Anti-Glycopeptide Antibody Development Service
Great Partners with Creative Biolabs
Defining Specificity: The Power of Anti-Glycopeptide Antibodies
In the landscape of oncology and infectious disease research, cell surface glycosylation patterns are among the most distinct biomarkers of pathology. Tumor cells frequently display aberrant glycosylation, resulting in the expression of Tumor-Associated Carbohydrate Antigens (TACAs) such as Tn, sTn, and T antigens. However, these glycans are not floating freely; they are covalently attached to specific protein backbones (glycoproteins) like Mucins (MUC1, MUC4, MUC16).
An anti-glycopeptide antibody is engineered to recognize a combined epitope: a specific glycan structure and the adjacent amino acid sequence of the carrier peptide. This dual-recognition capability is critical. Unlike antibodies that bind only to the glycan (which may be expressed on healthy tissues) or only to the peptide (which may be masked by glycosylation), anti-glycopeptide antibodies target the unique junction found only on diseased cells.
Creative Biolabs utilizes a proprietary glyco-optimization platform to overcome the inherent challenges of these targets, delivering antibodies with high affinity and exquisite specificity for research and pre-clinical applications.
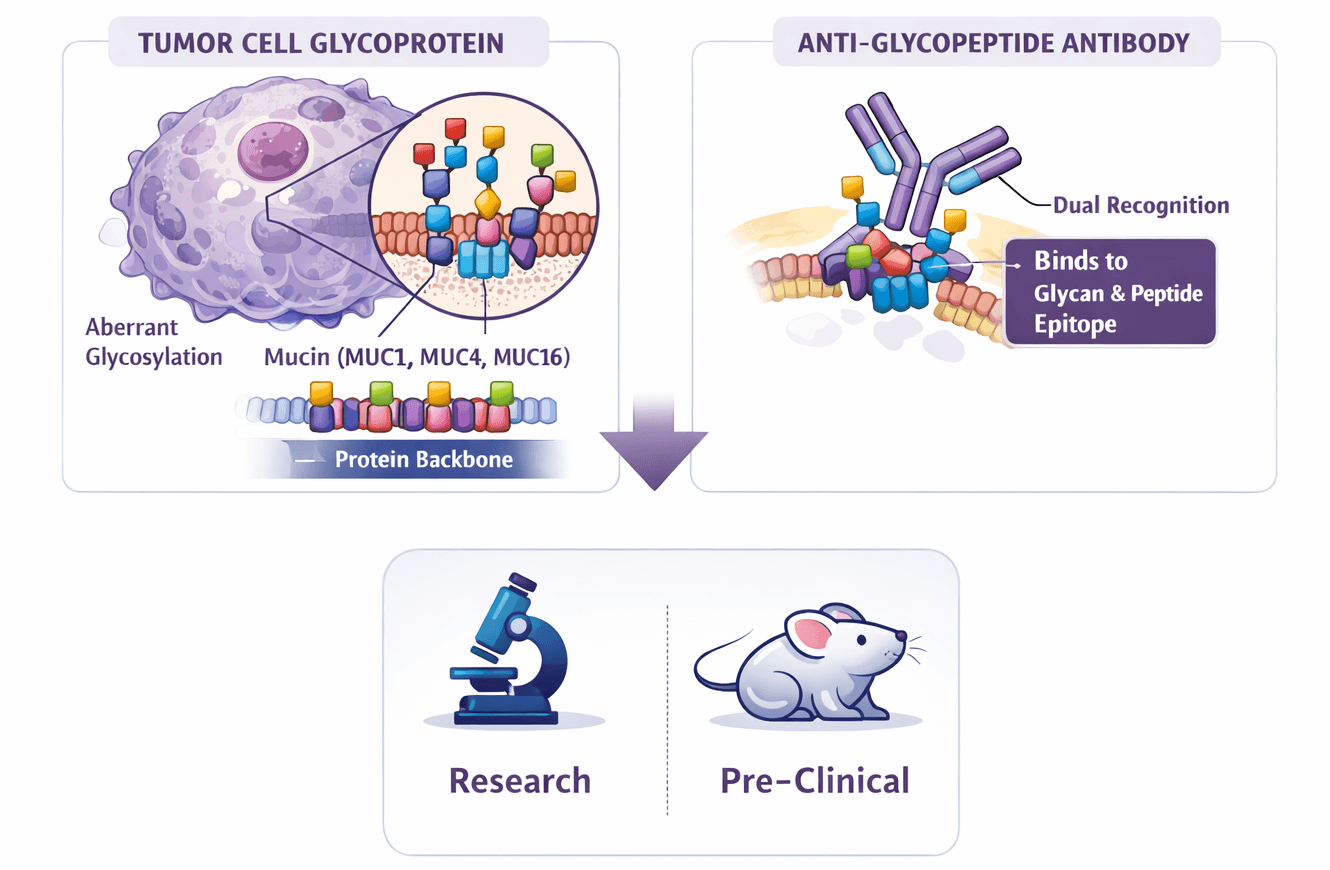
Fig.1 Anti-Glycopeptide Antibodies.
The Immunogenicity Challenge
Developing antibodies against glycopeptides is notoriously difficult due to the unique biological properties of carbohydrates. Researchers often face three primary hurdles:
T-Cell Independence
Carbohydrates are typically T-cell independent antigens. They fail to recruit T-cell help during immunization, often resulting in low-affinity IgM responses rather than high-affinity IgG class switching.
Immunological Tolerance
Many target glycopeptides (like MUC1) are self-antigens. The host immune system possesses strong tolerance mechanisms, actively suppressing the generation of auto-reactive antibodies.
Epitope Masking
The dense glycosylation on mucins can sterically hinder antibody access to the peptide backbone, while the peptide itself can influence the conformation of the glycan, complicating proper immunogen design.
Target-Specific Glycopeptide Antibody Services
We offer specialized antibody generation for high-value mucin targets and custom sequences, unlocking the potential of glycopeptides as precision biomarkers.
Anti-MUC1 Glycopeptide Antibody
Targeting MUC1 VNTR bearing Tn, sTn, or T antigens. Crucial for breast, ovarian, and pancreatic cancer research.
Anti-MUC4 Glycopeptide Antibody
Focusing on aberrantly glycosylated MUC4 domains, a key biomarker in pancreatic adenocarcinoma metastasis.
Anti-MUC16 (CA-125) Antibody
Generating antibodies against glycosylated repeats of CA-125, improving specificity for ovarian cancer.
Anti-MUC5AC Glycopeptide Antibody
Specialized development for MUC5AC, heavily implicated in gastric and colorectal cancer pathogenesis.
Anti-MUC2 Glycopeptide Antibody
Antibodies against intestinal mucin MUC2, essential for studying IBD and mucinous carcinomas.
Anti-PODXL Glycopeptide Antibody
Targeting sialylated glycoforms of Podocalyxin, a marker for aggressive tumor phenotypes.
Anti-CD43 Glycopeptide Antibody
Development of antibodies recognizing specific O-glycoforms of CD43, relevant in leukemia.
Custom Glycopeptide Antibody
From synthesis to screening, we develop antibodies against any novel target provided by you.
Anti-Glycopeptide Antibody Development Workflow
Immunogen Design
Precision synthesis of glycopeptides with defined sites (Tn, T, STn) and carrier protein conjugation.
Immunization
We use tailored adjuvants and host species to break self-tolerance; for challenging targets, phage display expands antibody diversity beyond hybridoma.
Dual Screening
Rigorous counter-selection against naked peptides and free glycans to isolate specific binders.
Validation
Verification via ELISA, FACS (tumor cell binding), and IHC on tissue arrays.
Why Partner with Creative Biolabs?
Synthetic Precision
We utilize synthetic glycopeptides with 100% defined glycosylation sites rather than heterogeneous natural extracts, ensuring the antibody targets the exact structure intended.
Tolerance Breaking
Our proprietary adjuvant systems (including liposomal formulations) are designed to overcome the weak immunogenicity of self-antigens and induce IgG class switching.
Platform Diversity
Choose between classic hybridoma technology for high-affinity IgG production or phage display libraries for discovering binders against difficult or toxic targets.
Stringent Validation
We deliver binders with optimal specificity. Our screening reports include data on negative controls (naked peptide, irrelevant glycan) to prove target specificity.
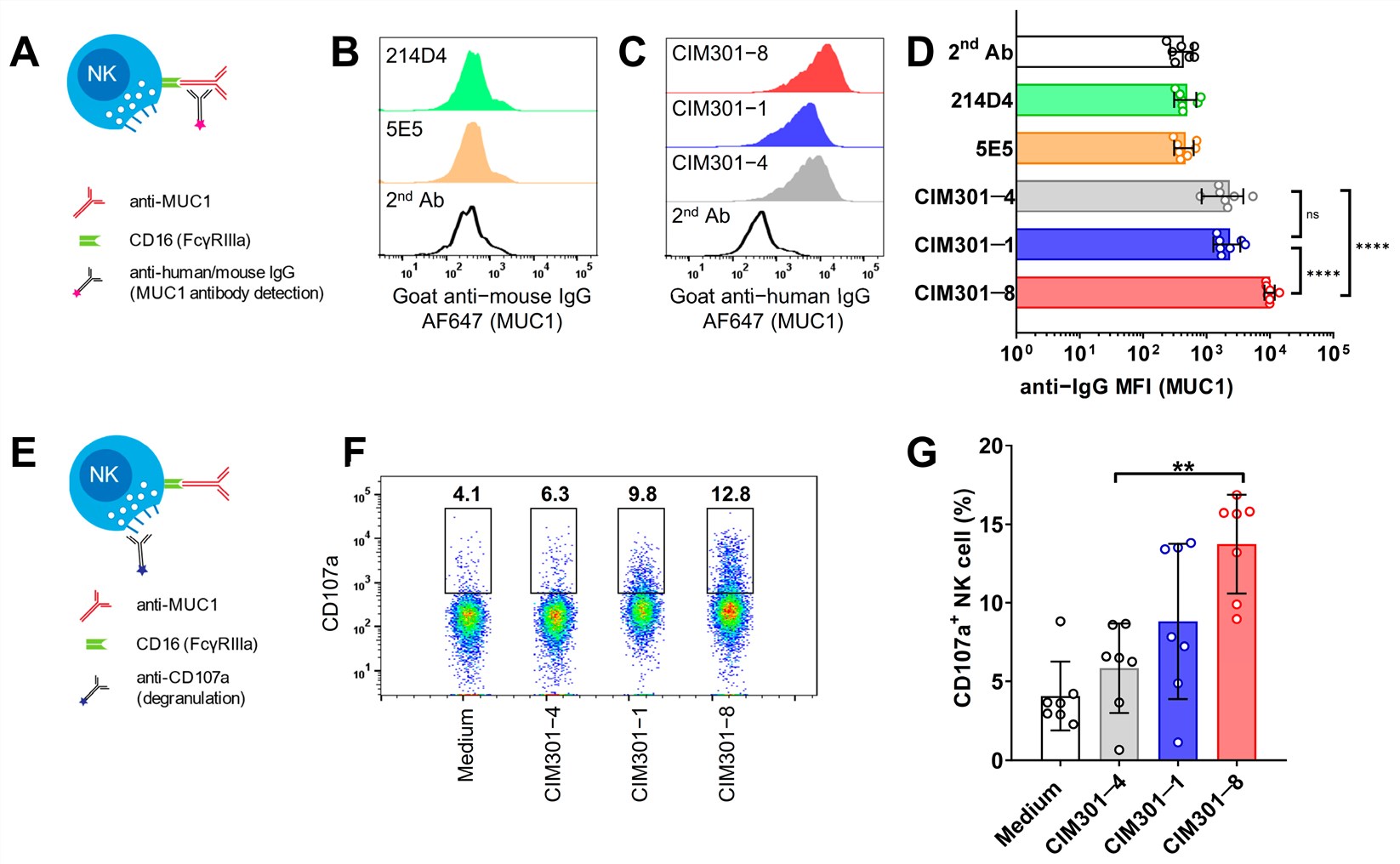
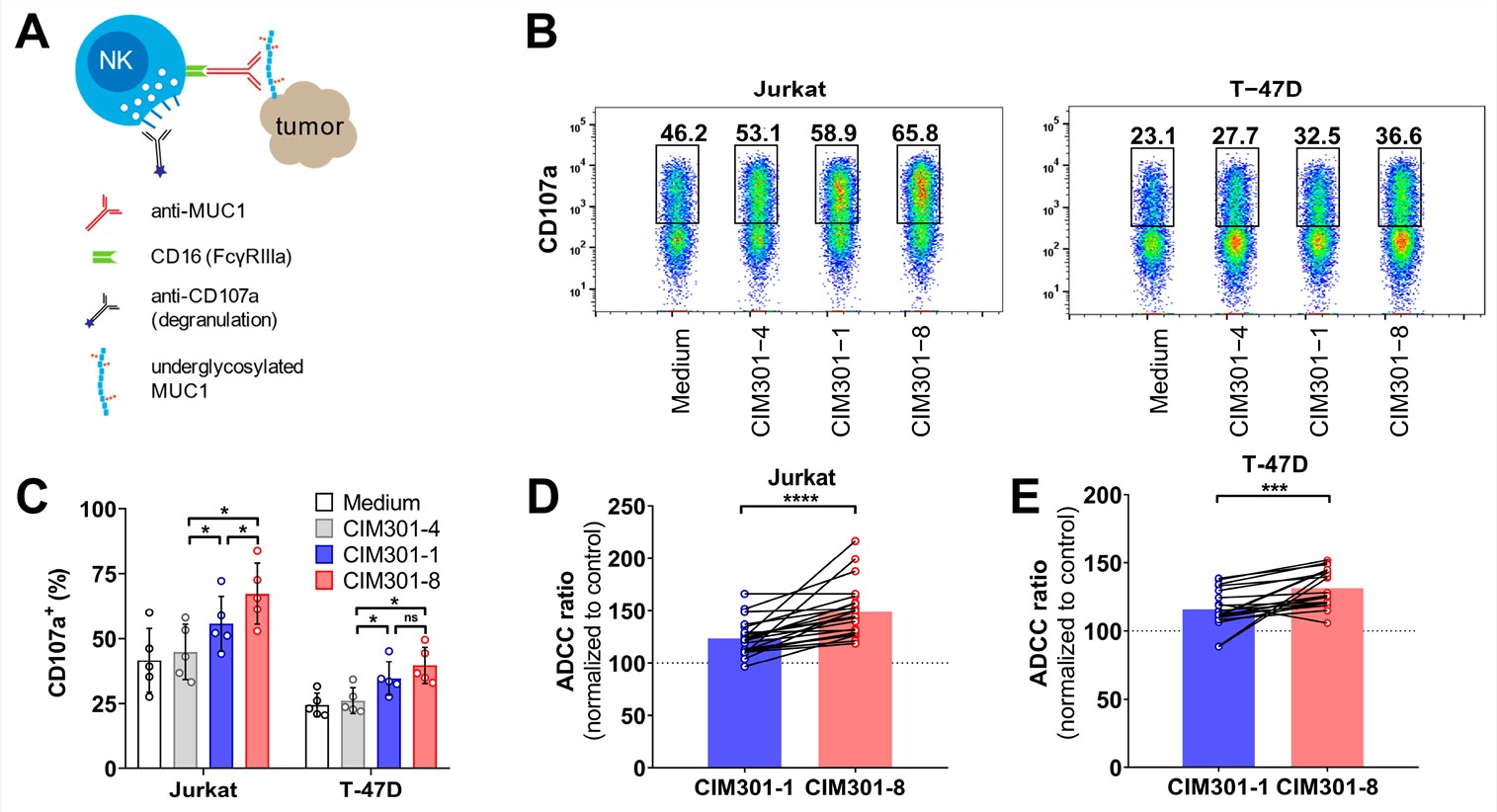
Published Data: Specificity of Humanized Anti-MUC1-Tn Antibodies
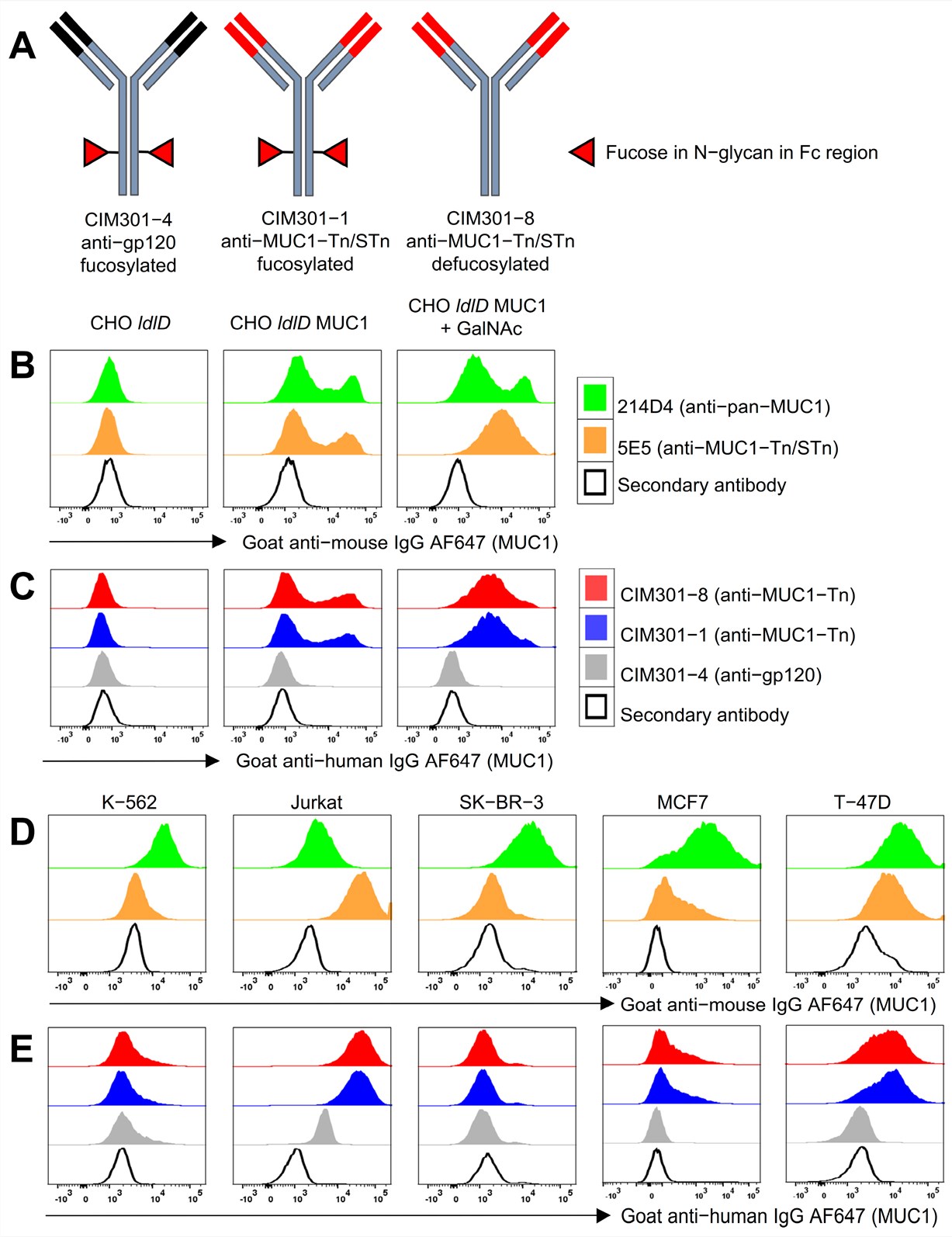
Precise Recognition of Tumor Glycoforms
The humanized antibody, derived from the murine 5E5 clone, exhibits exquisite specificity for the tumor-associated Tn and STn glycoforms of MUC1. Flow cytometric analysis confirms robust binding to MUC1-positive breast (T-47D, MCF7) and leukemia (Jurkat) cancer cell lines, while showing no cross-reactivity with MUC1-negative lines or healthy tissues. Crucially, the antibody distinguishes between aberrant cancer-specific glycosylation and normal mucin profiles, targeting the unique conformational epitope created by the interplay between the peptide backbone and truncated sugar chains. This precise recognition minimizes off-target effects, establishing a safety profile suitable for clinical translation.
Tumor Specificity
High-affinity binding to MUC1-Tn/STn epitopes on Jurkat and T-47D cells.
No Cross-Reactivity
Absence of binding to non-glycosylated MUC1 or healthy epithelial controls.
Frequently Asked Questions
How do you ensure the antibody binds the glycopeptide and not just the glycan?
We employ a stringent counter-screening strategy. After immunization, clones are screened against three targets: the immunizing glycopeptide, the naked peptide, and an irrelevant protein scaffold carrying the same glycan. We select only those clones that bind the glycopeptide but fail to bind the naked peptide or the irrelevant glycosylated scaffold.
What types of glycans can be incorporated into the immunogen?
Our synthetic chemistry capabilities allow us to incorporate a wide range of tumor-associated glycans, including Tn (GalNAc), sTn (Neu5Ac-GalNAc), T (Gal-GalNAc), sT, and core 1/core 2 structures. We can also synthesize longer, complex N-glycans if required for the project.
Can you develop antibodies for targets other than MUC1?
Yes. While MUC1 is the most common target, we have experience with MUC4, MUC16, MUC5AC, Podocalyxin, CD43, and CD44. Furthermore, our Custom Glycopeptide Antibody Development service allows us to target any protein where a specific glycosylation site is known or suspected to be a biomarker.
What host species are available for immunization?
We offer immunization in mice (Balb/c, CD-1), rats, rabbits, and llamas (for VHH development). For projects requiring human therapeutic candidates, we can utilize transgenic humanized mice or employ human synthetic phage display libraries.
What is the typical timeline for a custom project?
A standard murine monoclonal antibody project typically takes 4-6 months. This includes peptide synthesis (4-6 weeks), immunization (8-10 weeks), fusion and screening (4-6 weeks), and subcloning/characterization. Timelines can vary based on the complexity of the synthesis and the immune response.
Do you provide the glycopeptide antigen for our internal use?
Yes, as part of the service, we can synthesize additional quantities of the specific glycopeptide used for immunization and screening, allowing you to use it as a positive control in your downstream assays.
Client Testimonials
References:
- Gong, Ying, et al. "Defucosylation of tumor-specific humanized anti-MUC1 monoclonal antibody enhances NK cell-mediated anti-tumor cell cytotoxicity." Cancers 13.11 (2021): 2579. Distributed under Open Access license CC BY 4.0. https://doi.org/10.3390/cancers13112579
Featured Products
- Anti-MUC5AC Glycopeptide Antibody Development
- Anti-MUC1 Glycopeptide Antibody Development
- Anti-MUC4 Glycopeptide Antibody Development
- Anti-MUC16 (CA-125) Glycopeptide Antibody Development
- Anti-MUC2 Glycopeptide Antibody Development
- Anti-Podocalyxin (PODXL) Glycopeptide Antibody Development
- Anti-CD43 Glycopeptide Antibody Development
- Custom Glycopeptide Target Antibody Development