Creative Biolabs provides professional bispecific antibody (BsAb) design and construction services for diverse studies about therapeutic antibody engineering. IgG-v BsAb are a class of appended IgGs which include four different formats: IgG(H)-V, V(H)-IgG, IgG(L)-V, and V(L)-IgG to meet different customer’s special requirements.
In the past decades, various multi-specific and multi-valent biomolecules have been developed from immunoglobulin (Ig) or non-Ig sub-domains, heterodimeric mAbs or modified full-length antibodies. Sub-domain strategies are inherently mono-specific and mono-valent, but enable to be concatenated to produce low-order, multispecific and multi-valent biomolecules. These formats exhibit in vivo half-lives which are substantially less than those of typical mAbs, and need further engineering, for example, pegylation or fusion to an Fc domain, to extend half-life. Modified antibody formats are based on a full-length antibody scaffold, thus maintain many of the structural and functional features of typical mAbs. Nevertheless, as a consequence of their design, this group keeps largely bi-valent and bispecific. Heterodimeric mAbs usually produce thermodynamically stable proteins with inherent Fc-mediated effector function, but are also limited to bispecificity and mono-valent interactions with their targets.
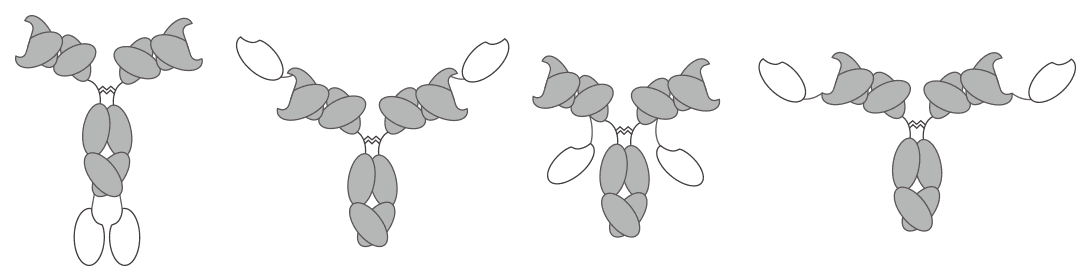
Fig. 1 Alternative formats of IgG-v BsAbs.
IgG-v BsAbs
IgG-v BsAbs are engineered by appending paired antibody variable domains Fv to the N or C terminus of the heavy or the light chain of an antibody, resulting in multi specific molecules with two binding sites for each antigen. They are the major class of appending IgG-like molecules which provide great starting points to design the most appropriate therapeutic antibodies so as to achieve the expected mechanisms and application potential. Depending on the strategy used for appending Fv to the whole IgG, there are four different formats of IgG-v BsAbs:
-
IgG(H)-V: a Fv domain linked to each of the C terminus of IgG HC;
-
V(H)-IgG: a Fv domain linked to each of the N terminus of the IgG HC;
-
IgG(L)-V: a Fv domain linked to each of the C terminus of the IgG LC;
-
V(L)-IgG: a Fv domain linked to each of the N terminus of the IgG LC.
Advantages of IgG-v BsAb
One outstanding advantage of IgG-v BsAb is that they are able to simultaneously bind to antigen to all variable domains and therefore give a higher specific binding capacity. This may be helpful in targeting low abundance proteins (e.g., Cytokines) and it may allow longer dosing intervals. Besides, the simultaneous binding of antigen holds the natural antibody avidity to cell surface receptors or dimeric antigens, and it offers an extended distance between the bound epitopes.
With the well-established IgG-v BsAb platform, the experienced scientists here at Creative Biolabs are dedicated to helping you develop therapeutic BsAbs. We also provide other various services regarding BsAbs development. Please feel free to contact us for more information and a detailed quote.
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY





