- Home
- UTC Development
- Antibody-Oligonucleotide Conjugate (AOC) Development
- AOC Conjugation Strategies
AOC Conjugation Strategies
Covalent and noncovalent bioconjugation chemistries are the main strategies for the preparation of antibody bioconjugates for various applications. With years of experience in antibody modification and bioconjugation, Creative Biolabs now provides a full range of conjugation strategies to help clients develop customized antibody-oligonucleotide conjugates (AOCs) in a time- and cost-efficient way.
Background
Noncovalent Approaches
A non-covalent bond is a special type of chemical bond which does not involve the sharing of a pair of electrons. Noncovalent interactions include affinity interactions, electrostatic, metal-mediated coordination, hydrophobic, and hydrogen bonding. Noncovalent approaches rely heavily on physical interactions via intermolecular noncovalent forces. The forces are weak by nature and they must work together to have a significant effect, namely cooperative effect. In recent years, advances in exploiting noncovalent interactions have led to the design of sophisticated biomolecules with complex architectures. Covalent-based synthetic methods become increasingly tedious as macromolecular structures and functional materials continue to evolve with a higher level of complexity and function. Noncovalent chemistries can overcome some of the drawbacks of covalent strategies such as lengthy synthesis and reagents' incompatibility. Currently, there are some widely-used methods include streptavidin-biotin and protein A/G-Fc mAb universal adapters.
- Streptavidin-Biotin System
Many supramolecular bioconjugation strategies employ biological recognition motifs, which are almost perfect guest-host systems. A classic example of such a system is the biotin-(strept) avidin interaction. Due to its small size (244.3 Da) and good stability, biotin can be easily noncovalently conjugated with many proteins, antibodies, polymers without any significant change in their biological activities.
- Protein A and Protein G
Antibody binding proteins such as protein A and protein G could also be used for antibody conjugation. For instance, the incorporation of additional unpaired cysteine residues into the backbone of protein A/G is a valuable strategy for maleimide-tagged ON conjugation.
Covalent Approaches
The covalent bond is the most common form of linkage between atoms in organic chemistry and biochemistry. The reaction of one functional group with another leads to the formation of a covalent bond via the sharing of electrons between atoms. Classical covalent bioconjugation approaches for the preparation of antibody conjugates are based on conjugation to either lysine residues, naturally present in native antibodies, or cysteine residues, which can, for example, be generated through reduction of interchain disulfide bonds. Nevertheless, these techniques afford heterogeneous mixtures comprising conjugate species with varying ON-to-antibody ratios. To generate more homogeneous AOCs, novel protein engineering and enzymatic approaches for conjugation have been actively developed and these techniques have been successfully applied for the preparation of site-specific AOCs. The following conjugation methods are the most commonly used to prepare AOCs.
- Conjugation on Lysine Residues of Antibodies
- Conjugation on Cysteine Residues of Antibodies
- Partially Reduced mAbs
- Chemical Protamine Conjugation to mAb
- Functionalization of Antibodies through Click Chemistry
- Strain-Promoted Alkyne-Azide Coupling (SPAAC)
- Inverse Electron-Demand Diels-Alder Reaction (iEDDA)
- Amine-to-Thiol Coupling
- Thiolation of Antibodies
- DNA-Templated Protein Conjugation (DTPC)
- Hydrazone Coupling
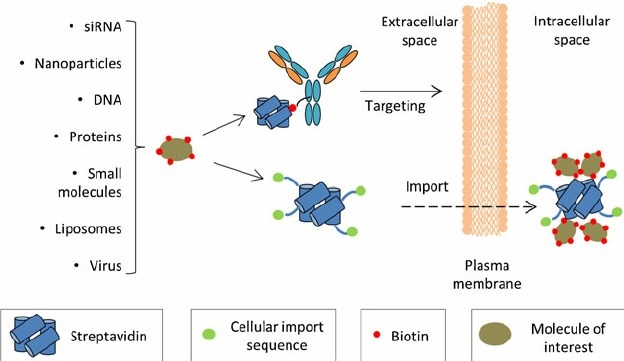 Fig.1 Streptavidin-based strategies for targeted drug delivery.1
Fig.1 Streptavidin-based strategies for targeted drug delivery.1
The advantages and disadvantages of the two methods are briefly compared as follows:
| Noncovalent Conjugation | Covalent Conjugation | |
| Advantages |
|
|
| Disadvantages |
|
Relatively complex processes and procedures |
Our Service
AOC Development Services
While a number of both covalent and noncovalent approaches exist to prepare AOCs, the choice of the appropriate conjugation method in each case usually depends on the specific purpose. If you are interested in our conjugation strategies for AOC development, please feel free to contact us for more information.
Highlights
- Advanced Site-Specific Conjugation: Creative Biolabs employs cutting-edge site-specific conjugation techniques for AOC development, ensuring precise attachment of oligonucleotides to antibodies, which enhances targeting accuracy and therapeutic effectiveness in complex applications.
- Optimized Stability and Functionality: Through covalent conjugation approaches, Creative Biolabs enhances the stability of antibody-oligonucleotide conjugates, maintaining the functional integrity of biomolecules, which is crucial for achieving long-term effectiveness in diagnostic and therapeutic applications.
- Improved Flexibility with Noncovalent Systems: Creative Biolabs leverages noncovalent bioconjugation strategies, offering flexibility in AOC design by using systems like streptavidin-biotin, which allow rapid assembly of conjugates without compromising functionality.
- Increased Homogeneity through Protein Engineering: Creative Biolabs utilizes protein engineering techniques to create homogeneous AOCs, reducing variability in conjugate composition, which significantly improves the consistency and reliability of experimental results and therapeutic outcomes.
- Comprehensive Conjugation Strategies: Creative Biolabs offers a full range of bioconjugation methods, including covalent and noncovalent approaches, ensuring customized antibody-oligonucleotide conjugates (AOCs) are efficiently developed.
FAQ
-
Q: What are the primary bioconjugation strategies employed by Creative Biolabs for AOC development?
A: Creative Biolabs utilizes both covalent and noncovalent bioconjugation chemistries for developing antibody-oligonucleotide conjugates (AOCs). These methods are tailored to meet the specific needs of clients, ensuring efficient and cost-effective development processes.
-
Q: Can you explain the noncovalent approach to AOC development at Creative Biolabs?
A: The noncovalent approach at Creative Biolabs involves using physical interactions via intermolecular noncovalent forces like affinity interactions and hydrogen bonding. This method is beneficial for its cooperative effect, overcoming some limitations of covalent strategies such as lengthy synthesis and reagent compatibility.
-
Q: How does the streptavidin-biotin system work for AOC development?
A: In the streptavidin-biotin system used by Creative Biolabs, biotin, a small and stable molecule, is noncovalently conjugated with proteins or antibodies. This system leverages the strong biotin-(strept)avidin interaction, facilitating effective bioconjugation without altering biological activities.
-
Q: What role do protein A and protein G play in antibody conjugation?
A: Protein A and protein G are utilized by Creative Biolabs for antibody conjugation through the addition of unpaired cysteine residues. This strategy supports maleimide-tagged oligonucleotide conjugation, enabling versatile antibody modifications.
-
Q: What are the covalent conjugation techniques used by Creative Biolabs for AOCs?
A: Creative Biolabs employs several covalent conjugation methods, including conjugation on lysine and cysteine residues of antibodies, chemically linking antibodies through click chemistry, and employing novel protein engineering for site-specific conjugations.
-
Q: How does Creative Biolabs ensure the quality and specificity of AOCs?
A: Creative Biolabs employs advanced protein engineering and enzymatic approaches to ensure the preparation of homogeneous and site-specific AOCs. These techniques optimize the functionality and stability of AOCs, catering to the precise needs of clients across various applications.
Published Data
In this experiment, antibody-oligonucleotide conjugation was achieved using a novel, site-selective method involving microbial transglutaminase (MTGase). This enzyme selectively conjugates oligonucleotides to monoclonal antibodies (mAbs), targeting the C-terminus of the antibody's heavy chain. The conjugation process creates homogeneous antibody-oligonucleotide conjugates, known as antibody-RNA conjugates (ARCs), with a consistent drug-antibody ratio (DAR) of 2. This uniformity avoids the heterogeneity and pharmacokinetic variability seen with random chemical conjugation methods. The resulting ARCs retain their antigen-binding specificity and offer potential for targeted oligonucleotide delivery to specific cell types, thus enhancing therapeutic efficacy while reducing off-target effects. These findings present a promising platform for expanding oligonucleotide therapies to tissues beyond the liver.
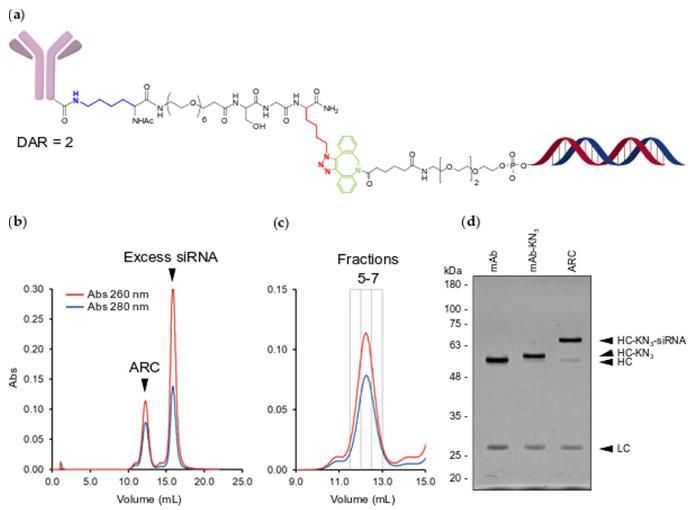 Fig.2 Conjugation and purification of ARC.2
Fig.2 Conjugation and purification of ARC.2
References
- Dundas, C.; et al. Streptavidin-biotin technology: improvements and innovations in chemical and biological applications. Applied microbiology and biotechnology. 2013, 97(21): 9343-9353.
- Huggins, Ian J., et al. "Site selective antibody-oligonucleotide conjugation via microbial transglutaminase." Molecules 24.18 (2019): 3287.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
