- Home
- Resources
- Featured Solutions
- One-Stop ADC Development Solution
- Antibody Design and Conjugation Solution for ADC Development
A Crucial Step in ADCs: Antibody Conjugation
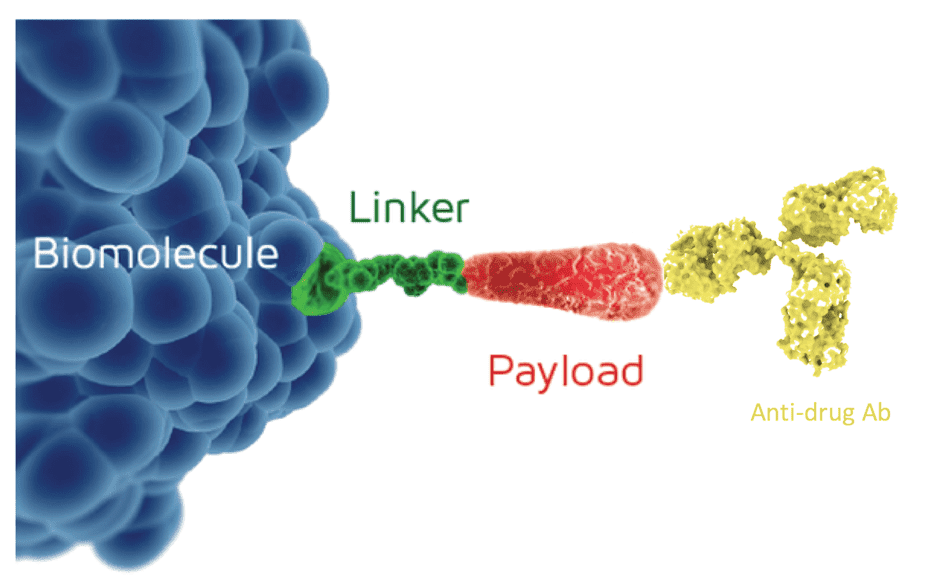
The core of an Antibody-Drug Conjugate (ADC) lies in accurately linking a potent cytotoxic drug (payload) to a monoclonal antibody that targets a specific antigen, via a stable linker. The Antibody Conjugation process is a critical step that determines the success or failure of an ADC. An ideal conjugation strategy must address the following key issues:
Site-specificity
Traditional random conjugation methods (e.g., lysine conjugation) produce a heterogeneous mixture of ADCs with unpredictable pharmacokinetics and efficacy. Site-specific conjugation ensures that the payload is attached to a specific location on the antibody.
Drug-to-Antibody Ratio (DAR)
Precise control over the number of drug molecules attached to each antibody is crucial. A DAR that is too high or too low can adversely affect the ADC's stability, efficacy, and toxicity.
Homogeneity & Stability
A highly homogeneous ADC product is easier to characterize and perform quality control on. The stability of the linker is directly related to whether the drug can remain attached until it reaches the target cells, avoiding off-target toxicity.
Comprehensive Conjugation Strategies
Classical and Amino Acid Residue-based Conjugation Strategies
Creative Biolabs is dedicated to helping clients overcome these challenges through advanced conjugation technologies to build next-generation ADCs with superior efficacy and safety.
-
Lysine-based Conjugation Strategy
Utilizes the abundant lysine residues on the antibody surface for conjugation, a mature and conventional method.
-
Cysteine-based Conjugation Strategy
Achieves relatively defined conjugation sites by reducing inter-chain disulfide bonds or introducing free cysteines.
-
Tyrosine-based Conjugation Strategy
An emerging site-specific conjugation method that utilizes specific tyrosine residues for precise modification.
-
Carbohydrate-based Conjugation Strategy
Targets the glycan chains in the antibody's Fc region for oxidation and conjugation, avoiding interference with the antigen-binding site.
Advanced Enzymatic and Engineered Conjugation Strategies
We employ cutting-edge enzymatic and protein engineering techniques to achieve unparalleled precision and control in ADC manufacturing.
-
EnCys-mAb based Conjugation Strategy
Achieves highly controllable conjugation by introducing a cysteine at a specific site via an enzymatic reaction.
-
Enzyme-mediated Modification & Conjugation Strategy
Uses enzymes like transglutaminase to achieve efficient and specific antibody modification and drug conjugation under mild conditions.
-
Intein-fusion based Conjugation Strategy
An innovative self-splicing technology that can introduce a non-natural amino acid at a specific position for subsequent conjugation.
-
Meditope-based Conjugation Strategy
Utilizes the unique pocket formed between the antibody's Fab arms (the Meditope) for non-covalent, reversible drug conjugation.
Featured Platform
PTLNX™ PT-based Conjugation
Precision & Homogeneity
Achieves precise control over the conjugation site and drug-to-antibody ratio (DAR), yielding well-defined and highly homogeneous ADCs, which simplifies the quality control process.
High Stability & Wider Therapeutic Window
The optimized conjugation technology ensures high stability of the ADC in serum, significantly reducing off-target toxicity, thereby widening the therapeutic window and enhancing drug safety.
Broad Applicability
The platform is flexible and compatible with various antibodies, linkers, and drug payloads, providing powerful technical support for different ADC projects.
Antibody Design and Conjugation Solution Workflow
Design
Preparation
Optimization
Purification
Characterization
Delivery
Consultation & Project Design
In-depth discussion with our expert team to clarify your project goals, target, antibody, and payload information. We tailor the optimal conjugation strategy and experimental plan based on your needs.
Antibody/Payload Preparation & Modification
Perform necessary engineering or modification of your antibody (e.g., introducing cysteine) according to the selected strategy. Synthesize or prepare high-quality linker-drug complexes.
Conjugation Reaction Optimization
Explore and optimize various parameters of the conjugation reaction (e.g., pH, temperature, reaction time, molar ratio) in small-scale experiments.
Scale-up Conjugation & Purification
Perform scale-up conjugation reactions under optimal conditions. Use advanced chromatography techniques (e.g., HIC, SEC) for efficient purification of the resulting ADC, removing unreacted antibodies and drugs.
Comprehensive ADC Characterization
Conduct a full analysis of the ADC's drug-to-antibody ratio (DAR), purity, aggregate content, endotoxin levels, in vitro cytotoxicity, and antigen-binding activity using various analytical methods such as mass spectrometry, HPLC, and ELISA.
Report Delivery & Technical Support
Provide a detailed experimental report, data analysis, and high-quality ADC product. Offer ongoing technical support to address any questions in your subsequent experiments.
Our Advantages
By choosing Creative Biolabs, you will receive:
Extensive Industry Experience
Over a decade of profound experience in the ADC R&D field, with hundreds of successful projects delivered.
Cutting-edge Proprietary Platforms
Access to internationally leading site-specific conjugation technology platforms, such as PTLNX™.
One-Stop Solution
Seamless services covering everything from antibody discovery and engineering to final ADC production and characterization.
Highly Customized Services
Our team of scientists will flexibly adjust plans to meet your specific needs and unique R&D goals.
Strict Quality Control
Adherence to a rigorous quality management system to ensure every batch of product has outstanding quality and consistency.
Our Services Trusted by Leading Researchers









What Our Customers Say About Us
Case Studies
Antibody Internalization Assay
With the success of these case studies, various approaches have been proven helpful in detecting internalization efficiency and are elucidated in this brochure.
- Case 1: Internalization of anti-HER2 mAb-vc-Toxin
- Case 2: Trafficking of anti-HER2 mAb and anti-HER2 mAb-vc-MMAE
- Case 3: Internalization kinetics of anti-Trop mAb-vc-auristatin
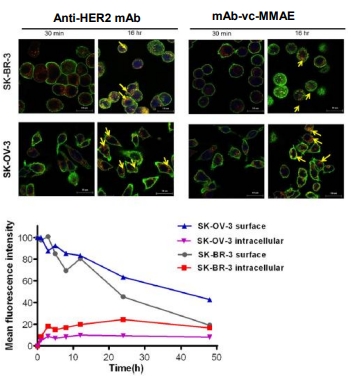
Anti-HER2 ADC Preparation & Potency Evaluation
This case study delves into key aspects such as optimized strategies for HER2 antibody selection, linker-payload conjugation techniques, in vitro and in vivo potency assessment data, and solutions to challenges encountered during development.
- Introduction
- Materials and Methods
- Results
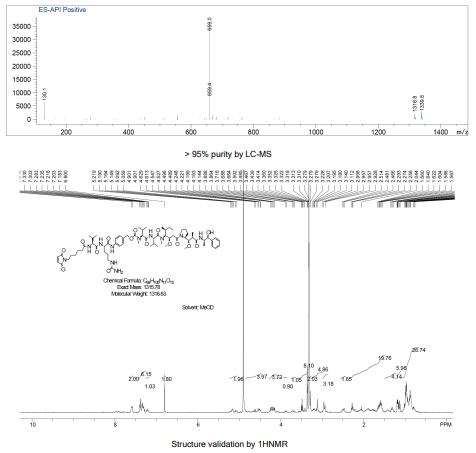
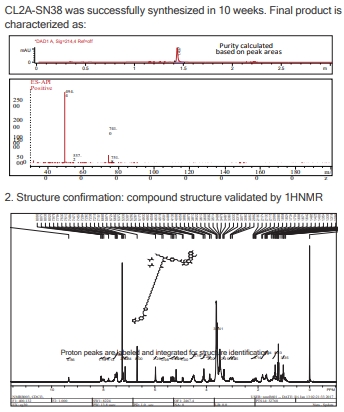
Custom Synthesis of ADC Linker-payload SET
Presented here is a case study for the synthesis of two customized payload-linker complexes via the "DrugLnk" organic synthesis services, ranging from traditional payloads to novel warheads.
- Introduction
- SN38: a novel payload for ADC development
- Case 1: Mc-vc-PAB-SN38
- Case 2: CL2A-SN38
FAQs of Antibody Conjugation Service
How should I choose the most suitable conjugation strategy for my project?
Our expert team will work with you to evaluate your antibody's characteristics, the chemical nature of your payload, and your project goals. For instance, if your antibody lacks available cysteines, we might recommend a lysine-based or enzyme-mediated strategy. We will recommend and validate the best approach for your specific situation.
What materials do I need to provide to start an ADC conjugation project?
Typically, you will need to provide your own monoclonal antibody. For the linker and payload, you can either provide them yourself or commission us to synthesize them. Please contact us to discuss your specific material availability.
How do you ensure the quality and homogeneity of the ADC product?
We ensure high quality and homogeneity by (1) using site-specific conjugation technologies for control from the source, (2) optimizing conjugation reaction conditions, (3) using efficient purification methods, and (4) performing comprehensive analytical characterization (e.g., mass spectrometry for DAR distribution, HIC for hydrophobicity).
How long does it take to complete a standard ADC conjugation and characterization project?
The project timeline depends on its complexity, including the choice of conjugation strategy and whether antibody engineering or payload synthesis is required. A typical project usually takes 6-12 weeks. We will provide you with a detailed timeline at the start of the project.
Can you handle my payload if it is very unique or highly hydrophobic?
Yes. We have extensive experience handling a wide variety of complex payloads. Our scientists will specifically develop and optimize conjugation and purification conditions to address the technical challenges posed by highly hydrophobic or other difficult payloads, minimizing aggregation and maintaining the ADC's solubility.
Featured Products
Featured Resources
For Research Use Only. NOT FOR CLINICAL USE.
Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.