ChimtoximAb Platform
Accurate targeting mechanism combined with lethal payloads, antibody-drug conjugates (ADCs) have become a new wave of next-generation immune therapeutics that are tipping the scale in the battle against cancer. Bearing similar concept, various forms of antibody (scFv, Fab, chimeric antibody, Fc-fusion protein…), referred to as “guidance chimeras”, can also be engineered into payload bearing warheads. With certain advantages over conventional full-length antibodies, the novel mAb chimera-toxin conjugates will greatly enhance the performance of the anti-tumor agents under suitable circumstances. Creative Biolabs is proud to provide the featured “ChimtoximAb” platform that is designated for the development of such mAb chimera-toxin conjugates.
Introduction of ChimtoximAb
Currently, the conjugation strategies used to join payload and a guidance chimera are similar to those used in developing full-length Ab-based immuno-conjugates. To develop a ChimtoximAb, the designer often utilizes the exogenous effector moieties and antibody fragment molecules to create the conjugate. ChimtoximAb development is trending in terms of discovering the next-generation, functionally enhanced immuno-therapeutics with fragment antibodies as well as Fc-fusion proteins. The payload adds a second dimension of efficacy into the ChimtoximAb to complement the original efficacy of the fragment antibody therapeutics, which depends mainly on the target binding by the single antibody domain. In the current clinical trial pipeline, about 50% of the fragment antibodies candidates are conjugated to exogenous functional moieties and they are used exclusively in cancer treatment, indicating ChimtoximAb development as a promising direction in scientific study and new drug discovery.
Antibody Fragment Conjugates
By far, a total of 24 conjugated antibody fragments have entered clinical trial stage, among which Metuximab-I131 (Licartin) is a murine IgG1 anti-CD147 F(ab)2 conjugated to iodine-131. It has been approved by the Chinese State FDA (CFDA) for the treatment of liver cancer. Meanwhile, eight related projects are being evaluated throughout the different phases of clinical development for Licartin. The chemical moieties used in develop different ChimtoximAbs vary. Among those 24 conjugated antibody fragments, 7 candidates are conjugated with radioisotopes, 12 with cellular toxins, 3 with cytokines, and one with an enzyme to target toxic metabolites.
- Fab Conjugates
Currently, a total of 12 Fab conjugates have entered clinical trials, including an anti-5T3 Fab conjugated to staphylococcal enterotoxin A, metuximab I-131, an active Phase 3 candidate, as well as naptumomab estafenatox (Anyara), a super-antigen that is designated to treat lung cancer by immune stimulation. Unfortunately, among the 12 Fab conjugates, 9 candidates have been withdrawn from clinical evaluation.
- scFv Conjugates
It has also been showed that by far, a total of 12 scFv conjugates have entered clinical trials, with six more under active development. Examples of the candidates under clinical evaluation include an anti-EpCAM scFv conjugated to pseudomonas exotoxin PE-38. This conjugate is developed for head and neck cancer treatment and it is currently in Phase 3 clinical trials.
- Fragment Ab-Fc Fusion Proteins Conjugates
Fragment Ab-Fc fusion proteins are also good candidates for ChimtoximAb development. For instance, a SNAP-fused single chain antibody fragments (scFvs) was conjugated to a BG-modified version of auristatin F (AURIF) to form a ChimtoximAb. The scFv-SNAP fusion protein in this construct was developed to target members of the epidermal growth factor receptor (EGFR) family which are frequently over expressed in breast cancer. The construct contained two scFv motifs for enhanced targeting and with the addition of the payload, this ChimtoximAb is highly efficient against breast cancer cell lines and caused apoptosis when applied at nanomolar concentrations.
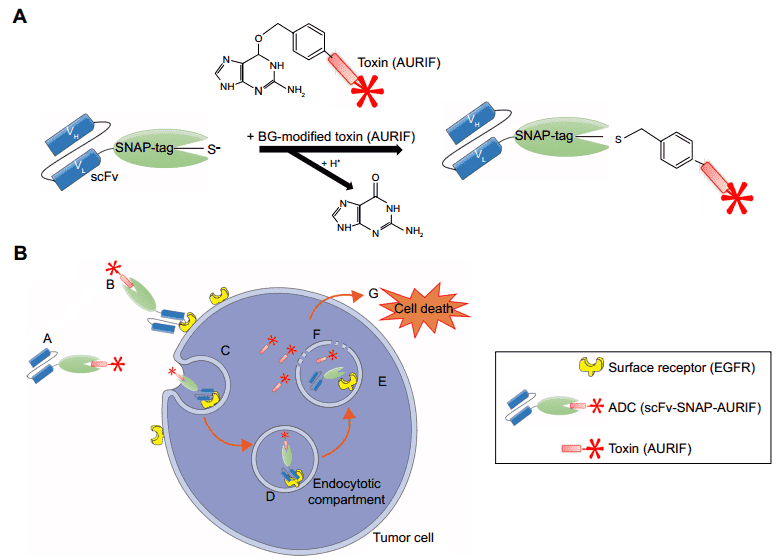 Fig.1 SNAP-tag technology and the mechanism of action of the scFv-SNAP-AURIF ADCs.1,2
Fig.1 SNAP-tag technology and the mechanism of action of the scFv-SNAP-AURIF ADCs.1,2
Advantages of ChimtoximAb
ChimtoximAb is a novel strategy in developing potent therapeutics for various application, for instance solid tumor therapy due to their higher tumor penetration, as well as diagnostic tools when conjugated with desired probes. Excellent tumor penetration and the feasibility for orthogonal conjugation site engineering highlight the advantages of ChimtoximAb.
Creative Biolabs offers high-quality ChimtoximAb services to promote the development of ADC, as well as therapeutic or diagnosis probes. Besides the types of ChimtoximAb mentioned above, scientists from Creative Biolabs also provide services to prepare selenomab conjugates (conjugation via engineered selenocysteine residues), camelid single-domain antibody-fragments conjugates, or any other special types of conjugates that you desire. Please contact us for more information.
References:
- Richards, Daniel A., Antoine Maruani, and Vijay Chudasama. "Antibody fragments as nanoparticle targeting ligands: a step in the right direction." Chemical science 8.1 (2017): 63-77.
- Distributed under Open Access License CC BY 3.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
