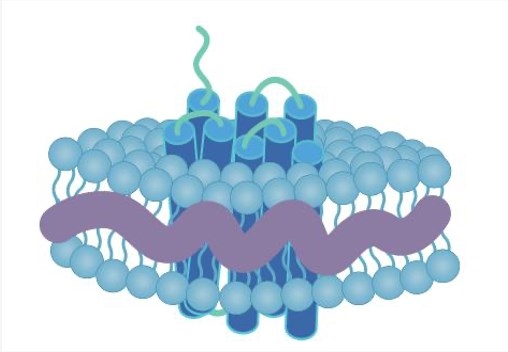
In most cases, membrane proteins are purified through recombinant methods and extracted using detergents to produce the proteins solubilized in detergent micelles. While detergents are the most common agents used for membrane proteins extraction, detergent addition strips the protein of its native lipid environment and thus membrane proteins’ stability and structure are compromised. To overcome this issue, an alternative polymer approach is designed to isolate and solubilize membrane protein in the most native format, completely without the use of detergents.
Amphipols are synthesized via the formation of amide bonds between octylamine and the carboxylic groups of low-molecular-weight polyacrylic acid precursors. The amphipols can make the transmembrane regions of proteins “trapped” around by these polymers, so as to keep hydrophobic domains stay folded.
The most significant advantage of the polymer system is the ability to encapsulate a membrane protein in a more “natural” environment than is provided by detergents.
Creative Biolabs has been dedicated to providing first-class membrane protein production service using a variety of strategies. Based on our leading-edge platform, we have successfully produced, purified, stabilized, and characterized many challenging membrane protein targets. We offer all-around, custom-oriented service of Magic™ membrane protein production. We are more than happy to leverage our advanced techniques and extensive expertise in this field to satisfy every single custom demand. Please feel free to contact us for more details.
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.