Anti-Glycosaminoglycan (GAG) Antibody Development Service
Great Partners with Creative Biolabs
Background Introduction
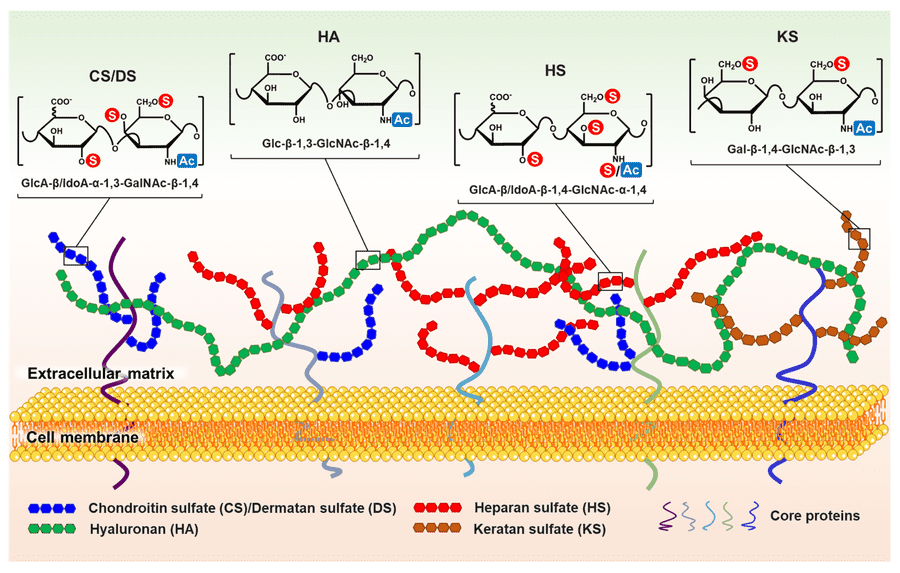
Glycosaminoglycans (GAGs) are linear, highly anionic polysaccharides composed of repeating disaccharide units. They occur either as free polymers (hyaluronic acid) or covalently attached to core proteins as proteoglycans, forming an essential part of the cell surface and extracellular matrix architecture. Structurally, mammalian GAGs are generally classified into:
- Heparan sulfate (HS)
- Chondroitin sulfate (CS)
- Dermatan sulfate (DS)
- Keratan sulfate (KS)
- Hyaluronic acid (HA)
Unlike template-driven biomolecules such as DNA or proteins, GAGs are synthesized through non-template enzymatic pathways, resulting in remarkable structural heterogeneity. Variations in sugar composition, sulfation pattern, epimerization, chain length, and spatial presentation can create distinct or selective binding interfaces for growth factors, cytokines, chemokines, morphogens, and extracellular enzymes. Through these interactions, GAGs regulate:
- Growth factor signaling (FGF, VEGF, HGF, Wnt)
- Chemokine gradient formation
- Cell adhesion and migration
- Angiogenesis
- Immune cell trafficking
- Tissue repair and regeneration
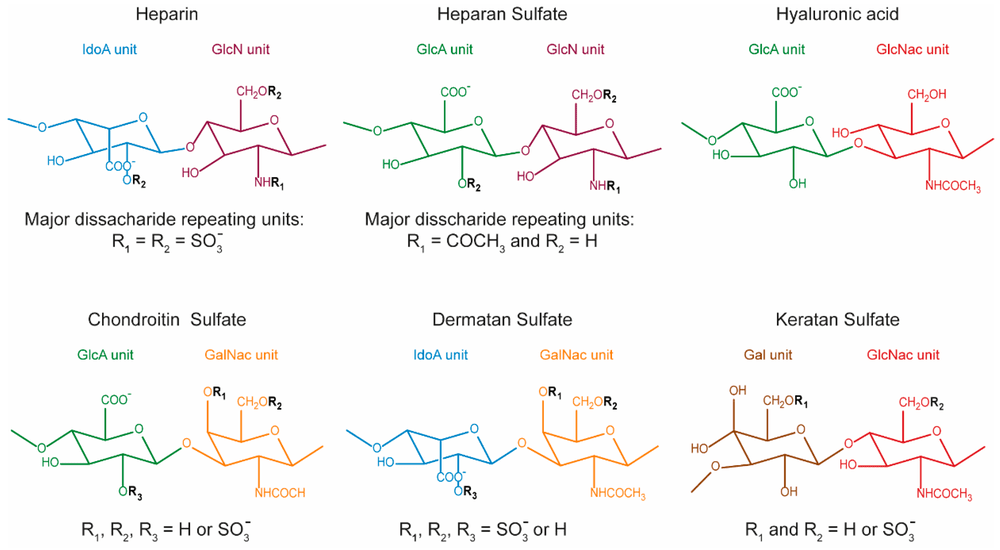
Fig.1 Glycosaminoglycans (GAGs) Structures.1
Why Anti-GAG Antibody Development Is Challenging
Developing antibodies against glycosaminoglycans presents challenges that are very different from protein antibody programs. For each of these hurdles, we detail how we provide a solution.
Specificity Challenges
GAGs share similar disaccharide backbones, and their key differences lie mainly in sulfation position and density. This often leads to HS/CS cross-reactivity or antibodies that bind broad polysaccharide regions rather than defined sulfation domains, limiting mechanistic clarity.
Our approach:
- Construct immunogens around defined sulfation motifs, not bulk polysaccharides
- Perform early cross-reactivity profiling across HS, CS, DS and related glycans
- Use structurally characterized GAG panels to map binding selectivity
- Apply orthogonal validation to confirm motif dependence
- Match specificity testing to intended applications before lead selection
Immunogenicity Barriers
GAGs are endogenous and conserved, making them weak immunogens. Traditional immunization may bias discovery toward low-affinity clones or carrier-dominant responses instead of the intended carbohydrate epitope.
Our approach:
- Enrich target sulfation domains within structurally optimized fragments
- Apply controlled conjugation to preserve epitope exposure
- Incorporate mimetic designs to enhance motif-focused immune recognition
- Integrate display-based selection to expand diversity and affinity space
- Enable advanced host systems when higher affinity maturation is required
Structural Complexity and Antigen Design
GAG chains are heterogeneous, flexible, and structurally dynamic. Poor control of sulfation pattern, chain length, or conjugation chemistry can produce antibodies that fail in native biological systems.
Our approach:
- Control or define sulfation pattern and chain length during antigen preparation whenever feasible
- Minimize structural masking through optimized conjugation strategies
- Drive screening by motif resolution rather than bulk binding strength
- Validate performance in native or tissue-relevant assay systems
Development Timeline and Risk
Hybridoma-only approaches may require repeated immunizations with uncertain outcomes, increasing budget and timeline risk. Later-stage translation may also be limited by affinity, specificity, or scalability issues.
Our approach:
- Deploy multi-platform discovery when appropriate to reduce epitope bias
- Introduce early affinity and specificity checkpoints
- Eliminate broadly reactive clones at pre-selection stages
- Support downstream engineering and scalable production readiness
Target-Specific Anti-GAG Antibody Development Services
Anti-Heparan Sulfate (HS) Antibody
Custom monoclonal or recombinant antibodies targeting HS motifs for use in mechanistic studies, imaging, or research evaluation of HS–growth factor blockade potential where appropriate.
Anti-Chondroitin Sulfate (CS) Antibody
Antibodies against total CS or specific subtypes (e.g., CS-A, CS-C, CS-D, CS-E) to dissect CS roles in tumor progression, neurobiology, and ECM remodeling.
Anti-Dermatan Sulfate (DS) Antibody
DS-focused antibodies for studying fibrosis, wound healing, and disease-associated CS/DS remodeling.
Anti-Keratan Sulfate (KS) Antibody
Antibodies recognizing KS in cartilage, cornea, and nervous system, supporting research in osteoarthritis, corneal disease, and neurodegeneration.
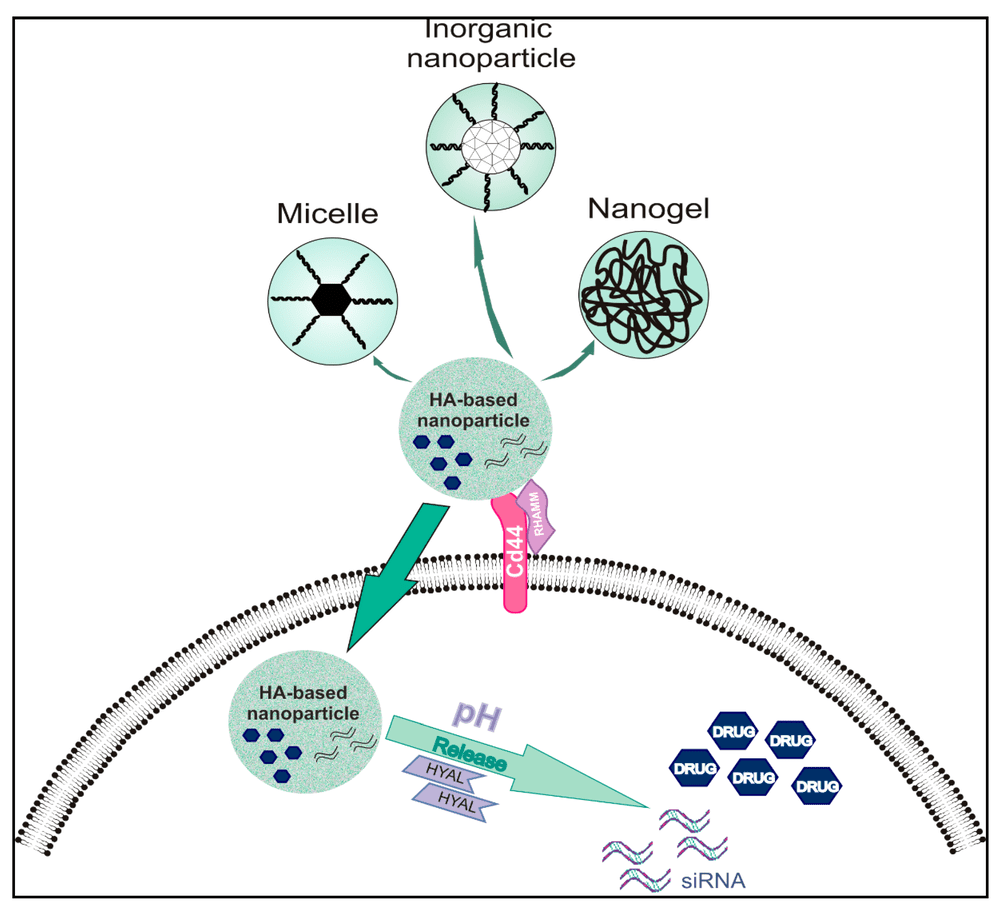
Anti-Hyaluronic Acid (HA) Antibody
HA-binding antibodies to visualize HA-rich matrices, interrogate CD44/RHAMM–HA axes, or support biomarker exploration and research stratification studies based on HA accumulation.
Anti-GAG Sulfation Motif Antibody
Motif-specific antibodies engineered to discriminate fine sulfation patterns within HS or CS/DS domains—ideal for mechanistic mapping.
Tumor-Associated GAG Antibody
Antibodies selectively targeting tumor-enriched GAG patterns (e.g., highly sulfated HS or HA-rich stroma) to support early-stage target validation for imaging or advanced therapeutic concept evaluation.
Need a customized anti-GAG strategy?
Tell us your target GAG and intended application. We will design a decision-driven antibody development roadmap.
Multi-Platform Discovery and Engineering
To address the intrinsic challenges of GAG antigens, we deploy multiple antibody discovery platforms selected according to antigen characteristics and project goals. Platform choice is driven by antigen nature, motif resolution requirements, intended application, and available evidence—not by a fixed workflow.
Available approaches include:
- Hybridoma immunization using chemically defined fragments or neo-epitope mimetics
- Phage or yeast display libraries to enrich rare motif-selective clones
- Humanized or transgenic rodent systems available upon project scope
Lead candidates may undergo CDR refinement, affinity maturation, format optimization, and additional developability review when justified by project scope.
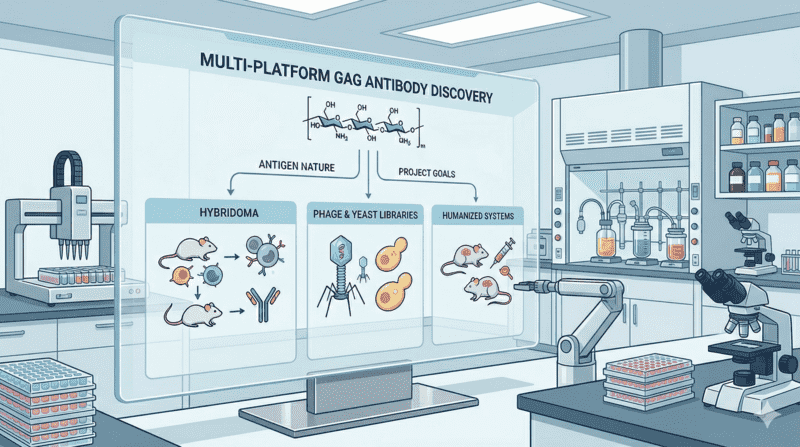
Fig.2 Multi-Platform Discovery and Engineering Workflow for Anti-GAG Antibody Development.
How the Glycosaminoglycan (GAG) Microarray Platform Strengthens Anti-GAG Projects
Our Glycosaminoglycan (GAG) Microarray platform is designed to support antigen definition, motif-level specificity assessment, and high-throughput screening decisions across structurally related GAG classes. Rather than relying on late-stage confirmation alone, the platform helps position specificity analysis as an early design variable.
| Platform Dimension | What the Platform Enables | Why It Matters for Anti-GAG Antibody Development |
|---|---|---|
| Broad GAG Coverage |
Parallel evaluation across multiple GAG classes, sulfation states, and related structures
|
Helps reveal cross-reactivity patterns early when distinguishing HS, CS, DS, KS, HA, or defined motif subsets
|
| Motif-Level Resolution |
Comparative binding analysis against defined sulfation motifs and structurally characterized fragments
|
Supports selection of antibodies directed to disease-relevant domain features instead of bulk polysaccharide recognition alone
|
| High-Throughput Specificity Profiling |
Rapid ranking of binders, counter-binders, and off-target interactions in one experimental format
|
Improves go/no-go decisions before resource-intensive downstream validation, reformatting, or scale-up steps
|
| Project Utility Beyond Screening |
Applicable to antigen feasibility assessment, clone characterization, and reagent validation packages
|
Creates a stronger evidence chain between antigen design, specificity control, and fit-for-purpose downstream use
|
Target Applications & Use Cases
Supporting breakthrough research across oncology, neuroscience, and inflammatory diseases.
Cancer Immunotherapy & TME Remodeling
Tumors frequently remodel HA, CS/DS, and HS landscapes, generating altered sulfation domains that support angiogenesis, immune evasion, and metastatic progression.
- Spatial mapping of tumor-restricted GAG patterns
- Functional blockade of HS–growth factor or HA–CD44 axes
- Patient stratification based on GAG enrichment
Neurodegenerative Disease & CNS Repair
CS/DS and HS remodeling shapes neuronal plasticity and glial scar formation. Disease-associated sulfation signatures have been implicated in neurodegenerative disease and other CNS pathologies.
- Visualization of perineuronal nets
- Motif-specific mapping of disease-associated GAG remodeling
- Functional studies of GAG–protein aggregation interfaces
Inflammation, Fibrosis & Autoimmune
GAG remodeling accompanies chronic inflammation, fibrotic injury, and metabolic complications.
- Monitoring HA or HS shifts during inflammatory progression
- Characterizing CS/DS enrichment in fibrotic lesions
- Developing biomarker capture reagents for exploratory serum or urine GAG profiling workflows
Comprehensive Project Deliverables
Each Anti-GAG Antibody Development project is supported by robust data and high-quality reagents to enable fit-for-purpose downstream application.

We encourage you to contact Creative Biolabs for a tailored consultation. Our scientific team will review your project goals, discuss feasible strategies and milestones, and provide a detailed quotation with timelines, deliverables, and optional add-ons. Actual deliverables depend on antigen definition, screening outcome, selected platform, and intended application.
Get Detailed Quotation →Published Data: GAG Remodeling and Target Evaluation
Disease-associated remodeling of glycosaminoglycans (GAGs), particularly at the level of sulfation motifs and domain organization, has been extensively documented in oncology and other pathologies. These observations support the research value of developing precision anti-GAG antibodies for mapping, selectivity analysis, and target evaluation.
Qingchi Wang and Lianli Chi provide a disease-oriented overview of glycosaminoglycan (GAG) remodeling across human pathology, including atherosclerosis, cancer, diabetes, neurodegeneration, and viral infection.
GAGs—heparan sulfate (HS), chondroitin/dermatan sulfate (CS/DS), keratan sulfate (KS), and hyaluronan (HA)—are described as highly diverse linear polysaccharides whose expression level, chain length, and sulfation pattern are closely associated with disease context and tissue type. The authors summarize shifts such as increased CS/DS content and altered 6-O/4-O sulfation in atherosclerotic vessels, as well as tumor-associated changes in HS and CS/CSPG expression with distinct sulfation signatures.
Implication for Antibody Design:
These findings underscore the value of distinguishing specific sulfation positions and domain features when developing research antibodies, rather than relying only on total GAG abundance.
Advance Your GAG-Targeted Program
Each glycosaminoglycan-targeting antibody program differs in structural complexity, motif specificity requirements, and translational scope, we provide project-specific strategy design and quotation.
We will design a decision-driven antibody development roadmap aligned with your objectives.
Contact Creative Biolabs Today →Frequently Asked Questions
How do you control specificity between closely related GAGs such as HS and CS/DS?
We manage specificity at three levels: antigen design, screening, and validation. First, we choose immunogens that emphasize motif features unique to your target (e.g., particular HS sulfation domains vs CS/DS patterns), guided by published structure–function data. Second, we screen against a panel that always includes the main off-target GAGs you want to avoid. Finally, we confirm specificity in cell and tissue models where the expression of HS, CS/DS, KS and HA is well characterized or experimentally manipulated. Any clone showing unacceptable cross-reactivity is deprioritized before advanced characterization.
Can you generate antibodies that recognize a defined sulfation motif rather than total GAG content?
Yes. Our Anti-GAG Sulfation Motif Antibody Development Service is specifically designed for that goal. We work with short, well-defined GAG fragments or mimetics enriched for your motif of interest, often supported by structural or microarray data indicating which sulfation pattern is functionally critical. We then design screening and counter-screening to maximize the chance of obtaining motif-selective clones.
What types of applications can your anti-GAG antibodies support?
Depending on your chosen configuration and validation package, our antibodies can be used in:
- ELISA and other plate-based assays
- Flow cytometry and imaging (IF/IHC)
- Immunoprecipitation of GAG–protein complexes
- Functional blocking or agonist assays in vitro
- Preclinical target evaluation for imaging agents or other advanced therapeutic concepts
We will discuss your intended applications early in the project so that assay conditions and validation endpoints are aligned from the start.
How do you address the risk that antibodies will bind to normal tissues and compromise therapeutic potential?
For programs with translational aspirations, we incorporate risk assessment and mitigation directly into the design. We prioritize tumor-associated or disease-enriched GAG patterns rather than ubiquitously expressed motifs. Early tissue panel screening can be included to identify problematic cross-reactivity. Where necessary, we can bias selection toward epitopes that depend on combinations of GAG pattern and co-expressed markers, such as proteoglycan context. These strategies help identify antibodies with more selective research profiles before any downstream translational decisions are made.
Can you help if I only have preliminary evidence that GAGs are involved in my system, but no clear target defined yet?
Absolutely. Many of our collaborations start with broad observations such as “HS seems upregulated” or “HA accumulates in late-stage disease.” We can help you interpret existing omics, histology or functional data, propose candidate GAG targets and motifs, and design a phased project: initial exploratory antibodies for mapping, followed by more focused motif-specific or tumor-associated GAG antibodies as your understanding deepens.
How long does a typical Anti-GAG Antibody project take from start to lead candidate?
Timelines depend on several variables, including the discovery platform, number of lead candidates desired, and extent of functional characterization and additional downstream review. When you contact us, we will provide a realistic timeline estimate tailored to your selected workflow and milestones, along with options for accelerated phases where feasible.
Can you support downstream scale-up and manufacturing of selected GAG-targeting antibodies?
Yes. Once you have selected your preferred candidates, Creative Biolabs can assist with recombinant reformatting, stable expression support, and additional characterization as defined by project scope. This allows you to move from initial concept to the next research stage with a single integrated partner.
What sample types can you support for anti-GAG antibody validation?
We routinely validate antibodies in tissue sections, cultured cells, serum, plasma, urine, and extracellular matrix-enriched preparations. For biomarker-oriented programs, we can design validation panels aligned with your intended research or preclinical sample type.
Customer Testimonials
Support Resources
GAG-focused projects often raise cross-cutting questions that extend beyond a single antibody campaign. Creative Biolabs offers additional support to help you build a coherent, long-term strategy around GAG biology:
References
- Neves, Mariana I., Marco Araújo, Lorenzo Moroni, Ricardo M. P. da Silva, and Cristina C. Barrias. Glycosaminoglycan-Inspired Biomaterials for the Development of Bioactive Hydrogel Networks. Molecules 25.4 (2020): 978. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/molecules25040978
- Wang, Qingchi, and Lianli Chi. The Alterations and Roles of Glycosaminoglycans in Human Diseases. Polymers 14.22 (2022): 5014. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/polym14225014
- Berdiaki, Aikaterini, Monica Neagu, Eirini-Maria Giatagana, Andrey Kuskov, Aristidis M. Tsatsakis, George N. Tzanakakis, and Dragana Nikitovic. Glycosaminoglycans: Carriers and Targets for Tailored Anti-Cancer Therapy. Biomolecules 11.3 (2021): 395. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/biom11030395
- Anti-Heparan Sulfate (HS) Antibody Development
- Anti-Chondroitin Sulfate (CS) Antibody Development
- Anti-Dermatan Sulfate (DS) Antibody Development
- Anti-Keratan Sulfate (KS) Antibody Development
- Anti-Hyaluronic Acid (HA) Antibody Development
- Anti-GAG Sulfation Motif (Neo-epitope) Antibody Development
- Tumor-Associated GAG Antibody Development