Creative Biolabs has successfully developed several different forms of bispecific antibodies (BsAbs). With experienced scientists hammering at Creative Biolabs, we are confident in providing best in class Fv (H,L)-IgG development services to contribute greatly to the success of your projects.
The molecular format of antibody-related biologics becomes more diverse to include different IgG subclasses and IgG with engineered Fc regions. Bi- and multispecific antibody formats, which are designed to exhibit improved clinical efficacy and unique functionality over common monoclonal antibodies, have exhibited great therapeutic potential. BsAb fragments may possess certain advantages over the full-length IgG-like molecules for tumor radioimaging and targeting, according to their better tissue penetration and faster clearance from the circulation. Nevertheless, full-length IgG-like bi- or multispecific molecules may be the superior choice for certain clinical applications, such as prolonged treatment for chronic immunologic indications and cancer. Comprising the Fc domain not only enhances long serum half-life but also supports secondary immune functions, including antibody-dependent cellular cytotoxicity (ADCC) and complement-mediated cytotoxicity (CDC).
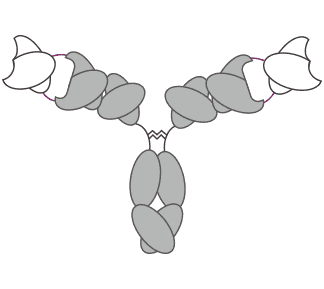
Figure 1. schematic diagram of the Fv (H,L)-IgG.
Fv (H,L)-IgG
Fv (H,L)-IgG is a format of appended IgG, which is IgG-like except that each light chain and heavy chain comprises two variable domains in tandem via a short peptide linkage, instead of one variable domain. It has been proven that the structural flexibility of IgG has functional significance for antigen binding. Therefore, based on the suitable peptide linkages between the two variable domains in both the heavy and light chains, the various motions within the Fab region enable to be maintained to offer sufficient structural freedom in the Fv (H,L)-IgG to allow for dual binding. There are several approaches for Fv (H,L)-IgG molecule optimization, such as the placement of the two variable domains in the correct orientation, as well as the adjustment and incorporation of peptide linkers connecting the two variable domains in the heavy and light chains.
Creative Biolabs adapts different linker to the different constructs of Fv (H,L)-IgG:
-
The light chain is Vλ-linker-Vκ-Cκ or Vλ-linker-Vλ-Cλ
-
The light chain is Vκ-linker-Vκ-Cκ or Vκ-linker-Vλ-Cλ
-
The heavy chain (g1) linkers
With the well-established Fv (H,L)-IgG technology platform, the experienced scientists here at Creative Biolabs are dedicated to helping you develop BsAbs. We also provide other various services regarding BsAbs development. Please feel free to contact us for more information and a detailed quote.
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY





