- Home
- ADC Development
- DrugLnk™ Custom Linker-Payload Synthesis
- Drug Module Synthesis
- Microtubule Toxin Synthesis
Microtubule Toxin Synthesis Services
As a next-generation cancer immunotherapeutic strategy, antibody-drug conjugates (ADCs) combines the targeting specificity of a monoclonal antibody with the high toxicity of a payload drug to achieve discriminated cancer cell elimination. Payload drugs disrupt important cellular pathways and result in cell death by apoptosis. Microtubule is a key intracellular target for a variety of payload drugs. With a deep understanding of the payload structure, chemistry, and effects, Creative Biolabs provides customized ADCs development using microtubule toxins as payloads, which include auristatins, epothilone, maytansinoids, taxoids, tubulysins, and vinorelbine.
Microtubules
Microtubules are filamentous intracellular components that form a framework of subcellular structures, for instance the spindle apparatus that arises during cell division…. They are dynamic macromolecules that are involved in a variety of important movements in eukaryotic cells, including chromosomal movement during mitosis, intracellular transportation, organelle motion and organization… Microtubules are constructed from the assembly of heterodimers that comprise of two tubulin isoforms, namely α-tubulin and β-tubulin. Upon the correct cellular signal, tubulin heterodimers assemble into linear protofilaments, which are later assemble into structurally more organized microtubules. Microtubule generation is well-regulated by multiple intracellular regulatory mechanisms, including feedback inhibition, GTP hydrolysis, protein cascade….
Dynamic instability of microtubules
Microtubules are dynamic bio-macromolecules under constant changes during most of the cellular activities. Within a population of microtubules, some are actively growing by the addition of heterodimers to their ends while others undergo shrinking and release the subunits in the form of heterodimers. Thus, microtubule structures are thought to be in a dynamic equilibrium, or a steady state, because of their intrinsic instability. A good representation of the microtubule dynamics is the haploid microtubules—a population that are able to oscillate between the growth and shortening phases. Interruption of this constant dynamics by microtubule-targeting payloads will inevitably disrupt various cellular activities and pathways, resulting in cell death.
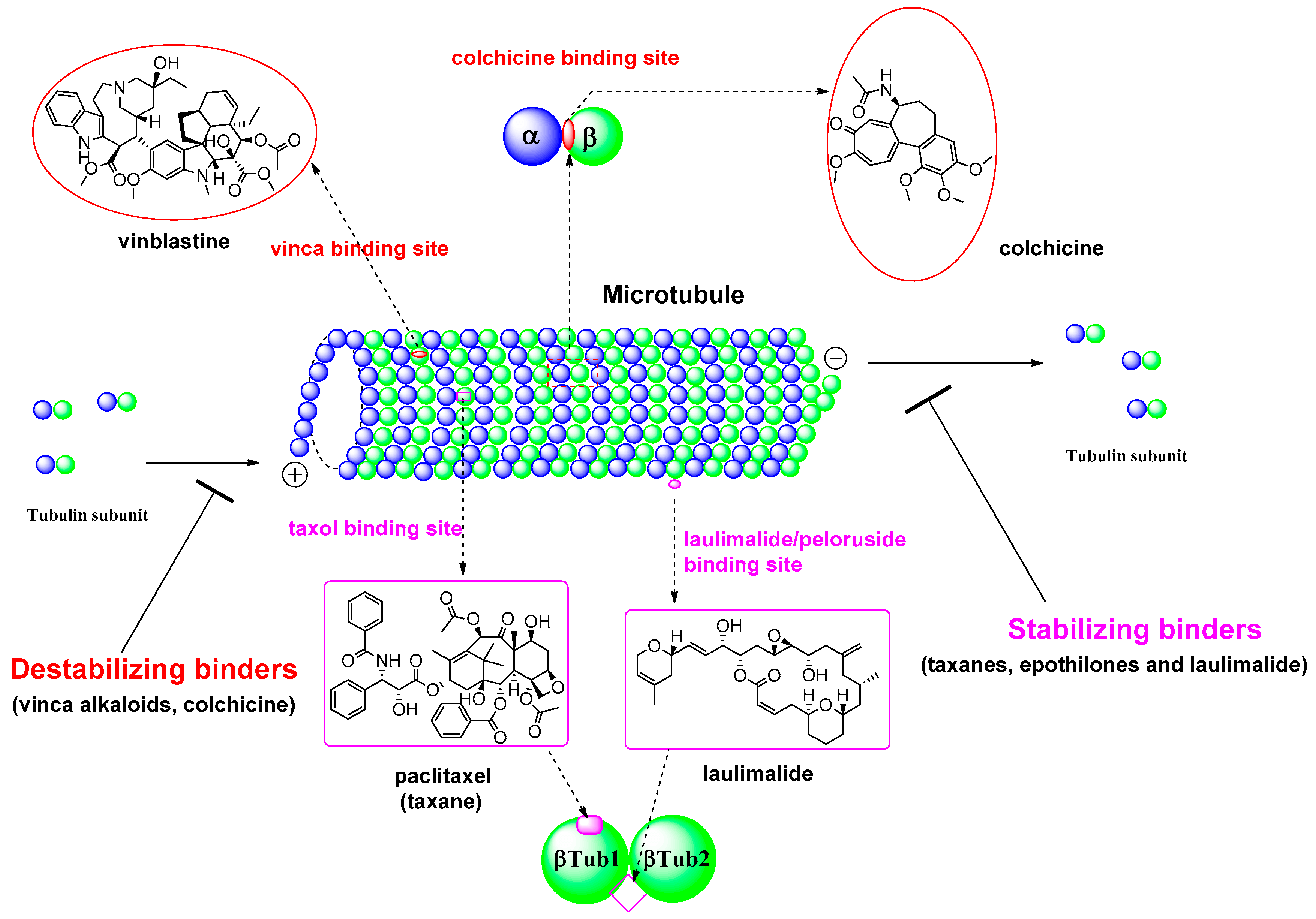 Fig.1 Microtubule formation and the binding sites of microtubule inhibitors.1, 2
Fig.1 Microtubule formation and the binding sites of microtubule inhibitors.1, 2
Microtubule toxin based ADCs
Several ADCs bearing anti-MT (Microtubule) agents have been produced for clinical applications while a large number of new ADCs in this category are under pre-clinical development and evaluation. The toxic moieties used in anti-MT ADCs mainly include auristatins, epothilone, maytansinoids, taxoids, tubulysins, and vinorelbine. These drugs disrupt microtubule function, arrest cell mitosis, and cause cell death via different mechanism of actions. Trastuzumab emtansine (T-DM1), a human epidermal growth factor receptor (HER2)-targeted ADC, is comprised of the Trastuzumab antibody, a small non-cleavable thioether linker, and the cytotoxic payload emtansine (DM1, a maytansine derivative). As one of the two current FDA approved therapeutic ADCs, T-DM1 obtained marketing approval in 2013 under the trade name Kadcyla®. Several other microtubule-targeting payloads based ADCs are under active development.
Advantages of microtubule toxin based ADCs
Microtubules serve as a confirmed oncologic target in a variety of tumor types, with many anti-microtubule cytotoxic drugs approved for cancer treatment. The use of strong microtubule-binding agents is restricted by their high toxicity to normal cells, resulting in severe side-effects including myelosuppression and peripheral neuropathy. The development of ADCs enables the use of those powerful microtubule inhibition agents that would otherwise be too toxic to deploy in their unbound form. Conjugating these agents to monoclonal antibodies could also avoid toxicity to normal tissues and thus improve drug concentration in target cancer tissues.
With our deep understanding of drug and linker chemistry and our advanced synthetic chemistry "DrugLnk" service platform, Creative Biolabs offers high quality customarily tailored ADC development service using microtubule-targeting agents as payloads. Our services include, but are not limited to linker design, synthesis of the drug-linker set, antibody conjugation, and ADC biochemical analysis. Please contact us for more information and a detailed quote.
References:
- Banerjee, Souvik, et al. "Current advances of tubulin inhibitors in nanoparticle drug delivery and vascular disruption/angiogenesis." Molecules 21.11 (2016): 1468.
- Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
