Scientists of Creative Biolabs provide high-quality membrane protein purification service using Synthetic Antigen Binders. With our solid platform and rich experience, we are able to satisfy your specific demands all through your project.
Synthetic antigen binders (sABs) are a novel and specific class of antibody-like molecules generated by phage display library technology which allows for displaying and screening antigen-binding fragments (Fabs) or single-chain variable fragments (scFvs) on the surface of bacteriophage. After several cycles of in vitro binding selection, antigen-specific binders are acquried. All of these binding molecules from this exquisite selection conditions are monoclonal, and their amino acid sequences can be decoded from the DNAs. Thus, this technology plays a significant role in structural and cell biology.
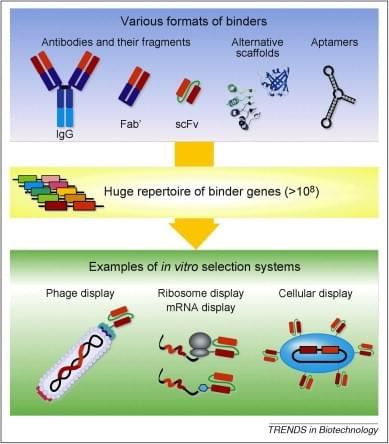 Figure 1. Schematic of synthetic antigen binders generation process in vitro. (Trends Biotechnol., 2010)
Figure 1. Schematic of synthetic antigen binders generation process in vitro. (Trends Biotechnol., 2010)
sABs can be applied to capture different types of targets, such as soluble proteins, multi-protein complexes, membrane proteins and functional DNAs or RNAs. The main utilities of sABs are described as below:
![]() Figure 2. Phage display cycle during membrane protein stalibization. (FEBS lett., 2004)
Figure 2. Phage display cycle during membrane protein stalibization. (FEBS lett., 2004)
As generation of sABs is highly conformation-specific, we can engineer the sABs to trap a specific conformational state of membrane proteins for further purification. We can perform a library of Fab fragments displaying on the surface of phage, then the phage library is exposed to the detergent-solubilized membrane protein with a biotin tag to form antibody/antigen complexes, which can be captured on streptavidin magnetic beads. After washing unbound phages, the membrane protein can be eluted from the magnetic beads. Remarkably, in this approach, the bound antibody fragment can form stable protein-protein interactions required for crystallization, moreover, the binding of antibody fragment can stabilize one specific conformation of the membrane protein to increase the structural homogeneity of the membrane protein of interest.
Creative Biolabs can also provide other various membrane protein production services. Please feel free to contact us for a detailed quote.
References
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.