Introduction of ANO4
ANO4 is encoded by the ANO4 gene and is also known as transmembrane protein 16D (TMEM16D). It belongs to the transmembrane protein 16 (TMEM16) family which is a large family of membrane proteins and is highly conserved in different eukaryotic organisms from fungi to invertebrate to mammals. TMEM16 family is also known as lipid scramblases because it moves lipids between the two layers of the membrane. Meanwhile, it was concluded that ANO4 has 8 transmembrane helices according to the structure of the fungal homolog nhTMEM16.
| Basic Information of ANO4 | |
| Protein Name | Anoctamin-4 |
| Gene Name | ANO4 |
| Aliases | Transmembrane protein 16D, TMEM16D |
| Organism | Homo sapiens (Human) |
| UniProt ID | Q32M45 |
| Transmembrane Times | 8 |
| Length (aa) | 955 |
| Sequence | MEASSSGITNGKTKVFHPEGGVDLQGYQLDMQILPDGPKSDVDFSEILNAIQEMAKDVNILFDELEAVSSPCKDDDSLLHPGNLTSTSDDASRLEAGGETVPERNKSNGLYFRDGKCRIDYILVYRKSNPQTEKREVFERNIRAEGLQMEKESSLINSDIIFVKLHAPWEVLGRYAEQMNVRMPFRRKIYYLPRRYKFMSRIDKQISRFRRWLPKKPMRLDKETLPDLEENDCYTAPFSQQRIHHFIIHNKETFFNNATRSRIVHHILQRIKYEEGKNKIGLNRLLTNGSYEAAFPLHEGSYRSKNSIRTHGAENHRHLLYECWASWGVWYKYQPLDLVRRYFGEKIGLYFAWLGWYTGMLFPAAFIGLFVFLYGVTTLDHSQVSKEVCQATDIIMCPVCDKYCPFMRLSDSCVYAKVTHLFDNGATVFFAVFMAVWATVFLEFWKRRRAVIAYDWDLIDWEEEEEEIRPQFEAKYSKKERMNPISGKPEPYQAFTDKCSRLIVSASGIFFMICVVIAAVFGIVIYRVVTVSTFAAFKWALIRNNSQVATTGTAVCINFCIIMLLNVLYEKVALLLTNLEQPRTESEWENSFTLKMFLFQFVNLNSSTFYIAFFLGRFTGHPGAYLRLINRWRLEECHPSGCLIDLCMQMGIIMVLKQTWNNFMELGYPLIQNWWTRRKVRQEHGPERKISFPQWEKDYNLQPMNAYGLFDEYLEMILQFGFTTIFVAAFPLAPLLALLNNIIEIRLDAYKFVTQWRRPLASRAKDIGIWYGILEGIGILSVITNAFVIAITSDFIPRLVYAYKYGPCAGQGEAGQKCMVGYVNASLSVFRISDFENRSEPESDGSEFSGTPLKYCRYRDYRDPPHSLVPYGYTLQFWHVLAARLAFIIVFEHLVFCIKHLISYLIPDLPKDLRDRMRREKYLIQEMMYEAELERLQKERKERKKNGKAHHNEWP |
Function of ANO4 Membrane Protein
The activity of ANO4 is activated by sub-micromolar Ca2+ concentration, which is a shared characteristic of TMEM16 proteins. The underlying regulatory ligand binding site and some of the interacting residues of Ca2+ binding have not been identified so far. ANO4 is a member of the TMEM16 family, which is negatively charged chloride ion. The structure of ANO4 has not been reported so far. However, the biochemical analysis in the ANO4 expressing cells shows that the whole-cell currents of variable magnitudes are activated by ATP and the activation of ANO4 by ATP can induce the increase of the intracellular Ca2+. The increase of Ca2+ may not only activate the Cl− channel, but may also trigger the inhibition of anoctamins via CAMKI, which may actually counteract further activation. For all anoctamins except of ANO4, membrane voltages were depolarized through stimulation of purinergic receptors and increase of Ca2+, which is expected upon increase of a Cl− conductance along with cation influx through store-operated Ca2+ channels.
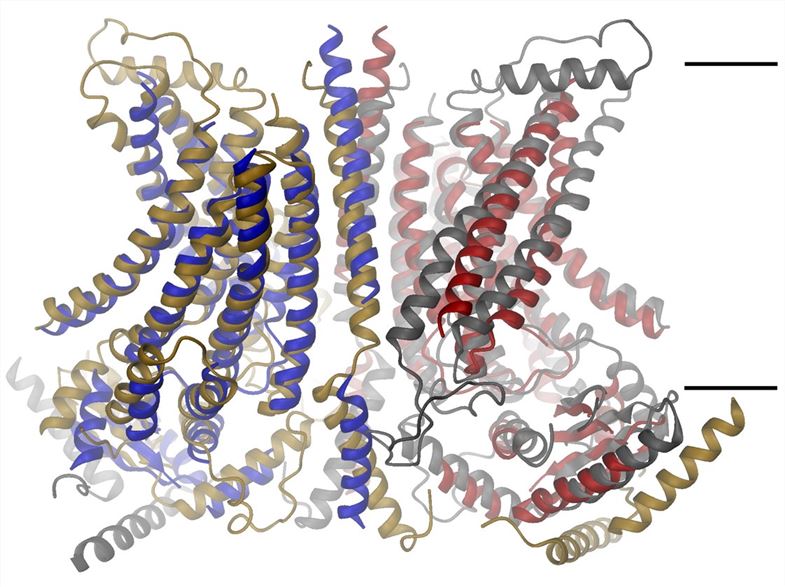 Fig.1 Proposed structure of ANO4 membrane protein (Paulino, 2017)
Fig.1 Proposed structure of ANO4 membrane protein (Paulino, 2017)
Application of ANO4 Membrane Protein in Literature
This article reports that in the ANO4 expressing cells, the whole-cell currents of variable magnitudes are activated by ATP and the activation of ANO4 by ATP can induce the increase of the intracellular Ca2+. The increase of Ca2+ may not only activate the Cl− channel, but may also trigger the inhibition of anoctamins via CAMKI, which may actually counteract further activation.
This article reveals that the anoctamin family exhibits characteristics most similar to those expected for the classical Ca2+-activated Cl− channels proteins, which perform many important functions in cell physiology processes.
Authors in this group apply cryo-electron microscopy and a complementary functional characterization to reveal the structural basis of anion conduction in a TMEM16 channel and it defines the foundation for the diverse functional behavior in the TMEM16 family.
The results of this article suggest that TMEM16 provides a cleft containing hydrophilic "stepping stones" for the outward translocation of phospholipids.
This article focuses on the X-ray structure of a fungal TMEM16 homologue, which can provide insight into the architecture of this protein family and into potential scrambling mechanisms.
ANO4 Preparation Options
To obtain the soluble and functional target protein, the versatile Magic™ membrane protein production platform in Creative Biolabs enables many flexible options, from which you can always find a better match for your particular project. Aided by our versatile Magic™ anti-membrane protein antibody discovery platform, we also provide customized anti-ANO4 antibody development services.
Creative Biolabs is a global research institute and a leading customer service provider in the field of membrane proteins, we have built impeccable reputations among our worldwide customers for successfully accomplishing numerous challenging projects including generation of many functional membrane proteins. Please feel free to contact us for more information.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.