Rodent Fluid Percussion Injury (FPI) Model of Traumatic Brain Injury
Fluid percussion injury (FPI) is capable of producing diffuse (midline FPI) and mixed (focal and diffuse; lateral FPI) injury pathologies that are frequently seen following human traumatic brain injury (TBI). Creative Biolabs conducts contract studies in this FPI model of TBI to test the efficacy of novel therapeutics. The injuries produced with this model are scalable and we can produce mild, moderate, and severe injury severities according to your research requirements.
Introduction of FPI Model
FPI is a highly characterized and widely accepted model that has been successfully applied in several animal species including rabbit, cat, rat, mouse, and pig. FPI is inflicted as a metal pendulum is released and strikes a piston which drives injection of a small volume of sterile saline into the cranial cavity of the animal through the plastic luer-loc needle hub. The hammer (pendulum) is controlled electrically and the voltage correlates to the pressure delivered. The injury can be applied centrally (midline FPI), over the sagittal suture midway between bregma and lambda, or laterally (lateral FPI), over the parietal cortex.
The lateral FPI (at least 3.5 mm lateral to the sagittal suture) leads to contralateral cortical damage with impact-both focal (such as hemorrhage) and diffuse similar to most human TBI. It allows for a direct unilateral injury with very little to no subsequent contralateral rebound and/or brainstem injury. The midline FPI impacts both cortices and subsequently shifts the brainstem which results in increased mortality thus limiting the dose-response. Each injury model induces similar signs and symptoms as in humans which allows for the study of pathology, physiology, and pharmacology.
Features of FPI Model
- The FPI model manifests pathologic features of TBI such as axonal injury, neuroinflammation, neuronal loss and glial activation. This model also leads to impairments in spatial learning and memory, cognitive deficits, and deficits in motor learning.
- The severity of the injury can be controlled by altering the strength of the pressure pulse and also the site of the craniotomy.
- In rodent models, FPI has been shown to produce cognitive deficits that can last for weeks to months postinjury, providing avenues to evaluate therapeutic efficacy.
- The use of fluid is less mechanically controllable because of its diffusivity and the accumulation of gas bubbles.
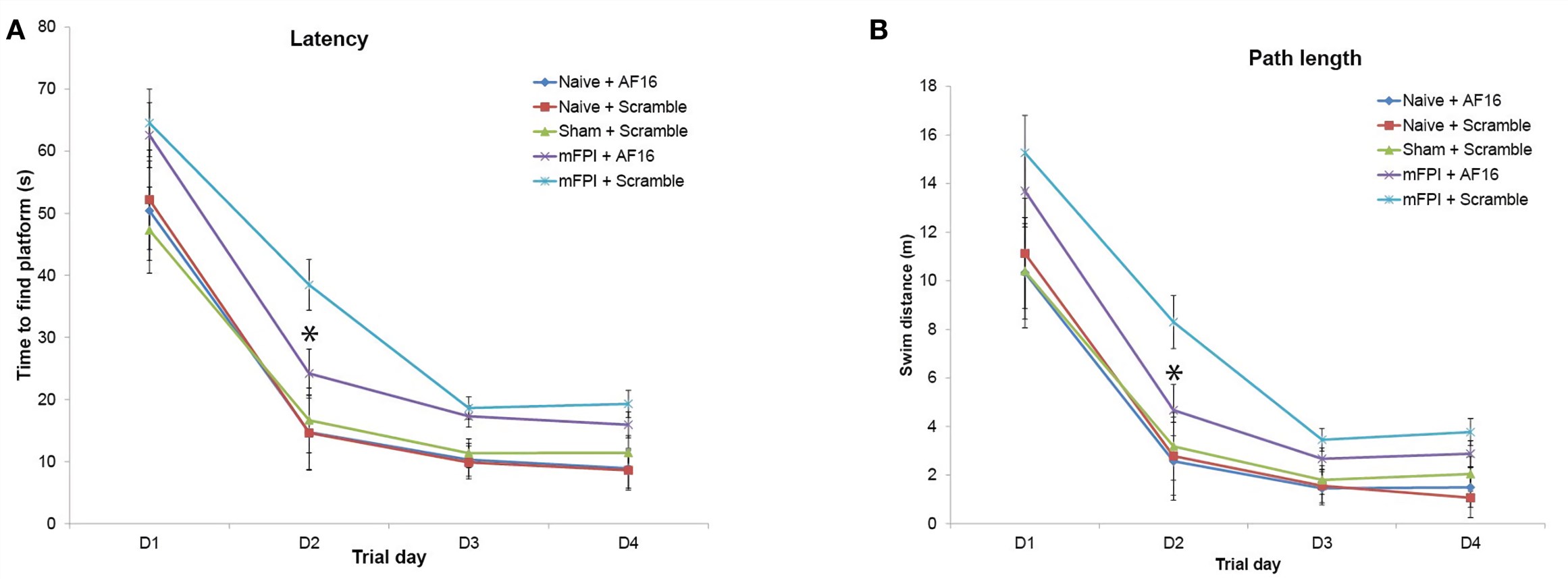 Fig. 1 Results from the Morris Water Maze learning trials for the 15-day survival study (means ± seM). Rats were subjected to sham- or midline fluid percussion injury (mFPI), and naïve controls were used. The animals were treated with either antisecretory factor (AF)-16 as the active, treatment compound or an inactive control compound (scramble). (Clausen et al. 2017)1, 2
Fig. 1 Results from the Morris Water Maze learning trials for the 15-day survival study (means ± seM). Rats were subjected to sham- or midline fluid percussion injury (mFPI), and naïve controls were used. The animals were treated with either antisecretory factor (AF)-16 as the active, treatment compound or an inactive control compound (scramble). (Clausen et al. 2017)1, 2
Assessments
To assess the injury severity level, there are multiple outcome measures that can be used (e.g., neurological, physiological, neurobehavioral, histological techniques). Neurological assessments mainly include the measurement of the duration of the reflex suppression (e.g., pinna, cornea, righting). For evaluating motor impairment, behavior tests such as the beam balance, beam walk, and Rotarod tests can be used. Moreover, the amount of weight loss is also recorded as it is correlated with the degree of injury severity. Briefly, Creative Biolabs provides assessments including, but not limited to:
- Body weight
- Acute neurological reflexes
- Behavioral tests (e.g., motor function, cognition, social behavior)
- Histology analysis
- Magnetic resonance imaging (MRI) and magnetic resonance spectroscopy (MRS)
Additionally, we provide other TBI rodent models that you may be interested in:
As a preclinical CRO focused on efficacy studies, Creative Biolabs has a strong expertise in Central Nervous System (CNS) preclinical efficacy data with a focus on different neurological disorders. The comprehensive list of rodent neurological disease models is placed below for you to review:
Choosing Creative Biolabs means having the opportunity of working closely with our highly-skilled, motivated and dedicated team members, fully adaptable to your processes. Contact us to discuss your requirements and enjoy the value of our expert services.
References
- Clausen, F.; et al. Intranasal administration of the antisecretory peptide AF-16 reduces edema and improves cognitive function following diffuse traumatic brain injury in the rat[J]. Front Neurol. 2017, 8(9):39.
- under Open Access license CC BY 4.0, without modification.
For Research Use Only.
