- Home
- ADC Development
- DrugLnk™ Custom Linker-Payload Synthesis
- Drug Module Synthesis
- Transcription Toxin Synthesis
Transcription Toxin Synthesis Services
As a pioneer in antibody-drug conjugates (ADCs) development, Creative Biolabs has excelled in this field with over a decade of experience. Using our advanced "DrugLnk" organic synthesis platform, our science team provides highly customized ADC design and preparation services using transcription toxin payloads, including amatoxins and thailanstatin A.
mRNAs are common regulators for many oncogenes and mediate several crucial biological processes, including angiogenesis, cell proliferation, and apoptosis. They are the products of gene transcription and they usually exert shorter half-lives. Agents that hinder gene transcription also inhibit mRNA synthesis and subsequently, prevent gene functionality and harm the neoplastic cells. During tumorigenesis, the mutated cells lose their natural growth control mechanisms and depend on the oncogene products for survival and to thrive. Treatments that impede the expression or function of mutated oncogenes have revealed great efficiency in killing the neoplastic cells. Since the initial step in oncogene expression is transcription, agents that prevent mRNA synthesis are considered to be an effective approach for gene expression suppression and offer novel approaches in cancer therapeutics. The most extensively studied agents that target and disturb the transcription process and mRNA biosynthesis are amatoxins and thailanstatin A.
Transcription inhibition
The complexity of gene transcription offers a number of potential drug targets. Therapeutics that target various steps along the progression of transcription have been extensively studied, among which only a few have arrived the clinic stage. As a targeting strategy, agents derived against RNA biosynthesis offer several strategic advantages. Firstly, accumulated evidence has showed that these agents do not cause an extensive cytotoxicity as those DNA-directed agents. Transcription inhibitions take advantage of the distinct sensitivity between normal and tumor cells, as the cytotoxic effects of inhibiting RNA synthesis is minimally observed in normal cells. For example, inhibition of Pol II in normal somatic cells such as mammary epithelial cells, normal lymphocytes, and fibroblasts does not readily lead to apoptosis. Secondly, it has been proven that in normal fibroblast and epithelial cells, transcription inhibitors result in a reversible growth arrest. In the case of tumor cells, on the other hand, transcription inhibition has been demonstrated to largely induce apoptosis. Thus, this unique "selectivity" of transcription inhibition agents makes them appealing agents for cancer therapy.
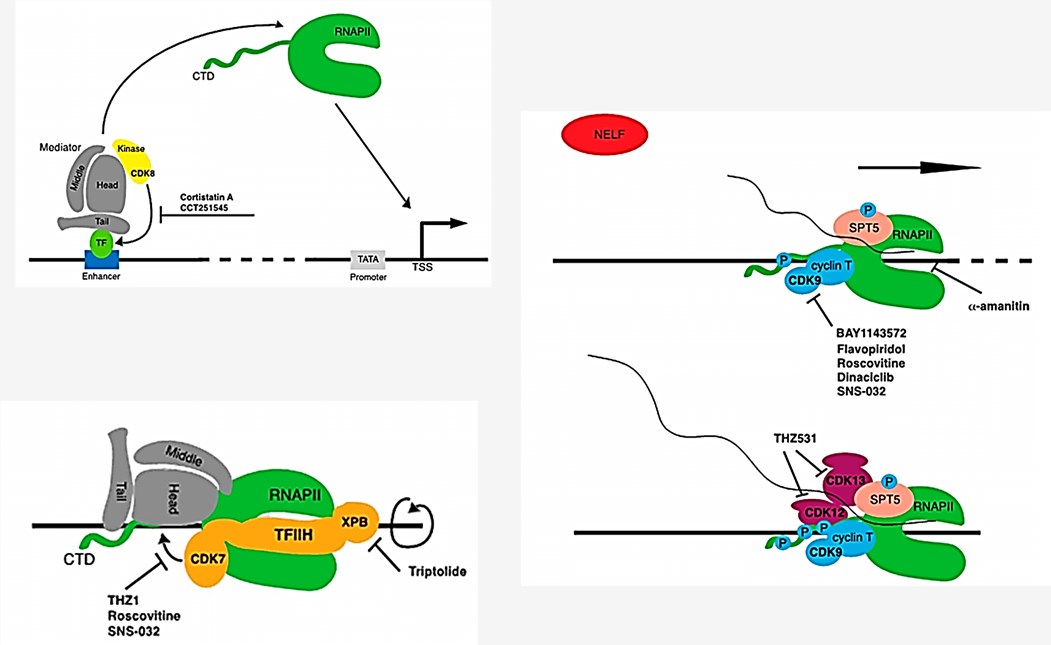 Fig.1 Inhibition of RNAPII transcription.1
Fig.1 Inhibition of RNAPII transcription.1
Spliceosome inhibition
Another target for transcription toxins is the mRNA maturation process. The main machinery for this process is spliceosome and the interruption of its assembly results in mis-processed, non-valid mRNAs and subsequently, impeded gene transcription. Spliceosome inhibitors are a class of potent anti-proliferative agents that target both actively dividing and quiescent cells. These agents bind firmly to the SF3b subunit of the U2 snRNA sub-complex, a crucial component of the spliceosome. Spliceosome inhibitors and their analogs are potential payloads for next generation ADCs and they are currently under active development and evaluation for cancer therapeutics. ADCs conjugated with spliceosome inhibitors, such as thailanstatin A, can selectively kill cancer cells expressing both high and low levels of the target antigen without causing collateral damage to normal tissues. High efficacy against multi-drug resistant (MDR) tumor phenotypes is another advantage for spliceosome inhibitors.
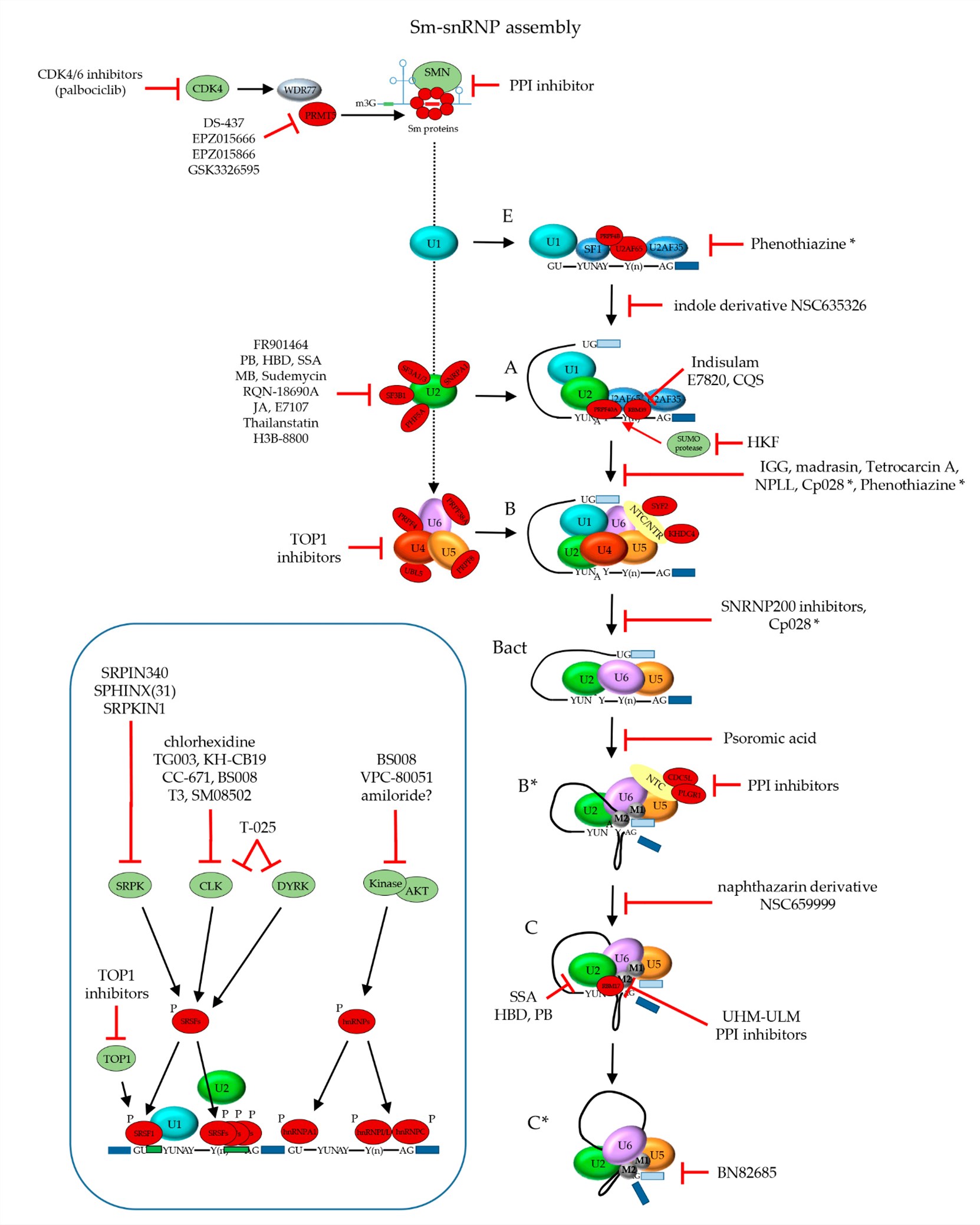 Fig.2 Inhibitors of the mRNA splicing reaction.2,3
Fig.2 Inhibitors of the mRNA splicing reaction.2,3
With our well-established "DrugLnk" organic synthesis platform, the experienced scientists here at Creative Biolabs is dedicated to help our clients develop unique payloads-linker complexes containing transcription toxins. Using readily available or customized linkers, we can promote your progress of the antibody-drug conjugate projects in a timely and cost-effective manner. Our customarily tailored services and high quality products will contribute greatly to the success of your projects.
Creative Biolabs also provides other various services regarding ADC development. Please feel free to contact us for more information and a detailed quote.
References:
- Martin, Ryan D., Terence E. Hébert, and Jason C. Tanny. "Therapeutic targeting of the general RNA polymerase II transcription machinery." International Journal of Molecular Sciences 21.9 (2020): 3354. Distributed under Open Access License CC BY 4.0, remixed a part of Figure 1, figure 2, and figure 3.
- Blijlevens, Maxime, Jing Li, and Victor W. van Beusechem. "Biology of the mRNA splicing machinery and its dysregulation in cancer providing therapeutic opportunities." International Journal of Molecular Sciences 22.10 (2021): 5110.
- Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
