Brain Organoids Introduction
Brain organoids are 3D self-aggregating structures derived from human embryonic or induced pluripotent stem cells. These organoids closely mimic the cellular composition and structural organization of the developing human brain, making them valuable in vitro models for investigating neurogenetic diseases. Their utility has led to widespread adoption in neurogenetic research.
What Are Brain Organoids?
Brain organoids are at the forefront of the organoid revolution. They are tiny, self-assembled 3D cellular clusters that resemble aspects of the developing human brain. They contain various neural cell types, including neurons, astrocytes, and oligodendrocytes, and can exhibit rudimentary neuronal activity, forming synaptic connections and even demonstrating patterns reminiscent of brain waves. While they are far from being "mini-brains" in the sense of possessing consciousness or complex cognitive functions, their remarkable structural and functional similarities to early human brain development make them invaluable tools for research.
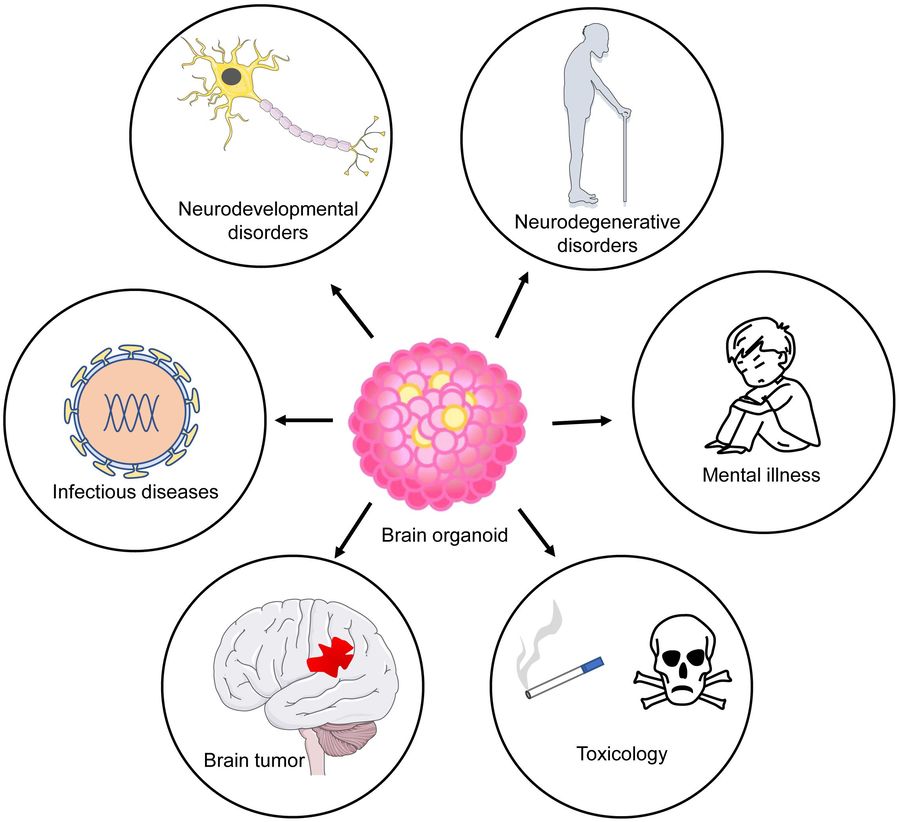 Figure 1 Application of brain organoids as disease models. Brain organoids have been used to model neurodevelopmental and degenerative diseases1,3.
Figure 1 Application of brain organoids as disease models. Brain organoids have been used to model neurodevelopmental and degenerative diseases1,3.
Characteristics of Brain Organoids

-
Advanced Experimental Model: Brain organoids stand out as an advanced experimental model due to their self-renewal, self-organization, functionality, and multicellularity, offering unparalleled insights into human brain development and function.
-
3D Clusters: Brain organoids are 3D clusters formed from human embryonic stem cells (hESCs) or induced pluripotent stem cells (hiPSCs), exhibiting remarkable structural and cellular fidelity to the human brain.
-
Patient-Specific Genetic Information: A significant advantage is their ability to carry patient-specific genetic information, allowing for more precise and clinically relevant research.
-
Diversity and Flexibility: The versatility of organoid technology allows for the creation of both global brain models and region-specific constructs (e.g., cerebral cortex, midbrain, striatum).
-
Self-Organization Capability: Ultimately, brain organoids' superior self-organizing capabilities and ability to accurately recapitulate human brain complexity make them an indispensable model for understanding neurological disease pathogenesis.
How to Culture Brain Organoid
Current methods for generating brain organoids mainly use hESCs or hiPSCs as starting cells. hESCs or hiPSCs are guided to form embryoid bodies and then differentiate into neuroepithelial tissues in an inductive culture environment. To enhance their development, these tissues are then transferred to Matrigel droplets and maintained in a rotating bioreactor for 1-2 months. This innovative culture system improves nutrient delivery and provides essential spatial support, enabling the formation of brain organoids with integrated neuroepithelial tissue. These comprehensive 'whole brain organoids' contain a rich array of cell lineages, such as the ventral forebrain, midbrain, choroid plexus, dorsal cortex, retina, and hippocampus. This remarkable cellular complexity allows these organoids to robustly simulate the intricate interconnections and dynamic interactions characteristic of the human brain.
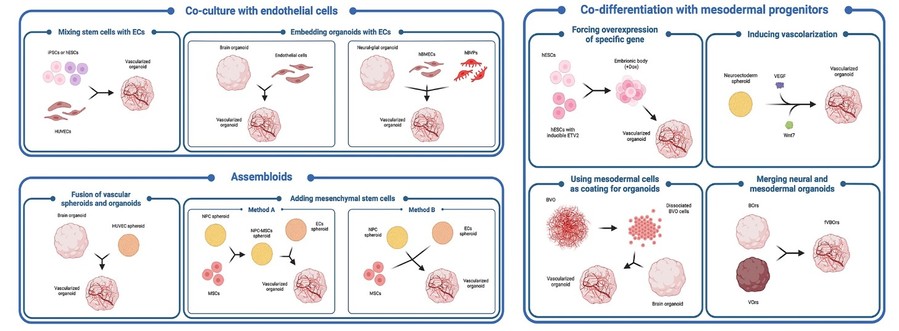 Figure 2 Schematic pictorial of different in vitro strategies for the generation of vascularized brain organoids.2,3.
Figure 2 Schematic pictorial of different in vitro strategies for the generation of vascularized brain organoids.2,3.
Applications of Brain Organoids in Genetic Diseases of Nervous System
|
Disease
|
Cell
|
Dedifferentiation/gene editing/disease induction
|
Organoid type
|
Phenotype
|
|
Microcephaly (MCPH)
|
Patient-derived fibroblasts
|
Dedifferentiation using lentiviral OSKM
|
Whole-brain organoid
|
Small organoid size
Immature neural differentiation
|
|
Autism spectrum disorder (ASD)
|
Patient-derived fibroblasts
|
Dedifferentiation using retroviral OSKM
|
Forebrain organoid
|
Increased FOXG1 expression
Increased GABAergic inhibitory neurons
|
|
Rett's syndrome (RTT)
|
Patient-derived fibroblasts
|
Dedifferentiation using retroviral OSKM
|
Whole-brain organoid
|
Abnormal miR-199 and miR0214 expression
Abnormal migration of neural progenitor cells
|
|
Alzheimer's disease (AD)
|
Pluripotent stem cells
|
PSEN2 mutation correction through gene editing
|
Whole-brain organoid
|
Small organoid size
High Aβ42/Aβ40 ratio
Abnormal Ca2+ homeostasis
Excessive neuron activation
|
|
Parkinson's disease (PD)
|
Patient-derived fibroblasts
|
Dedifferentiation using a lentiviral OSKM
|
Midbrain organoid
|
Increased sensitivity to toxicity
Increased alpha synuclein
Increased thioflavin T-positive cells
|
Challenges of Brain Organoids in Genetic Diseases of Nervous System Models
-
Heterogeneity and Reproducibility Challenges
Brain organoid research faces significant hurdles due to issues of heterogeneity and reproducibility. Diverse organoid morphology and structure arise from variations in source cells, experimental protocols, and inter-laboratory differences. This complexity is compounded by the absence of exogenous induction factors and reliance on stem cell self-organization, complicating batch-to-batch comparisons. Furthermore, subtle differences in personnel, operational details, and environmental conditions contribute to low reproducibility.
-
Application Limitation
A key challenge is the absence of a vascular system, which hinders oxygen and nutrient supply, potentially leading to tissue necrosis. Their developmental timeline also diverges from in vivo brain development, with current mature organoids primarily modeling embryonic or fetal stages. This restricts their ability to fully replicate the intricate physiological and pathological processes seen in the adult brain. Furthermore, conventional brain organoids lack immune cells and struggle to recapitulate complex neural circuits, thereby limiting their utility in neurological disease research.
Optimization Strategies for Brain Organoids
01 Standardized Experimental Procedures and Consistency Control
Establish standard operating procedures in the laboratory to ensure the consistency of operations for each construction of brain organoid models. All operators should be trained and follow standard procedures, and the experimental materials and environment should be kept consistent as much as possible to reduce differences caused by heterogeneity.
02 Cell Selection and Sterile Operation
Select hESCs or hiPSCs with good growth status and in the logarithmic growth phase as raw materials for constructing brain organoids, and expand the initial number of cells to increase the volume of embryoid bodies. At the same time, pay attention to sterile operation throughout the process to avoid bacterial contamination and ensure the success of the experiment.
03 Culture Technology and Microwell Array Application
Prepare fresh culture medium to maintain its molecular activity, and maintain gentle force during operation to avoid damaging brain organoids. At the same time, a microwell culture platform based on 3D printing can be used to improve the homogeneity and repeatability of cell aggregates.
Advantages of Brain Organoids
-
Higher Clinical Relevance
-
Self-Renewal and Organization
-
Convenience of Genetic Manipulation
-
Robustness and Speed
Frequently Asked Questions (FAQs)
Q: What is the primary advantage of using brain organoids over animal models for neurological research?
A: The primary and most compelling advantage of using brain organoids over traditional animal models (such as mice or rats) for neurological research lies in their human specificity and relevance.
Q: Can brain organoids be used to study psychiatric disorders like schizophrenia or autism?
A: Absolutely! Brain organoids represent a truly transformative tool for studying complex neuropsychiatric disorders like schizophrenia, autism spectrum disorders (ASD), bipolar disorder, and major depressive disorder. Understanding the biological basis of these conditions has historically been incredibly challenging due to their complex genetic and environmental influences, and the inherent inaccessibility of living human brain tissue.
Q: How long can brain organoids survive and function in the lab?
A: The lifespan and functional maturity of brain organoids in the laboratory can vary significantly, ranging from weeks to many months, and in some advanced cases, even over a year. The duration depends on several factors, including the specific protocol used, the type of organoid (e.g., forebrain vs. midbrain), the culture conditions, and the research goals.
Q: What does the future hold for brain organoid research?
A: The field of brain organoid research is rapidly advancing. Future developments aim to overcome current limitations by incorporating vascularization, immune cells, and more mature brain structures to create even more physiologically relevant models. Researchers are also working on developing higher-throughput screening platforms and integrating organoids with advanced bioengineering techniques to further enhance their utility for drug discovery, personalized medicine, and a deeper understanding of brain function and dysfunction.
Conclusion
Brain organoids represent a promising new model for neurological diseases. They offer significant advantages, including high cellular heterogeneity, self-renewal capacity, and ease of genetic manipulation. This makes them a powerful tool for investigating the pathogenesis of neurological genetic diseases and developing novel therapies. However, practical application of brain organoids faces technical, biological heterogeneity, and ethical challenges. Collaborative efforts are essential to continuously refine and improve this technology, enabling its full potential in understanding and treating neurological genetic diseases.
Elevate Your Research with Creative Biolabs' Advanced Brain Organoids Model
Creative Biolabs is a professional biotechnology company and has made continuous efforts into studying brain organoid models. Contact us today to learn more!
References
-
Shou, Y.; et al. The application of brain organoids: from neuronal development to neurological diseases. Front Cell Dev Biol. 2020, 8: 579659. https://doi.org/10.3389/fcell.2020.579659
-
Rizzuti M, Melzi V, Brambilla L, et al. Shaping the neurovascular unit exploiting human brain organoids. Molecular Neurobiology, 2024, 61(9): 6642-6657. https://doi.org/10.1007/s12035-024-03998-9
-
Distributed under Open Access license CC BY 4.0, without modification.
Research Model
Related Sections:

 Figure 1 Application of brain organoids as disease models. Brain organoids have been used to model neurodevelopmental and degenerative diseases1,3.
Figure 1 Application of brain organoids as disease models. Brain organoids have been used to model neurodevelopmental and degenerative diseases1,3.

 Figure 2 Schematic pictorial of different in vitro strategies for the generation of vascularized brain organoids.2,3.
Figure 2 Schematic pictorial of different in vitro strategies for the generation of vascularized brain organoids.2,3.