Organ-on-a-Chip
Basic Definition
An organ-on-a-chip (OOC) is a microfluidic cell culture device that mimics the physiological function, mechanical properties and biochemical responses of organs or organ systems. Such devices, usually made using soft lithography techniques with polydimethylsiloxane (PDMS), seek to replicate the dynamic cellular microenvironment in vivo and to be a more predictive and human-relevant system compared to static 2D cultures.
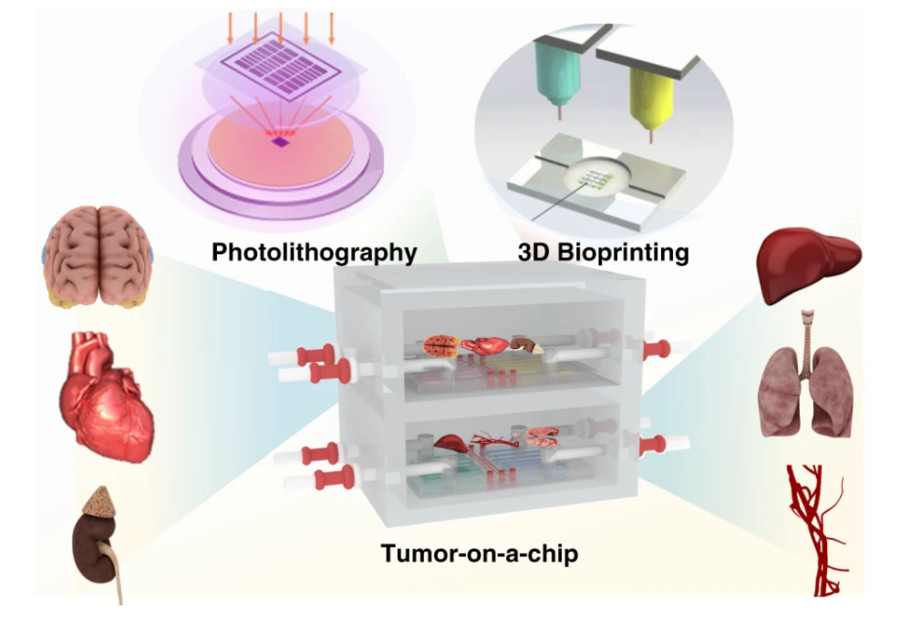 Figure 1 Schematic of the tumor-on-a-chip platform for modeling tumor cell tissue structure and functional units in vitro.1,3
Figure 1 Schematic of the tumor-on-a-chip platform for modeling tumor cell tissue structure and functional units in vitro.1,3
Composition Structure of Organ-on-a-Chip
An Organ-on-a-Chip (OOC) is a device that replicates the morphology, functions, and microenvironment of a living organ. Its components have been engineered to fulfill these functions:
-
Microfluidic Channels: These simulate blood vessels and control the continuous flow of media, nutrients, and cells, while modulating shear stress.
-
Cell Culture Chambers: Organ-specific and immune cells are seeded into these compartments to create tissue-like structures and co-cultures.
-
Porous Membranes: Permeable barriers that separate cell compartments for the modeling of barrier tissues and immune cell transmigration.
-
Biomaterials and Extracellular Matrix (ECM): Provide a 3D scaffold that simulates native tissue morphology and influences cell shape and function.
-
Sensors and Actuators: These integrated components allow for real-time monitoring of physiologic parameters (e.g., oxygen, pH) and dynamic mechanical stimulation (e.g., stretch).
Immunology Research Review
Immunology is a scientific field that involves the highly organized biological defense system of the body. Its function is to prevent foreign pathogens, non-self-cells, and environmental toxins, and to distinguish between self and non self. It can be divided into two categories: innate immunity and adaptive immunity. Innate immunity is a rapid, non-specific immunity that immediately responds to pathogens, while adaptive immunity provides long-term specific immunity against antigens. The role of cell populations (lymphocytes, phagocytes, and antigen-presenting cells) in maintaining homeostasis and generating immune responses to pathogens or toxins is a research focus in this field. The study of immunology traditionally has the following disadvantages:
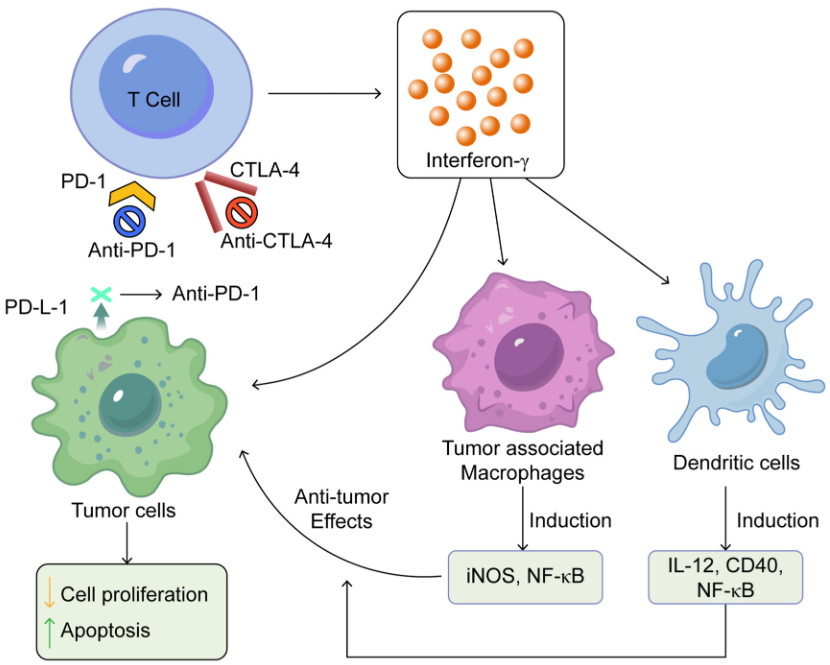 Figure 2 Summarises the mechanisms of action of immune checkpoint inhibitors.2,3
Figure 2 Summarises the mechanisms of action of immune checkpoint inhibitors.2,3
-
In vitro models (traditional 2D culture): These models lack physiological stimuli such as 3D environment, shear stress, and cell-cell interactions, and cannot accurately replicate in vivo immune responses; Therefore, they perform poorly in predicting in vivo immune responses.
-
Animal models: They provide an understanding of the system, but due to species barriers, high costs, and variability, they lack direct applicability to humans. This translation gap highlights the urgent need for more physiologically relevant humanoid platforms.
Cell Sources of Organ-on-a-Chip for Immunology Research
|
Cell Source Category
|
Examples
|
Characteristics & Advantages
|
Disadvantages
|
|
Primary Cells
|
PBMCs, T cells, B cells, macrophages, endothelial cells
|
- Physiological Relevance: Most accurately reflect in vivo behavior, morphology, and function.
- Direct from Human Tissue: Ideal for patient-specific studies.
|
- Limited Availability: Scarce, requiring fresh samples.
- Donor Variability: Significant batch-to-batch variation.
- Limited Proliferation: Finite lifespan.
- High Cost & Technical Demand: Expensive and requires specialized expertise.
|
|
Immortalized Cell Lines
|
Jurkat, THP-1, HUVEC, Caco-2
|
- Infinite Proliferation: Easily scalable for high-throughput screening.
- Reduced Variability: Homogeneous populations ensure high reproducibility.
- Cost-Effective: Less expensive to acquire and maintain.
|
- Reduced Physiological Relevance: Often lose key in vivo characteristics.
- Chromosomal Abnormalities: Can have altered gene expression.
- Species Origin: Many are non-human.
|
|
Induced Pluripotent Stem Cell (iPSC)-Derived Cells
|
iPSC-derived immune cells (e.g., macrophages, T cells), organ-specific cells
|
- Patient-Specific Models: Can generate disease-relevant or patient-specific cell types.
- Unlimited Supply: Overcomes limited availability of primary cells.
- Ethical Advantages: Avoids concerns associated with embryonic stem cells.
- Disease Modeling: Excellent for genetic predispositions.
|
- Differentiation Challenges: Complex and time-consuming protocols.
- Maturation Status: May not fully mature to adult primary cells.
- Cost & Expertise: High cost and specialized expertise required.
|
Organ-on-a-Chip for Immunology Research
Organ-on-a-chip is a game changer in understanding immune phenomena that cannot be studied in 2D or in vivo environments. OOC can reproduce the complexity of real life by simulating various conditions such as gradients, perfusion, and interactions between cells, and can be used in many fields of immunological research.
Immunology Modeling
One of the most promising applications of organic technology on chips in immunology is the immunotoxicity screening of drugs and environmental compounds.
-
Inflammatory Diseases: Use the lung, gut, and joint chips to model chronic inflammation (e.g., IBD) and analyze inflammatory pathways and anti-inflammatory therapies.
-
Host-Pathogen Interactions: Study pathogen infection in the lung chip (e.g., SARS-CoV-2, influenza virus) with analysis of viral replication, barrier breach, immune cell infiltration, and antiviral therapy efficacy.
At present, the process of testing vaccine immunogenicity and efficacy largely relies on animal models. Dongfang Overseas has provided an exciting new avenue for preliminary screening. We are developing chip-based lymph nodes or chip-based spleen chips that simulate the initiation of immune responses in secondary lymphoid organs. They exposed APCs to vaccine components and added T and B cells to test antigen presentation, T cell activation, clone amplification, and antibody production. This can accelerate the selection and mechanism research of early vaccines.
The OOC platform has also been applied to the modeling of specific autoimmune diseases to understand disease mechanisms and develop targeted therapies. For example, on-chip joints containing synovial cells, chondrocytes, and immune cells isolated from rheumatoid arthritis (RA) patients can be used to study inflammatory mediators, cartilage destruction, and anti-rheumatic drug activity. Similarly, a pancreatic chip containing islets and immune cells can be used to simulate autoimmune insulitis in type 1 diabetes.
Limitations of Organ-on-a-Chip in Immunology Research
Complexity vs. Whole Organism: While OOCs simplify the full complexity of the human immune system (without mimicking systemic circulation, hormonal regulation, neural innervation, and interactions between different organs), they lack an effective scale up strategy.
-
Because most of the OOCs in development are designed for use at the laboratory scale, their use for high throughput screening for new therapeutics is impeded by the lack of automated systems and need for highly skilled labor.
-
Due to a lack of standard protocols and validated platforms, different research groups have difficulty reproducing the OOC experiments, hindering adoption in general and regulatory acceptance.
-
Immune cell culture and long-term maintenance inside an OOC device is still an open challenge, and viability and functionality of primary human immune cells in the OOC environment remains a significant challenge.
Conclusions
OOCs are a promising new technology aimed at bridging the gap between low complexity in vitro systems and high complexity in vivo studies. By closely replicating the physiological microenvironment, mechanical forces, and cell-cell interactions found in human organs, OOCs allow for the study of high-fidelity immune responses. They can be used to evaluate drug immunotoxicity, simulate inflammatory diseases such as IBD and pulmonary inflammation, analyze host pathogen interactions, and accelerate the development of cancer immunotherapy and vaccines. In addition, by using patient derived cells, OOCs have the potential to be used for personalized medicine and personalized drug responses. There are challenges in scalability, standardization, and reproducibility of systemic effects, however, OOC technology is changing the landscape of immunology, enabling more accurate preclinical data and accelerated clinical translation.
Frequently Asked Questions
Q: What are the main benefits of Organ-on-a-Chip (OOC) compared to in vitro and animal models in immunology research?
A: One of the main benefits of OOCs is the ability to create a physiologically relevant microenvironment. This is achieved by providing 3D tissue structure, mechanical forces (such as shear stress, tension), and dynamic fluid flow, which would otherwise not be possible in 2D culture. Compared to animal models, they are targeted towards humans, minimizing the challenges of species translation, reducing the cost of high-throughput screening, and minimizing ethical considerations. More precise control of the experimental environment and real-time monitoring of cellular responses can be achieved.
Q: How accurate are Organ-on-a-Chip models in mimicking in vivo immune responses?
A: The OOC model significantly improves in vitro immune response simulation by combining various elements of the in vivo microenvironment, such as tissue barriers, mechanical stimulation, and multicellular interactions. They have limitations in simulating the systemic complexity of the entire organism, such as the central nervous system and endocrine system, but can be very accurate in simulating immune function at specific organ levels, host pathogen interactions, and drug-induced immune toxicity.
Q: What types of immune responses can be specifically studied on Organ-on-a-Chip platforms?
A: OOC platforms are versatile for studying a wide range of immune responses, including:
-
Immune cell activation, proliferation, and differentiation.
-
Cytokine and chemokine production.
-
Immune cell extravasation and transmigration across endothelial or epithelial barriers.
-
Phagocytosis and antigen presentation.
-
Host-pathogen interactions (bacterial, viral, fungal infections).
-
Drug-induced immune activation or suppression.
-
Tumor-immune cell interactions and immunotherapy efficacy.
-
Inflammatory processes in specific organs (e.g., gut inflammation, lung inflammation).
Overview of What Creative Biolabs Can Provide
As one of the most advanced biotechnology companies in the world, Creative Biolabs aims to provide cutting-edge solutions for biomedical research through Organ-on-a-Chip. Our technology will support you in the research of your immunology study and immunotherapy development, including designing, producing, as well as analyzing all the results for better quality, physiology-relevant, and reproducible data.
Creative Biolabs provides comprehensive end-to-end support in OOC-based immunology studies. From customized OOC design and fabrication to pre-validated immune-component-integrated OOC models, expert cell sourcing (human primary cells and iPSC derivatives), comprehensive immunoassay development tailored for OOC, to consultation and R&D support. Contact us today to learn more!
References
-
Liu X, Fang J, Huang S, et al. Tumor-on-a-chip: from bioinspired design to biomedical application. Microsystems & Nanoengineering, 2021, 7(1): 50. https://doi.org/10.1038/s41378-021-00277-8
-
Justiz-Vaillant A, Pandit B R, Unakal C, et al. A Comprehensive Review About the Use of Monoclonal Antibodies in Cancer Therapy[J]. Antibodies, 2025, 14(2): 35. https://doi.org/10.3390/antib14020035
-
Distributed under Open Access license CC BY 4.0, without modification.
Research Model
Related Sections:

 Figure 1 Schematic of the tumor-on-a-chip platform for modeling tumor cell tissue structure and functional units in vitro.1,3
Figure 1 Schematic of the tumor-on-a-chip platform for modeling tumor cell tissue structure and functional units in vitro.1,3
 Figure 2 Summarises the mechanisms of action of immune checkpoint inhibitors.2,3
Figure 2 Summarises the mechanisms of action of immune checkpoint inhibitors.2,3