Creative Biolabs provides the high quality CBD phage display library construction service by the innovative Hi-Affi™ phage display platform. This powerful platform is specific developed for generating scaffold protein with high affinity and diversity.
CBD, known as the C-terminal cellulose-binding domain of cellobiohydrolase I from the fungus Trichoderma reesei, is independently folding domain required for efficient binding and hydrolysis of solid substrates. It is a member of the knottin family which is a kind of small, disulphide-bonded proteins that bind with high specificity to their target molecules. The CBD is a 36 amino acids wedge-shaped protein retained cellulose binding activity, one rounded face is hydrophobic, another face is flat and hydrophilic which mediates the cellulose binding ability of the protein. Relevant three-dimensional structure analysis of CBD indicates the protein is formed by triple-stranded, anti-parallel β-sheets and stabilized by disulfide bridges.
Selecting a specific CBD scaffold is generally based on the construction of phage display library. The variation is introduced by randomized the residues that form the flat face of CBD which functional binding cellulose and then displayed this repertoire on the surface of M13 filamentous phage. The library is then selected against cellulose to isolate novel CBD scaffold with either original cellulose binding activity or other different specificity. Consequently, the engineered CBD scaffold can be selected to against different macromolecular targets and provide greater structural diversity in the primary repertoire.
Hi-Affi™ phage display platform is an advanced proprietary technology of Creative Biolabs. On the basis of common phage display technology, which is an exogenous gene expression method through fusing the target genes to bacteriophage coat proteins then displaying on the phage surfaces to select specific binders, our Hi-AffiTM phage display platform also integrates trimer codon technology and NNK method together to introduce more randomicity and make it more suitable for sorting and isolating the high affinity protein or peptide targets. As a result, our scientists can finally generate the CBD phage display library with the best quality, 100% precise mutant and over 1010 diversity.
As one of the industry leader in scaffold protein library field, Creative Biolabs has extensive experience to tailor the best products for customers’ satisfaction. Up to now, our scientists have generated more than 50 kinds of scaffold libraries for clients all over the world. We commit to offer unique scaffold library with strictly controlled quality and meet each specific demand from our brilliant clients.
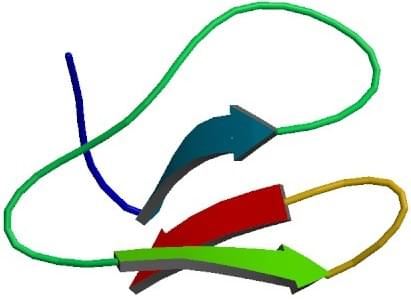
Fig. 1 Cellulose binding domain from cellobiohydrolase CeI7A. (PDB ID: 1CBH)
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.