Creative Biolabs is a well-recognized expert in the field of antibody generation and production. Especially, we have launched a series of in vitro diagnostic (IVD) antibody/immunoassay development services for different infections and diseases. Here, we focus on the fecal lactoferrin as a biomarker of inflammatory bowel disease (IBD).
Introduction to Lactoferrin
Lactoferrin is an 80 kDa iron-binding glycoprotein of the transferrin family which plays a role in transporting serum iron. It is mainly neutrophil-derived, but may also by several mucosal epithelial cell types. It can be found in mucosal secretions and bodily fluids, but most highly in milk and colostrum. Lactoferrin is a multifunctional protein involved in several physiological functions, including antimicrobial activity against a large panel of microorganisms; immunomodulatory and anti-inflammatory activity; regulation of iron absorption in the bowel; antioxidant and anticarcinogenic properties. Considering these physiological capabilities of lactoferrin in host defense, it has been produced as a nutraceutical to exert its health benefits.
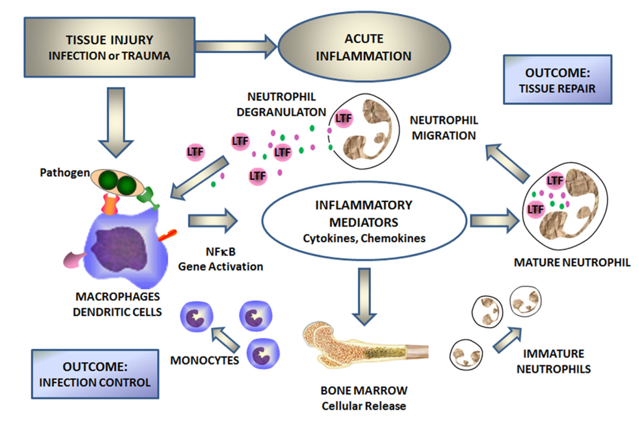 Fig.1 Lactoferrin mediates cellular responses to inflammation caused by infection and trauma.1
Fig.1 Lactoferrin mediates cellular responses to inflammation caused by infection and trauma.1
Fecal Lactoferrin as A Diagnostic Marker of IBD
In response to inflammation in the intestinal lumen, an increase in fecal lactoferrin levels occurs due to mucosal infiltration and degranulation of polymorphonuclear neutrophils. Elevated fecal lactoferrin levels have been reported in IBD with a sensitivity of 78%, and specificity of 90% in identifying inflammation in adults with ulcerative colitis (UC) and Crohn’s disease (CD). The fecal lactoferrin has been shown to correlate well with disease activity (endoscopic and histopathologic grading). Besides, it was reported that lactoferrin had good sensitivity and specificity in distinguishing IBD from irritable bowel syndrome (IBS). What’s more, lactoferrin may also be useful in monitoring IBD therapy and predicting relapses in both UC and CD.
Due to its potential as a marker of IBD, lactoferrin can be measured semi-quantitatively or quantitatively using immunoassay-based technologies, such as ELISA. Often, fecal lactoferrin is measured because samples can be collected easily and lactoferrin is stable in the stool for at least 48 hrs because of its resistance to proteolysis. Commercial tests have been developed to offer simple, sensitive, and non-invasive methods to determine intestinal inflammatory activity, to monitor treatment response in these patients, and to predict the risk of relapse.
IVD Antibody/Immunoassay Development Services Provided by Creative Biolabs
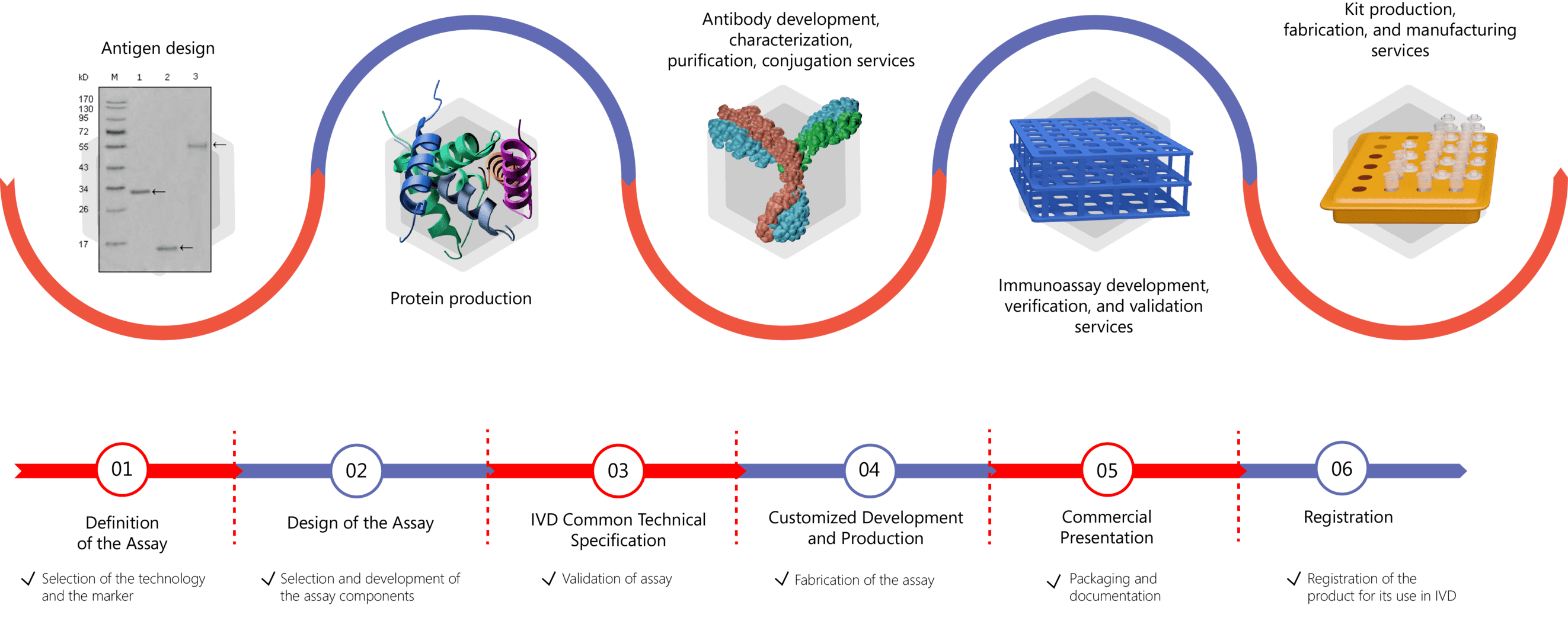
Creative Biolabs has extensive experience in IVD antibody and diagnostic immunoassay development with expertise in bioconjugate design and synthesis, antibody generation, recombinant protein expression, as well as immunodiagnostic assay development, verification, validation, and production. Our highly qualified staff, years of experience and expertise establish us as a center of excellence in IVD antibody and immunoassay kit development. Creative Biolabs offers comprehensive contract development services including:
- IVD Antibody Development
- Antibody Pair Development
- Antibody & Protein Conjugation
- IVD Immunoassay Development
Features of Our Services
- Long-standing know-how and experience across a broad array of immunoassay technology platforms
- Strong scientific background and wide-ranging and multi-disciplinary expertise
- Flexibility and adaptability to deliver services designed to suit specific demands
- Efficient customer communication and regular project updates
Please feel free to contact us to discuss how our expertise can lead to your success.
References
- Kruzel, Marian L., Michal Zimecki, and Jeffrey K. Actor. "Lactoferrin in a context of inflammation-induced pathology." Frontiers in immunology 8 (2017): 1438. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.