Creative Biolabs is a well-recognized expert in the field of antibody generation and production. Especially, we have launched a series of in vitro diagnostic (IVD) antibody and immunoassay development services targeting various disease biomarkers. Here, we focus on the Leukotriene E4 (LTE4) biomarker, the diagnostic value of which for asthma and/or other conditions has been reported in different studies.
Introduction to LTE4
LTE4 is a cysteinyl leukotriene (CysLT) that belongs to the leukotriene family of lipid mediators. It can be produced by different types of white blood cells, including eosinophils, mast cells, tissue macrophages, and basophils. The leukotriene family is derived from arachidonic acid through the 5-lipoxygenase pathway and LTE4 is formed from the sequential conversion of LTA4 to LTC4, then to LTD4, and finally LTE4. LTC4, LTD4, and LTE4 are called CysLTs but LTE4 is the most stable end product. LTE4 can be detected in biologic fluids, including urine, plasma, sputum, exhaled breath condensate, and bronchoalveolar lavage, in clinical research studies. Studies have demonstrated that measurement of urinary LTE4 can be a useful noninvasive method to assess changes in the rate of total body cysteinyl leukotriene levels.
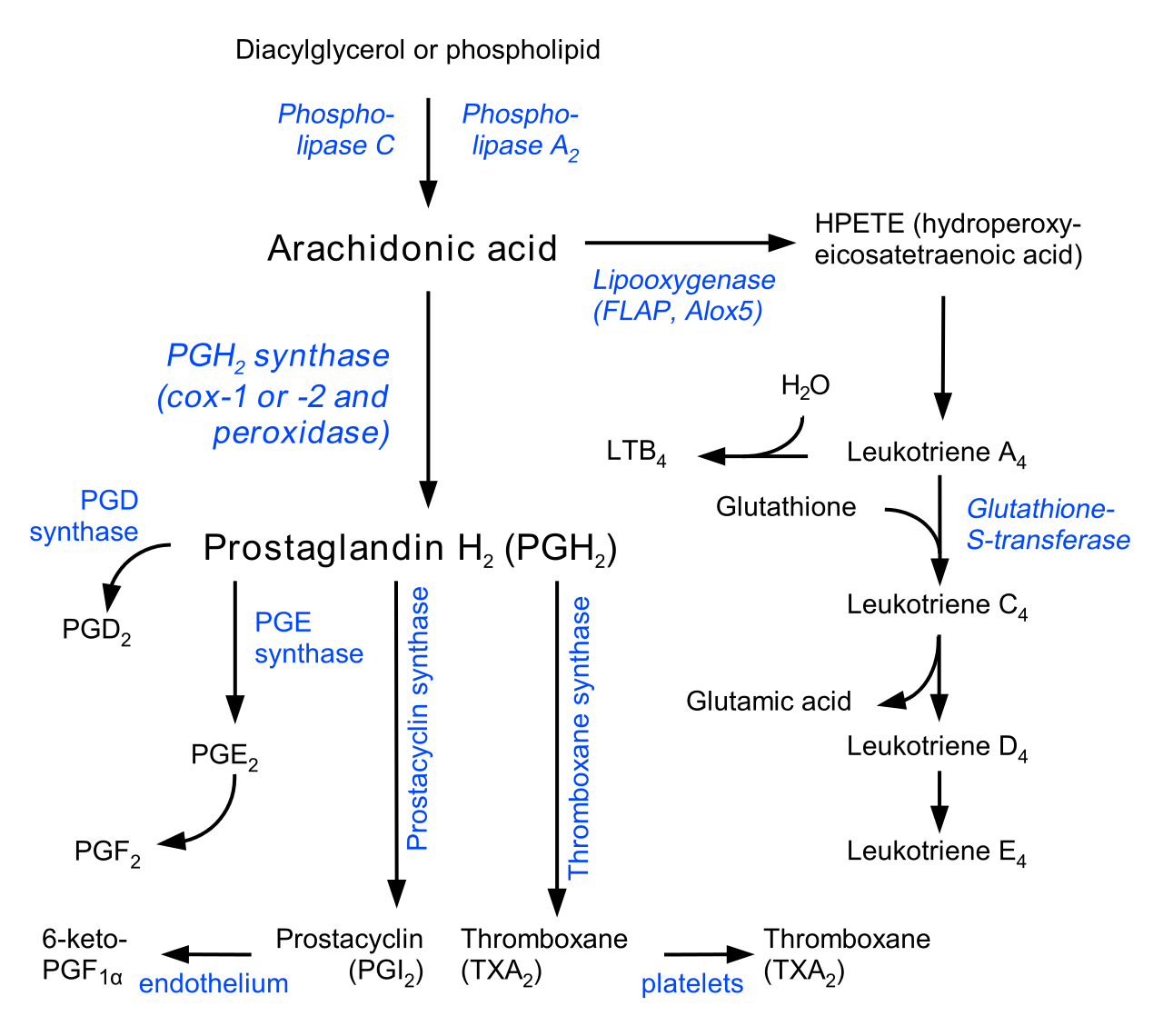 Fig.1 Synthesis of LTE4. Distributed under CC BY-SA 3.0, from Wiki, without modification.
Fig.1 Synthesis of LTE4. Distributed under CC BY-SA 3.0, from Wiki, without modification.
LTE4 as A Diagnostic Marker for Asthma
LTE4 has been shown to be a reliable biomarker in inflammatory states such as asthma, allergic rhinitis, and aspirin sensitivity. Researchers explored the diagnostic utility of 24-hour urinary LTE4 in patients with allergic rhinitis, asthma, CRS with/without nasal polyps, and aspirin sensitivity. Similarly, another research team measured the levels of urinary LTE4 in patients with chronic rhinosinusitis (CRS), asthma, and allergies using a validated immunoassay. The results showed that patients with CRS demonstrated significantly higher concentrations of urinary LTE4 than healthy controls and that comorbid asthma also correlated with elevated urinary LTE4 levels within patients with CRS. Chiu et al. (2014) investigated the utility of urinary LTE4 levels for the diagnosis of atopic diseases in early childhood, and they demonstrated that measuring urinary LTE4 provided a strategy for the diagnosis and management of asthma in preschool children.
IVD Antibody & Immunoassay Development Services for LTE4 Marker
Various methods for the detection of LTE4 in biologic fluids have been reported, including mass spectrometry, radioimmunoassay, and enzyme immunoassays. Among these, antibody-based immunoassays have been shown to be a sensitive method. At Creative Biolabs, we offer high-quality target-specific IVD antibody & immunoassay development services for global clients, covering antigen production, antibody development, labeling, and conjugation, as well as immunoassay development. For more information, please click the following links:
- IVD Antibody Development
- Antibody Pair Development
- Antibody & Protein Conjugation
- IVD Immunoassay Development
Contact us to discuss your project and experience the great value of our services.
References
- Chiu, C. Y., (2014). “Urinary LTE4 levels as a diagnostic marker for IgE-mediated asthma in preschool children: a birth cohort study.” PLoS One, 9(12). Distributed under Open Access license CC BY 4.0, mentioned by summarizing some parts of the article.
For Research Use Only.