- Home
- ADC Development
- DrugLnk™ Custom Linker-Payload Synthesis
- Drug Module Synthesis
- Microtubule Toxin Synthesis
- Auristatin Synthesis
Auristatin Synthesis Service
Creative Biolabs offers customized services for the design and production of antibody-drug conjugates (ADCs) using auristatin and auristatin derivatives as payloads. With years of experience in the ADC research, scientists at Creative Biolabs are confident to provide clients with high-quality auristatin ADCs with appropriate linkers to meet your demands.
Auristatin Mode of Action (MOA)
Auristatins are a family of complex analogues to the native antineoplastic product dolastatin 10. They are 100 to 1000 times more toxic than Doxorubicin, a conventional cancer chemotherapy medication. The potent agent, dolastatin 10 is a five-subunit penta-peptide discroverde by Pettit et al. from the sea hare Dolabella auricularia in 1987 and chemical modification to dolastatin 10 were applied to generate auristatins. It was later realized that the remarkable cytotoxicity of auristatins owes to their powerful capability to inhibit microtubule formation through the interaction with tubulin at the "peptide sub-site" of tubulin's "Vinca domain" and disrupt tubulin-dependent GTP hydrolysis. Auristatins lead to the arrest of cancer cells in the mitosis stage and eventually, apoptosis. Unfortunately, due to their high cytotoxicity, harmful side-effects and a narrow therapeutic window were reported in clinical trials of auristatins, rendering them unsuitable as anticancer medications.
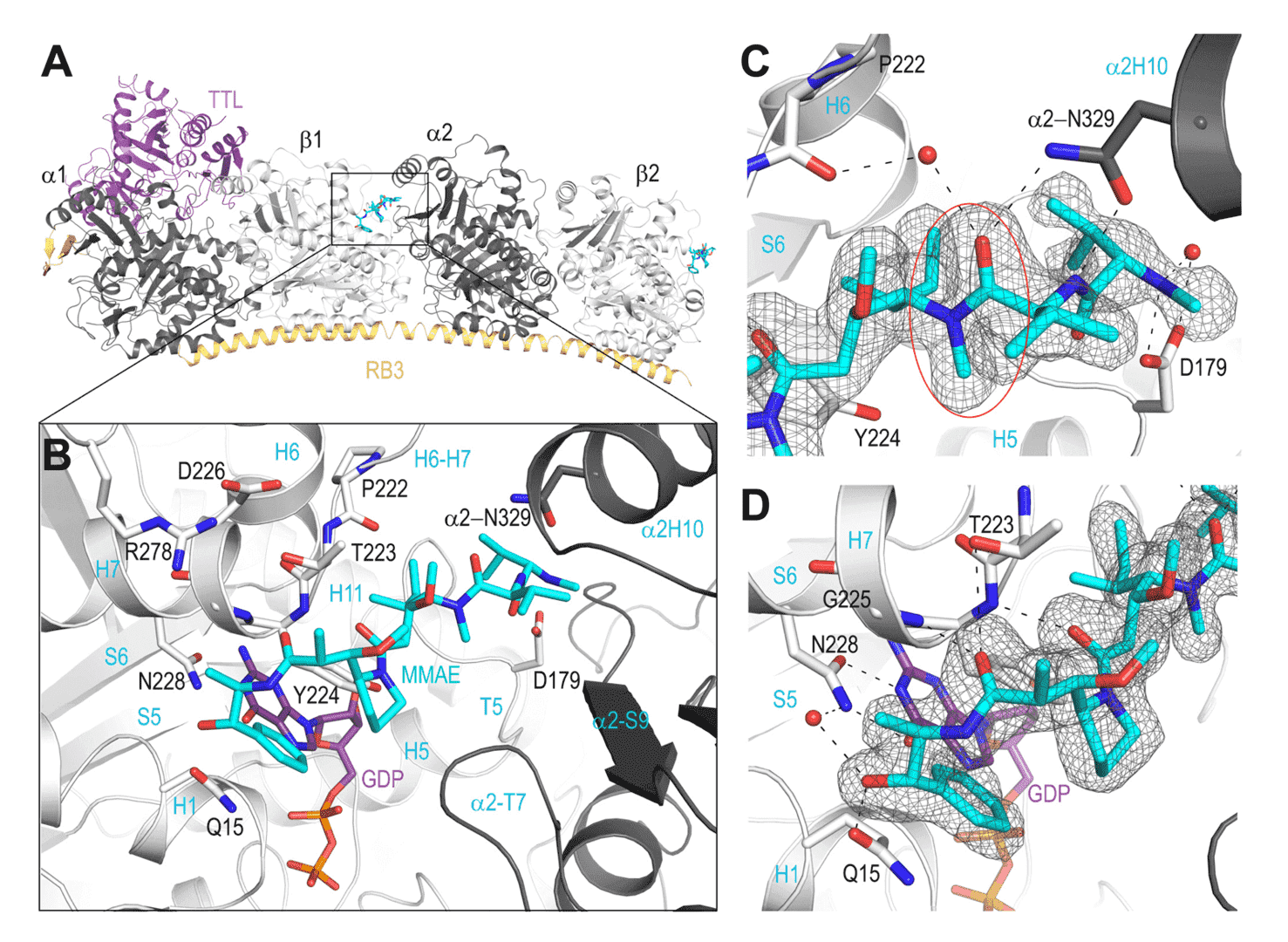 Fig.1 Binding of MMAE, an Auristatin derivative, to the α and β tubulin subunits solved by co-crystallography.1,2
Fig.1 Binding of MMAE, an Auristatin derivative, to the α and β tubulin subunits solved by co-crystallography.1,2
Modifications of Auristatins
A crucial discovery that finally changed the perspective for auristatins was the removal of one methyl group at the N-terminus of dolavaline to give rise to a series of auristatin analogues with only slightly decreased potency. Those new auristatins contain a secondary amine at their N-termini that enables their coupling with a linker and subsequent conjugation to monoclonal antibodies. This minor yet important modification resulted in the generation of extremely powerful ADCs. Generally, there are four strategies for the structural modification of auristatin analogues: N-terminal extensions, C-terminal modifications, displacements of the N-terminal dolavaline, and major peptide core structure modifications.
Auristatins-based ADCs
Utilizing the highly specific targeting mechanism of a monoclonal antibody, ADCs selectively deliver extremely powerful cytotoxic agents to tumor cells that express the particular cell surface membrane receptor. Made into an ADC, the drug efficacy of auristatins has been dramatically increased, along with an expanded therapeutic window.
At this moment, an increasing number of auristatin analogues are being synthesized and the application of auristatin derivatives will become one of the cornerstones for ADCs development. What’s more, other targeting strategies for the selective delivery of auristatins to tumors are being investigated, such as MMP-targeting prodrug analogues and albumin conjugates with targeting peptide ligands. Creative Biolabs is specialized in designing and creating ADCs with tailored auristatins payloads, optimized linkers, and suitable conjugation strategies using our advanced "DrugLnk" research platform to meet every client’s unique requirements. In the meantime, Creative Biolabs also provides other microtubule toxins as well as various ADC-related services. Please contact us for more information and a detailed quote regarding your ADC design and production.
References:
- Waight, Andrew B., et al. "Structural basis of microtubule destabilization by potent auristatin anti-mitotics." PloS one 11.8 (2016): e0160890.
- Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
