Creative Biolabs proudly offers our TEM-1 β-lactamase phage display library construction service to assist our customers’ research goal. Through the proprietary Hi-Affi™ phage display platform, our scientists can generate the best scaffold library with high affinity and diversity.
TEM-1 β-lactamase is chosen as a novel scaffold different from the others in many respects. In practice, TEM-1 is the most commonly encountered β-lactamase in gram-negative bacteria, such as Escherichia coli, Haemophilus influenzae and Neisseria gonorrhoeae, and provides resistance to β-lactam antibiotics such as ampicillin, penicillin and cephamycin. Compared with other scaffold proteins, TEM-1 β-lactamase is a much larger (263 residues) protein with intrinsic enzyme activity and has a protein backbone consisting of numerous α-helices and β-sheets, and a disulfide bond.
An ideal TEM-1 β-lactamase scaffold is generally selected from TEM-1 β-lactamase phage display library. Because the TEM-1 β-lactamase can accommodate insertions of random sequences in two loops surrounding its active site without compromising its activity. Thus, libraries of TEM-1 β-lactamase is constructed by introducing one or two insertions of random peptides into permissible surface loops that surround the catalytic site that complex formation with a target protein could affect enzyme activity. These libraries can be firstly selected for retained β-lactamase activity and then be used for biopanning on antibodies or other target proteins to isolate variants with novel binding activities. As a consequence, the specific engineered TEM-1 scaffold with integrated target-binding activity can be potentially used as signaling molecules in direct or competitive homogeneous immunoassay.
Hi-Affi™ phage display platform, developed by Creative Biolabs, is an advanced system combines conventional phage display technology with trimer codon technology and NNK method. Other than common phage display technology, which is an exogenous gene expression method through fusing the target genes to bacteriophage coat proteins then displaying on the phage surfaces to select specific binders, more library randomicity can be achieved by our platform. In addition, it is more suitable for using Hi-Affi™ phage display platform to sort and isolate the high affinity protein or peptide targets. This technology can help our scientists to generate high quality TEM-1 β-lactamase phage display library with 100% precise mutant and over 1010 diversity.
Creative Biolabs own a professional team of scientists who are experts of scaffold protein library. We have generated about 55 kinds of scaffold libraries for clients all over the world by now, and are pleased to share our rich experience and advanced technology to serve our customers for generating the best scaffold protein library.
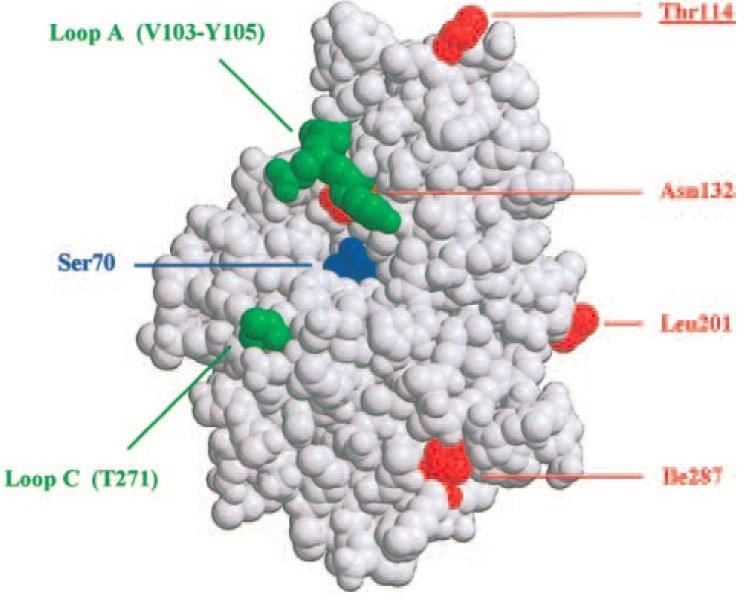 Fig. 1 Localization of insertion and mutation sites on the three-dimensional structure of TEM-1 β-lactamase. Insertions are represented in green, the active site is labeled in blue, additional residues mutated are indicated in red. (Legendre et al. 2002)
Fig. 1 Localization of insertion and mutation sites on the three-dimensional structure of TEM-1 β-lactamase. Insertions are represented in green, the active site is labeled in blue, additional residues mutated are indicated in red. (Legendre et al. 2002)
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.