With unchallenged experience in bispecific antibodies (BsAbs) synthesis, Creative Biolabs has long been well-known in the field of antibody production. We elaborately integrate our multiple platforms for providing customer a desired diabody with high affinity and low immunogenicity for both academic and clinical purposes.
Linking protein components to produce a predictable and rigid structure is a prior condition for producing complex protein assemblies in a predetermined fashion. A majority of the chemical cross linkers available process long and flexible spacers. According to this, the linked proteins have prominent structural flexibility, and the relative orientation and distance between their protein components is mostly unpredictable. Recombinant BsAbs are artificial proteins comprising the antigen- binding domains of two different antibodies. They are being widely researched on account of their “two-target” functionality can enhance the therapeutic value of the individual antibodies by inducing hetero-dimerization of antigens. Until now, more than 50 different recombinant formats are being created for producing BsAbs and a number of the latter are in various stages of clinical trial against cancers and inflammatory diseases.
Diabody
Diabodies are among the smallest of these BsAbs. They are formed by incorporating heavy-chain variable fragments (VH) and light-chain variable fragments (VL) with short “GGGGS” peptide linkers. However, the five-amino-acid linker is too short to span the distance of ~35 angstroms from the VH C terminus to the VL N terminus. Thus, the two VH-linker-VL chains associate to generate a dimer with two antigen binding sites, each contained the VH and VL fragments from the different chains.
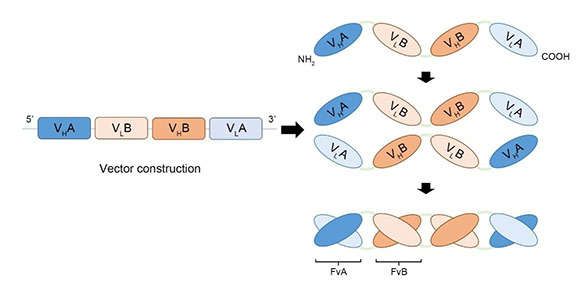
Fig. 1 Diagram of tandem diabody procedure.
When the two Fv domains in the diabody are identical, the structure is named as a monospecific diabody. Such diabodies lead to homo-dimerization of target proteins science they are able to simultaneously recognize two identical protein molecules. By the contrast, bispecific diabodies have Fv domains deriving from two different antibodies and enable to bridge different proteins resulting in their hetero-dimerization.
Diabody Engineering
Based on development of Diabody, some other engineered BsAbs have also been generated including scDiabody, Diabody-CH3, scDiabody-CH3, Diabody-Fc, scDiabody-Fc. In these derivate molecules, a disulfide bond can be introduced into the diabody to increase the stability, or a flexible linker is engineered to connect two antibody chains.
With the well-established diabody generation platform, the experienced scientists here at Creative Biolabs are dedicated to help you develop unique diabody. To meet the requirements of every client, we provide custom diabody and other diabody-based fragments, including Tandem diabody, single-chain diabody, diabody-Fc, etc. We also provide other various services regarding BsAbs development. Please feel free to contact us for more information and a detailed quote.
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY





