Creative Biolabs offers clients design and construction services of a novel form bispecific antibodies (BsAbs), the KIH IgG-scFab, for many different studies about antibody engineering. We are confident to deliver you a BsAb that can meet all of your required specifications.
Generally, BsAb are divided into five distinct structural groups: bispecific IgG (BsIgG), IgG appended with an additional antigen-binding moiety, bispecific fusion proteins, BsAb fragments, and BsAb conjugates. Each of these different BsAb formats processes different properties in binding valency for each antigen, pharmacokinetic half-life, and geometry of antigen-binding sites. Monospecific IgG is able to be designed for bispecificity via appending either the amino or carboxy termini of either light or heavy chains with additional antigen-binding units. Several additional antigen-binding units are available, for example, single domain antibodies (unpaired VL or VH), engineered protein scaffolds, as well as paired antibody variable domains (e.g., Fv or scFv). Creative Biolabs offers one of the appending IgG, KIH IgG-scFab, for custom.
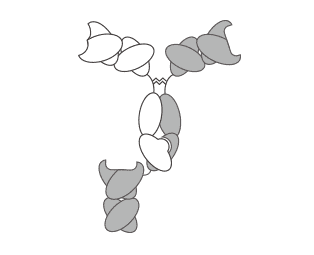
Fig. 1 Schematic of the KIH IgG-scFab antibody
KIH IgG-scFab
KIH IgG-scFab, a new form of appended IgG-like BsAbs, is engineered by fusing a scFab domain to the C-termini of IgG heavy chain modified by knobs-into-holes (KIH) technique. Hence, when co-expressed in one cell, Knob-modified Fv tend to pair with Hole-modified Fv, which effectively results in the production of asymmetric antibodies with dual specificities. Up to now, variant approaches have been established based on KIH method, such as using alternative mutations, electrostatic steering effects, or hybrid CH3 domains derived from IgG and IgA. KIH IgG-scFab can effectively prevent the formation of homodimer which inevitably happens during conventional bispecific IgG synthesis. Furthermore, this strategy makes it feasible to produce BsAb in industrial scale.
Advantages of Appended BsIgGs
A distinct advantage of appended BsIgG is that they are able to simultaneously bind antigen to all variable domains, therefore, provide a higher specific binding capacity. This is very useful in targeting low abundance proteins (e.g. cytokines), and it allows longer dosing intervals. Besides, the simultaneous binding of antigen protects the natural antibody avidity to cell surface receptors or dimeric antigens, and it offers an extended distance between the bound epitopes by placing the additional binding domains at the distal end of the heavy chain. What’s more, the flexibility to choose between the 2 + 2 and 2 + 1 antigen-binding valencies offers the opportunity to eliminate avidity through placing a single binding unit at the carboxy-terminus to generate monovalent binding while conserving the binding capacity to the second antigen.
With our well-established BsAb develop platform, the experienced scientists here at Creative Biolabs are dedicated to helping you develop unique BsAbs. We also provide other various services regarding BsAbs development. Please feel free to contact us for more information and a detailed quote.
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY





