Circulating tumor cell (CTC) is an important biomarker for early diagnosis of cancer. Among a number of greatly advanced strategies, electrochemical CTC sensors show absolutely advantages such as fast response, simplicity, low cost and the capability for miniaturization. Electrochemical sensors can provide quantitative or semi-quantitative analytical information by converting the interaction between CTCs and biorecognition probes into electrical readouts in the form of the changes in the current, the potential, or the impedance.
Common Detection Principles
- Electrochemical impedance spectroscopy (EIS) / transport blocking
- Catalytic amplification
Since the electron tunneling takes place within a distance less than 20 Angstroms, the presence of the electrochemically inert CTCs on the electrode surface would largely hinder the electron transfer between the electrode and the electroactive reporter molecules in solution. As a result, an increase in the impedance of the electrode as well as a decrease in the current output of the electroactive reporter molecules are expected. Detection based on this principle is able to quantify CTCs in a label-free manner. The label-free feature of EIS/diffusion blocking sensors can greatly simplify the operation procedures and save the analysis time. However, the downside of such sensors lies in the low sensitivity due to the high original background current.
When labeling CTCs using materials with catalytic activity towards specific substances in the electrolyte, the electrochemical signals can be amplified. Functional nanomaterials are commonly used labels which show great potential of improving the performance of the sensors due to their biocompatibility, highly catalytic performance, and stability.
Detection Method
The electrochemical CTC detection methods can be roughly divided into direct assays, sandwich assays and other assays. The direct assays use capture probe to (antibody or aptamer) bind and detect CTC. The sandwich assays add a signal probe on the basis of direct assays. After CTC is bound to the capture probe on the electrode, a signal probe is added. After the signal probe is combined with CTC, it can significantly amplify the signal, thereby increasing the sensitivity and reducing the detection limit.
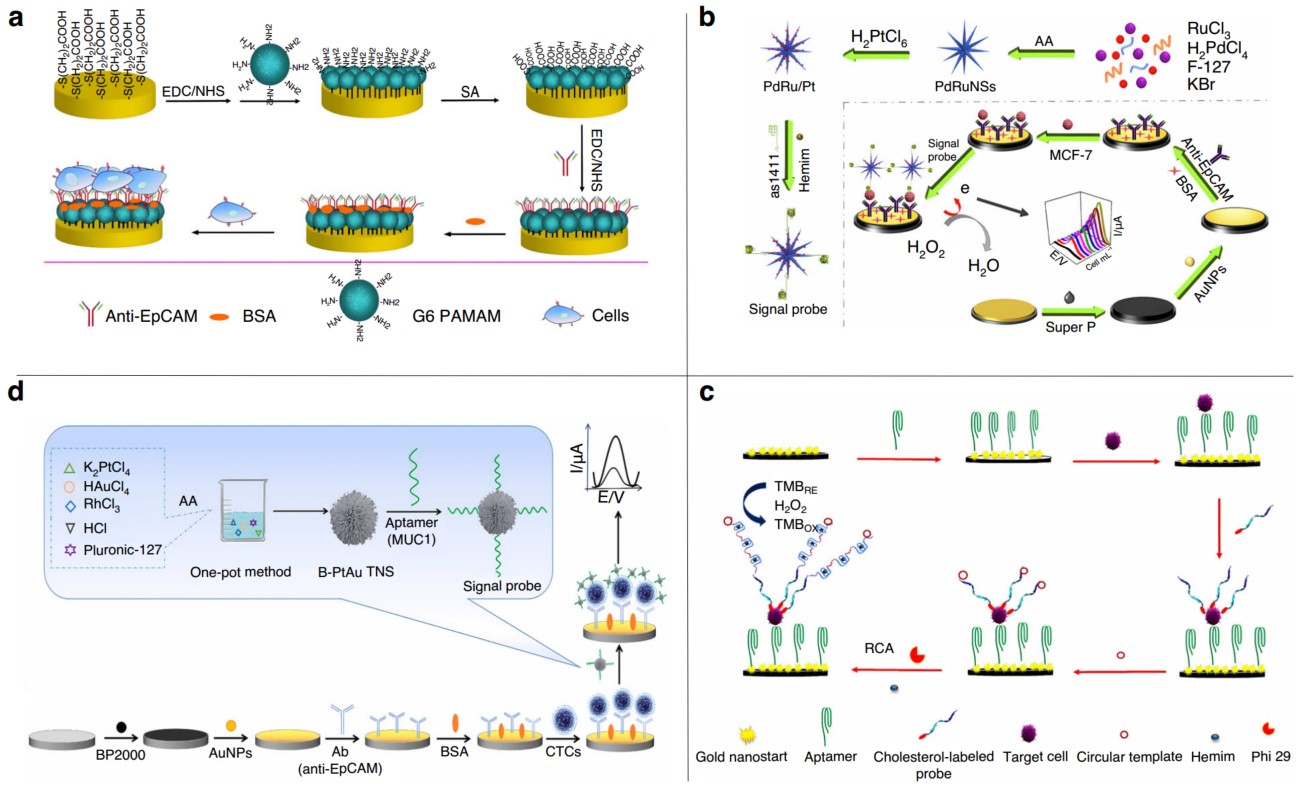 Fig.1 Electrochemical sensor for detecting CTCs.1
Fig.1 Electrochemical sensor for detecting CTCs.1
Case
Due to the extremely rare counts of CTCs in complex blood sample, efficient enrichment of CTCs become the first step for the precise detection of CTCs. Thus, functionalized magnetic nanospheres (MNs) were utilized to bind with tumor specific antigens associated with CTCs, in most cases is the epithelial cell adhesion molecule, EpCAM.
After enrichment of CTCs from blood, signal amplification is another important step for the sensitive detection of CTCs. Combing the MNs for enrichment of CTCs with DNA generated electrochemical current as well as rolling circle amplification (RCA) for signal amplification, scientist developed a sensitive electrochemical sensor for detection of CTCs in peripheral blood.
Reference
- Jiang, Liying, et al. "Current research status of tumor cell biomarker detection." Microsystems & Nanoengineering 9.1 (2023): 123. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.