Bispecific antibody (BsAb) is one of the antibodies with two or more binding sites directed at different antigens simultaneously, enabling therapeutic strategies not possible with traditional monoclonal antibodies. Many bispecific design modalities have been evaluated since BsAb is regarded as a promising agent. Here, Creative Biolabs would like to introduce our high-efficiency cell-free bispecific antibody expression systems, which produce various bispecific antibody products in innovative ways and continue to offer considerable utility particularly in functional proteomics. And the development of higher yield expression systems has expanded their applications in many fields.
Introduction
Cell-free protein synthesis, also termed in vitro translation, facilitates the production of a target protein by translational machinery without relying on living cells. As such, protein or antibody synthesis is disconnected from cell context and the synthesis reaction itself will not be constrained by a cell wall, indicating that reaction conditions are accessible from the outside of the container.
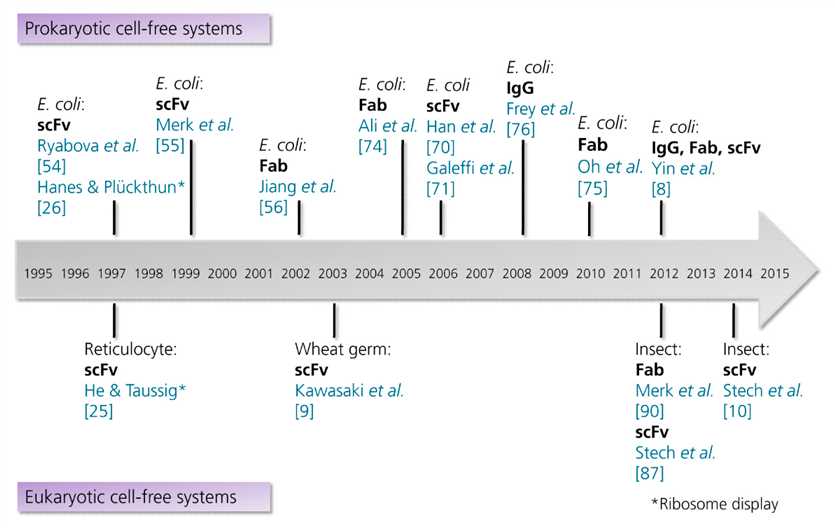
Fig.1 Timeline showing the milestones in the cell-free synthesis of antibody fragments and antibodies. (Marlitt, 2015)
Cell-free system was originally designed as a tool to elucidate the genetic code. Nowadays, this method has proven its potential to employ as a reliable, versatile and flexible protein production platform, ensuring the synthesis of difficult-to-express proteins, such as membrane proteins and even toxic proteins. Cell-free protein synthesis is scalable from the microliter to liter scale, which has been successfully used for the high-throughput production of protein libraries and for the high-yield manufacture of given target proteins.
Applications
BsAbs have emerged in recent years for therapies in oncology, infectious diseases, and inflammatory diseases. Their capability of dual target recognition permits new therapeutic hypotheses to be tested. Among extremely diverse architectures of BsAbs, knobs-into-holes (KIH) technology has been known applied widely, that involves engineering CH3 domains to form either a "knob" or "hole" in every heavy chain to promote heterodimerization. The current report described the usage of a cell-free expression system to generate KIH BsAbs in multiple scaffolds.
Cell-free BsAb Expression Services at Creative Biolabs
Cell-based production techniques of antibodies and antibody fragments are well-established, allowing scientists to invent high-specific molecular recognition tools as for example hybridoma technology and prokaryotic or mammalian cells. However, these techniques are accompanied by the drawbacks of being rather cost-intensive and time-consuming, thus resulting in the development of strong and powerful cell-free protein synthesis systems over the last decade as alternatives.
There’re two categories of cell-free expression systems. So far, prokaryotic in vitro translation cell-free systems have been the hotspot of interest. Recently, eukaryotic cell-free systems have enriched the antibody production pipeline, as the reactions can mimic the natural pathway of antibody synthesis within eukaryotic cells. At Creative Biolabs, we make use of advanced cell-free expression systems for the synthesis of various BsAbs and many other antibody fragments of varying origins, classes and properties.
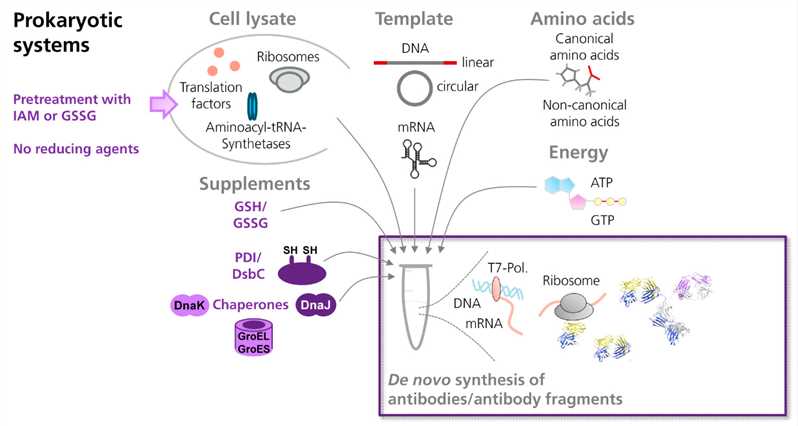
Fig.2 Scheme showing the cell-free synthesis of antibodies and antibody fragments in prokaryotic in vitro translation systems. (Marlitt, 2015)
-
Prokaryotic Cell-Free Systems
Prokaryotic cell extracts are the prominent source of recombinant antibody formats as well as full-length antibody modalities. Creative Biolabs can provide E. coli-based cell-free translation system, which is the simplest system for the synthesis of soluble full-length proteins and high-yield production of prokaryotic proteins, for required BsAb products. In addition, we’re able to perform laborious purification steps to refrain BsAbs from contaminated with endotoxins in E. coli.
-
Eukaryotic Cell-Free Systems
Eukaryotic cell-free systems have been used to synthesize functional disulfide bonded proteins such as Fab and scFv antibody fragments. At Creative Biolabs, we intend to offer a more suitable environment for the folding of complex antibody molecules, that includes but not limited to rabbit reticulocyte lysate (RRL), wheat germ lysate, insect cell lysate, and human cell lysate. Of them, RRL is one of the most popularly applied eukaryotic cell-free reactions. Notably, this system provides post-translational modifications (e.g. core glycosylation or phosphorylation), and natural membrane elements (e.g. microsomes) for membrane protein insertion.
Our Highlights
-
It is an open system, in which many components such as chaperones, reducing/oxidizing elements, labeled amino acids, or detergents) can be added to the reaction mixture and then directly used in applications like NMR;
-
Cell-free systems can often eliminate other effects in living cells such as protease digest or DNA template instability since responsible cellular components have been removed;
-
Multimeric complexes can be expressed and the ratios of different subunits affected by DNA template titration.
BsAbs are important potential tools in diagnostics, therapy, and research applications. Increasingly, they become the key detection elements for biological targets. Creative Biolabs is committed to an ultimate solution of cell-free bispecific antibody production for clients worldwide. Our service can be tailored based on your special requirements. If you have any issues, please contact us for more details.
Reference
-
Stech, Marlitt, and Stefan Kubick. "Cell-free synthesis meets antibody production: a review." Antibodies 4.1 (2015): 12-33. (Under open access license CC BY 4.0, without modification.)
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY






