Knobless Adenovirus Vector Construction Service
Recombinant adenoviral (Ad) vectors are being used as gene delivery vectors in a variety of gene therapy strategies. However, the lack of specific targeting of Ad vectors in vivo may inhibit the efficient transduction into cells of the therapeutic gene. This has led to the need for high vector dose transduction, increasing the risk of unwanted side effects during gene therapy. Therefore, if a particular gene is only transferred to the desired target cells, the Ad vector may be greatly improved. Creative Biolabs is doing this work to improve the effectiveness of gene therapy by modifying the Ad capsid
Introduction of Knobless Ad Vector
Ad vector systems for gene therapy can be greatly improved by targeting specific cell types of vectors. It has been previously determined that the Hi ring of the fibrous nodal domain is the preferred site for targeting ligand binding. The structural properties of the Hi ring allow the insertion of multiple ligands, including large polypeptide molecules. Previous studies have determined that the structure of Ad fibrin imposes strict size restrictions on ligands bound to its carboxyl end, thus limiting the range of potential candidate ligands and making them short peptides. Therefore, in order to eliminate this restriction, it is necessary to completely eliminate the tendency of natural adenovirus and introduce a new binding affinity into the capsid of the virus. Current studies have shown that these requirements can be met by deleting the entire knob domain of adenovirus fibrin and replacing it with two distinct parts, providing trimerization of knobless fibers and specific binding with target cells, respectively. Hence, these knobless fibers are prototype substrates for multi-purpose addition of targeting ligands to produce truly targeted adenoviruses.
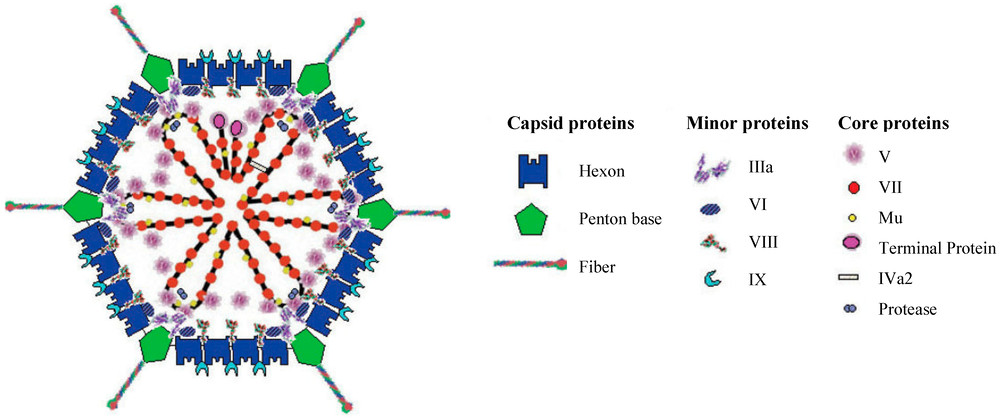 Figure 1. Adenovirus Structure. Schematic representation of the capsid and core proteins of an adenovirus.1
Figure 1. Adenovirus Structure. Schematic representation of the capsid and core proteins of an adenovirus.1
Knobless Ad Vector Construction Services
The way we develop knobless Ad vector is to delete the entire fiber knob and replace it with two different protein parts, which provide the trimerization function for the knobless fiber and specifically bind to target cells. Scientists have constructed an Ad vector carrying knobless fibers containing the alpha-helix trimerization domain of glycoprotein encapsulated by Moloney murine leukemia virus. Two simulated targeting ligands, Myc-epitope and 6*His-tag, are linked by the flexible conjugated peptide. The results showed that targeted knobless fiber molecules were correctly expressed and introduced into the nucleus of adenovirus packaging cells, where they were incorporated into the adenovirus capsid as functional trimers. Both ligands are exposed to the surface of viral particles and can be used to specifically bind to their target molecules. In addition, knobless fibers mediate gene delivery to cells that display coupled ligand receptors.
The advantages of our non-binding adenoviral vector include:
- Increased range of potential ligand candidates and short peptides;
- As a multi-functional prototype substrate for adding targeted ligands to produce truly targeted adenoviruses;
- Improving the targeting of Ad vectors and the efficacy of adenovirus-based gene therapy.
Core Advantages of Our Knobless Ad Vectors
Our engineered knobless adenovirus vectors combine structural innovation with functional optimization, delivering unparalleled advantages over traditional Ad vectors and other engineered variants, making them the optimal choice for precision gene therapy:
Expanded range of targeting ligand candidates
By eliminating the size constraints of the natural knob domain, we unlock the potential to use not only short peptides but also large polypeptide molecules, protein ligands, and fusion proteins as targeting moieties. This drastically expands the pool of available targeting molecules, allowing you to select the most optimal ligand for your specific target cell type and significantly improving the specificity and efficiency of targeting.
Flexible multi-targeting design capability
The Hi ring of the knobless fiber supports the insertion of multiple distinct targeting ligands, enabling the construction of multi-targeted adenoviral vectors. This is particularly valuable for the treatment of complex diseases such as heterogeneous tumors, where multiple cell subtypes need to be targeted simultaneously, and for combating drug resistance by targeting multiple signaling pathways.
Enhanced gene therapy efficacy and safety
Precise targeting of therapeutic genes to diseased cells maximizes the concentration of the therapeutic gene at the disease site, improving the efficiency of gene transduction and expression. At the same time, specific targeting reduces the non-specific binding of the vector to normal cells, allowing for a significant reduction in vector dosage. This not only lowers the risk of immune responses and off-target side effects but also addresses the clinical challenge of high-dose toxicity associated with traditional Ad vectors.
Stable and functional structural integration
Our modified knobless fibers form stable, functional trimers that are efficiently incorporated into the adenoviral capsid, maintaining the structural integrity and infectivity of the viral particle. Targeting ligands are stably exposed on the viral surface, ensuring reliable and sustained binding to target cell receptors—avoiding the structural instability and functional loss common in other engineered Ad vector variants.
Versatile prototype substrate for customized engineering
Knobless fibers serve as a universal and modifiable scaffold for the addition of targeting ligands. We can flexibly modify and optimize the vector according to your research needs, including adjusting ligand types, linker peptides, and trimerization modules—providing a highly customizable platform for the development of personalized gene therapy strategies.
Knobless Adenovirus Vector Construction Strategy
Our Knobless adenovirus vector construction strategy does not merely modify this interaction; it eliminates it entirely. The process involves a precise genetic re-engineering of the viral capsid:
Instead of attempting to insert targeting ligands into the structurally constrained "Hi loop" of the fiber—which limits ligand size to short peptides—we take a more radical and effective approach. We delete the entire knob domain from the adenovirus fiber protein. This deletion, however, renders the fiber incapable of trimerization, a process essential for its stability and incorporation into the viral capsid.
To resolve this, we replace the deleted knob with two distinct, functional protein components:
- An Exogenous Trimerization Domain: We introduce a heterologous trimerization motif, such as the alpha-helical domain from the Moloney murine leukemia virus (MoMuLV) envelope glycoprotein. This domain efficiently drives the trimerization of the modified, knobless fiber, ensuring proper structural integrity and assembly into the virus particle.
- A Targeting Ligand: This is the customizable "navigation system" you define. The chosen ligand—which can range from short peptides and single-chain antibodies (scFvs) to DARPins and full-length antibody domains—is genetically fused to the trimerization domain via a flexible peptide linker. This ensures the ligand is correctly displayed on the mature virus surface, free from the steric hindrances of the original knob structure.
Features of Our Knobless Adenovirus Platform
Choosing Creative Biolabs' Knobless Adenovirus Vector Construction Service provides you with a next-generation delivery platform that offers unparalleled safety, flexibility, and efficacy:
1. Eliminated Natural Tropism for Maximum Safety
- Reduced Off-Target Toxicity: By completely abrogating CAR-mediated binding, you can virtually eliminate non-specific transduction of healthy tissues, particularly the liver. This dramatically widens the therapeutic window and reduces the risk of systemic side effects.
- Enabling Safe Systemic Administration: Freed from rapid sequestration by non-target cells, knobless vectors gain the potential for effective and safe intravenous (IV) administration, opening doors for treating metastatic cancers or systemic genetic disorders.
2. Unlocked Targeting Flexibility for Any Ligand
Conventional insertion sites on the fiber knob impose severe size restrictions on targeting moieties. Our knobless design completely liberates you from these constraints. Our platform allows for the seamless incorporation of ligands ranging from small peptides to large, complex protein domains (e.g., full-length antibodies). This vastly expands the range of potential targets you can engage, enabling strategies that were previously impossible.
3. A True Platform for Genuinely Targeted Vectors
The knobless vector functions as an ideal "multifunctional prototype substrate." Because all cell binding is mediated solely by the engineered ligand, you can create a virus that truly infects only your cell type of interest. This platform can be readily integrated with the latest advances in targeted therapeutics, such as bispecific antibodies or immune cell engagers, to create sophisticated, next-generation gene therapies.
4. Potential for Reduced Immunogenicity
By removing the immunodominant knob domain, knobless vectors may partially evade pre-existing neutralizing antibodies present in the human population due to natural adenovirus infections. Furthermore, the reduction in non-specific uptake by antigen-presenting cells could lead to a blunted innate immune response, further improving the safety and efficacy profile of your therapy.
5. Increased Range of Potential Ligand Candidates
Unlike conventional vectors limited to short peptides, our platform accepts a wide variety of ligands. This allows researchers to utilize high-affinity, highly specific binders developed through various discovery platforms, ensuring robust and selective targeting from the outset.
Frequently Asked Questions
Q: How does the yield of knobless adenoviruses compare to conventional ones?
A: With the use of specialized CAR-expressing packaging cell lines and our optimized production protocols, we are able to achieve yields that are comparable to high-yielding conventional adenovirus preparations. We will work with you to define expected yields based on your specific construct and project scale.
Q: How do you confirm that my targeting ligand is correctly displayed and functional?
A: Functional validation is a key part of our service. We perform in vitro transduction assays on a panel of cell lines, including those that are positive and negative for your ligand's target receptor. By measuring reporter gene expression or vector genome copy numbers, we can quantitatively confirm the specificity and efficiency of your ligand-mediated transduction.
Q: Can this technology be applied to different adenovirus serotypes?
A: Yes, absolutely. While Ad5 is the most common serotype used, our platform is adaptable. We can construct knobless vectors based on other serotypes, such as Ad35, which may offer advantages in evading pre-existing immunity or targeting different cell populations.
Q: What QC data will I receive with my final knobless adenovirus?
A: Upon project completion, you will receive a comprehensive QC report. This typically includes: physical and infectious titers, results from knob-deletion and structural validation (PCR/Sequencing/Western Blot), a certificate of analysis for sterility and endotoxin levels, and the functional validation data demonstrating targeting specificity. We are also happy to discuss additional QC assays to meet your specific needs.
Partner with Us
If you are looking for a reliable and professional partner for your adenovirus-based gene therapy research, Creative Biolabs is your best choice. Our knobless adenovirus vector construction service will provide you with a precision-targeted gene delivery platform, empowering your gene therapy projects to achieve breakthrough results.
Reference
- Coughlan L, Alba R, Parker A L, et al. Tropism-modification strategies for targeted gene delivery using adenoviral vectors[J]. Viruses, 2010, 2(10): 2290-2355. https://doi.org/10.3390/v2102290 Distributed under Open Access license CC BY 4.0, without modification.
