C. diff toxin B
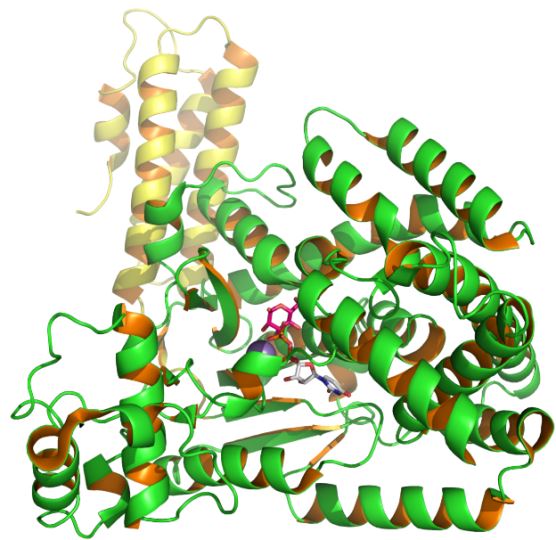 Clostridium difficile is a common nosocomial pathogen and a major cause of infectious diarrhea in hospitalized patients. Two structurally similar toxins denoted as TcdA and TcdB are the main virulence determinants associated with Clostridium difficile-associated disease. The TcdB is encoded by the tcdB gene and is pro-inflammatory, cytotoxic, and enterotoxic in the human colon. The molecular weight of TcdB is 270 kDa. TcdB is a glucosyltransferase that catalyzes the monoglucosylation of threonine 35/37 of small GTP-binding proteins Rho, Rac, and Cdc42 within target cells, and thus modulates several physiological cellular events resulting in cell death. Structurally TcdB contains three domains: the enzymatic activity is located in the N terminus of the protein, the middle domain is the putative translocation region, and the C terminus is involved in receptor binding. The N termini of TcdA and TcdB demonstrate 74% homology that accounts for their similar substrate specificity. The carboxy-terminal domain of both toxins carries a number of short homologous regions termed combined repetitive oligopeptides (CROPs).
Clostridium difficile is a common nosocomial pathogen and a major cause of infectious diarrhea in hospitalized patients. Two structurally similar toxins denoted as TcdA and TcdB are the main virulence determinants associated with Clostridium difficile-associated disease. The TcdB is encoded by the tcdB gene and is pro-inflammatory, cytotoxic, and enterotoxic in the human colon. The molecular weight of TcdB is 270 kDa. TcdB is a glucosyltransferase that catalyzes the monoglucosylation of threonine 35/37 of small GTP-binding proteins Rho, Rac, and Cdc42 within target cells, and thus modulates several physiological cellular events resulting in cell death. Structurally TcdB contains three domains: the enzymatic activity is located in the N terminus of the protein, the middle domain is the putative translocation region, and the C terminus is involved in receptor binding. The N termini of TcdA and TcdB demonstrate 74% homology that accounts for their similar substrate specificity. The carboxy-terminal domain of both toxins carries a number of short homologous regions termed combined repetitive oligopeptides (CROPs).
A number of studies have demonstrated that the presence of TcdB in mammalian cells leads to rapid changes in cell morphology and cell signaling. Within a short period of time, cells have the appearance of plaque, even with small dosages of TcdB and TcdA.
If you can’t find your product on this list? Please directly send email to .
For Research Use Only. NOT FOR CLINICAL USE.
Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
