Regulated Adenovirus Vector Construction
The virus represents an appealing tool for therapeutic gene transfer because of its high transfection efficiency in a wide range of human cell types. Approximately 70% of the vectors used in gene therapies are performed by viral-based delivery systems. As it belongs to a pathogenic agent, the virus needs to be attenuated to be safely applied in research applications. To this end, Creative Biolabs has designed virus-derived vectors that stem from different viral classes, including adenovirus, adeno-associated virus (AAV), lentiviruses, and retroviruses. Here, we offer a multitude of regulated adenovirus vector construction services to achieve gene transfer properly, particularly specific cell-targeted constructions, specific cancer-targeted constructions, and inducible constructions.
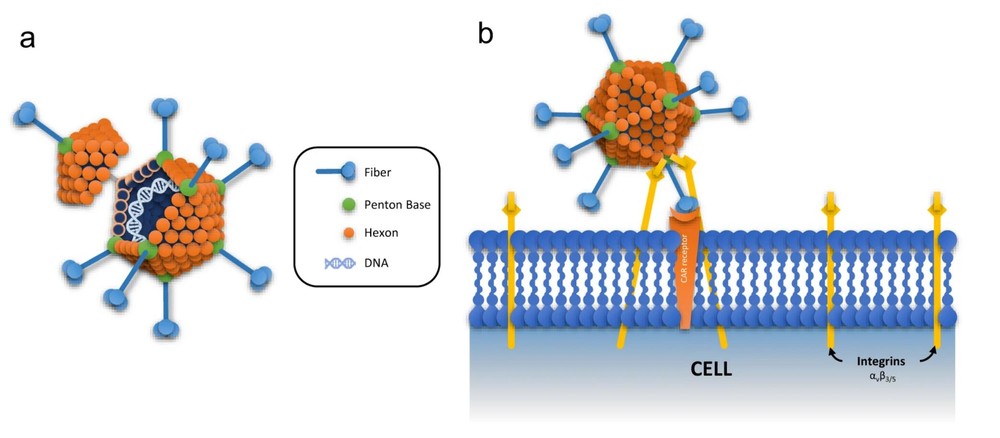 Figure. 1: Adenovirus structure organization and interaction with host cell.1
Figure. 1: Adenovirus structure organization and interaction with host cell.1
Regulated Modification
Adenovirus is a double-stranded DNA virus and is a member of Group I family in Baltimore classification. It is a non-enveloped icosahedral virus which carries a medium-sized genome of 35 kb, harboring numerous essential genes that determine its biological activities and distinguishable serotypes. So far, there're seven species of adenoviruses (A to G), which can be further divided into 57 serotypes (HAdV-1 to 57). To generate non-replicative yet infective adenoviral vectors, helper cells are required to be transfected with vectors and express key proteins for efficient vector production. Up to now, HEK293T cell lines are found the most prominent to work as helper cells. They're specific lab-adapted immortalized cells and can be easily transfected with vector-packaging plasmids, showing robust protein output and ideal titered adenoviral vectors. Recombinant adenoviral vectors are versatile, easily-regulated systems for gene delivery and expression in mammalian cells.
The Challenge: From a Blunt Tool to a Precision Instrument
Despite their advantages, traditional first-generation adenovirus vectors lack specificity. They transduce cells indiscriminately, which presents two major hurdles:
- The Safety Challenge: High immunogenicity and off-target transduction can lead to significant toxicity and inflammatory responses, limiting therapeutic windows.
- The Efficacy Challenge: In cancer therapy, a non-specific virus dilutes its therapeutic effect in healthy tissue, failing to concentrate the "payload" where it is needed most.
The solution lies in "Regulation." By engineering the virus at the genetic level, we transform it from a simple delivery tool into a "smart" therapeutic agent that acts only in designated cells, at specific times, or under particular environmental conditions.
Regulated Adenovirus Vector Construction Services
Adenoviral vectors for gene therapy can be significantly enhanced by targeting vectors to specific cell types or disease classifications. This needs the complete elimination of native adenovirus tropism and the introduction of a new binding affinity in viral capsids. Meanwhile, adenoviruses can be altered to selectively infect or replicate in cancer cells. To further improve targeting ability, cell-killing capacity and hiding talent of the virus from host immunity, we focus on the modifications of adenoviral E1 region and the systems allowing for genome-wide modification.
High transduction efficiency and short-term gene expression make adenoviral vector a preferred instrument for in vivo gene delivery. Creative Biolabs has developed a series of technologies to improve adenoviral packaging protocols in terms of purity, titer, viability, and consistency. Our regulated adenovirus vector construction services include but not limited to the following items.
Specific Cell-targeted Modifications
- Liver-targeting Adenovirus Vector Construction
- Erythroid Cells-targeting Adenovirus Vector Construction
- Pituitary-targeting Adenovirus Vector Construction
- Melanocytes-targeting Adenovirus Vector Construction
- Muscle-targeting Adenovirus Vector Construction
- Glial Cells-targeting Adenovirus Vector Construction
- Neurons-targeting Adenovirus Vector Construction
Specific Cancer-targeted Modifications
- Hepatocellular carcinoma-targeting Adenovirus Vector Construction
- Pancreatic Cancer-targeting Adenovirus Vector Construction
- Colorectal Carcinoma-targeting Adenovirus Vector Construction
- Glioma-targeting Adenovirus Vector Construction
- Breast Cancer-targeting Adenovirus Vector Construction
- Lung Cancer-targeting Adenovirus Vector Construction
- Epithelial Tumor-targeting Adenovirus Vector Construction
Inducible Modifications
- Radiation-induced Adenovirus Vector Construction
- Heat-induced Adenovirus Vector Construction
- FK506/Rapamycin-Inducible Adenovirus Vector Construction
- Hypoxia Inducible Adenovirus Vector Construction
- Tetracycline Inducible Adenovirus Vector Constructio
Core Regulatory Technologies Comparison
Table 1: Core Regulatory Technologies.
| Technology Type | Modification Strategy | Mechanism of Action | Key Applications |
|---|---|---|---|
| Cell-Specific Targeting | Fiber/hexon protein engineering; incorporation of cell-type specific binding domains | Redirects vector binding to cell-type specific receptors | Basic research on cell-type specific gene functions; tissue-specific gene therapy |
| Cancer-Specific Targeting |
1. Tumor-specific promoter-driven transgene expression 2. Tumor receptor-binding ligand modification 3. Conditional replicative adenovirus (CRAd) design |
1. Restricts transgene/replication to tumor cells via promoter selectivity 2. Enhances tumor cell binding via overexpressed receptors 3. Selective replication and lysis of cancer cells |
Oncolytic virotherapy; targeted immunomodulator delivery; tumor microenvironment engineering |
| Inducible Expression Systems |
1. Small molecule-inducible switches (Tet-On/Off, Tamoxifen-responsive) 2. Stimulus-responsive promoters (hypoxia, radiation, inflammatory cytokines) |
1. Temporal/spatial control via exogenous small molecules 2. Activation by pathological/physical cues in target tissues |
Controlled gene expression studies; prodrug activation therapy; combination treatment regimens |
Service Workflow & Deliverables
Our process is designed for transparency and rigor, ensuring you receive a product perfectly suited for its intended application.
Workflow Diagram (Visual Flowchart):
-
Consultation & Strategy Design
- In-depth discussion of your research goals and target cell type.
- In silico design of the optimal regulation strategy (promoter selection, targeting peptide, inducer system).
- Deliverable: Detailed vector design proposal and timeline.
-
Vector Cloning & Verification
- Synthesis/amplification of regulatory elements and GOI.
- Subcloning into a shuttle vector and homologous recombination into an E1/E3-deleted AdV backbone (e.g., Ad5).
- Deliverable: Sequence-verified plasmid DNA and detailed maps.
-
Virus Rescue, Amplification & Purification
- Transfection of linearized genome into packaging cells (e.g., HEK293) for virus rescue.
- Small-scale amplification followed by large-scale expansion.
- Purification via two rounds of CsCl density gradient ultracentrifugation or advanced column chromatography to ensure high purity and removal of replication-competent adenoviruses (RCAs).
-
Comprehensive Quality Control & Functional Validation
- Titration: Infectious titer by TCID50/PFU assay and physical titer by qPCR/OD260.
- Purity & Safety: HPLC/SDS-PAGE for purity, endotoxin and sterility testing, RCA assay.
- Functional Validation: In vitro testing in permissive and target cells to confirm regulation specificity (e.g., GFP expression, cell killing, target gene expression).
Final Deliverables
| Deliverable Item | Specification & Description |
|---|---|
| High-Titer Viral Stock | Purified, regulated adenovirus; ≥ 1×10¹¹ PFU/mL (or ≥ 1×10¹² VP/mL). Formulated for your application (e.g., in vivo grade). |
| Certificate of Analysis (CoA) | Detailed report including infectious titer, physical titer, purity (SDS-PAGE), sterility, and endotoxin levels. |
| Technical Report | Full methodology, vector map, and sequencing data. Includes functional validation data from our QC process. |
Technical Advantages & Differentiators
- Deep Customization: We offer true "boutique" design. Whether you need a specific promoter from literature, a novel targeting peptide, or a complex dual-regulated system, our platform can accommodate it.
- Advanced Backbone Technologies: We utilize next-generation AdV backbones (including E1/E3 deleted and helper-dependent "gutless" vectors) with larger cargo capacities (~7.5 kb to >30 kb) and significantly reduced immunogenicity, ensuring your therapeutic gene is the primary focus.
- Stringent RCA Testing: Safety is paramount. Our production processes are optimized to minimize the generation of Replication Competent Adenoviruses (RCA), and we offer rigorous RCA testing to meet the highest standards for preclinical and clinical research.
- Integrated One-Stop Shop: From initial vector design and construction to high-titer production and downstream in vitro/in vivo efficacy testing, Creative Biolabs provides continuous support, streamlining your project management and accelerating your timeline.
Broad Applications
Our regulated adenovirus vectors are enabling breakthroughs across multiple fields:
- Oncology Gene Therapy: Conditionally replicating oncolytic viruses, targeted expression of immunomodulators (e.g., cytokines, checkpoint inhibitors), and CAR-T cell generation.
- Vaccine Development: Rapid construction of adenovirus-based vaccine candidates expressing antigens from emerging pathogens like influenza or SARS-CoV-2.
- Genetic & Rare Diseases: Targeted delivery and regulated expression of therapeutic genes to minimize systemic side effects.
-
Basic & Translational Research:
- Neuroscience: Cell-type specific expression of opsins, calcium indicators, or chemogenetic receptors (DREADDs).
- Immunology: Studying gene function in specific immune cell subsets in vivo.
- Metabolic Disorders: Regulated expression of enzymes for pathway analysis.
Featured Case Studies
Case Study 1: A Dual-Regulated Oncolytic Adenovirus for Hepatocellular Carcinoma
- Challenge: A client required a virus that would replicate and express a therapeutic transgene only in liver cancer cells.
- Our Solution: We constructed an oncolytic adenovirus with a dual-regulation mechanism. The E1A gene was placed under the control of the AFP promoter (transcriptional targeting), and the virus fiber was modified with a peptide targeting Glypican-3, a receptor highly expressed on liver cancer cells (physical targeting).
- Result: The virus demonstrated potent, selective cytotoxicity in AFP+/GPC3+ hepatocellular carcinoma cell lines, with no observable effect on AFP-negative primary human hepatocytes. Tumor burden in a xenograft mouse model was significantly reduced.
Case Study 2: An Inflammation-Inducible Vector for Treating Acute Lung Injury
- Challenge: To create a vector that produces an anti-inflammatory protein only when and where inflammation is occurring, avoiding systemic immunosuppression.
- Our Solution: We utilized a C3 promoter, which is activated by inflammatory signals like LPS and influenza virus, to drive expression of the HMGB1 antagonist, Box A. This "inflammation-inducible" cassette was cloned into a non-replicative AdV vector.
- Result: In an influenza-infected mouse model, therapeutic administration of AdV.C3-Box A significantly mitigated lung and systemic inflammation by producing the antagonist only in response to the ongoing viral infection, demonstrating a powerful new approach for treating inflammatory diseases.
Frequently Asked Questions
Q1: What is the key difference between regulated and non-regulated adenoviral vectors?
A: Regulated vectors incorporate additional control elements (tumor-specific promoters, inducible switches, targeting ligands) that restrict transgene expression or vector replication to specific cell types, tissues, or conditions. Non-regulated vectors express transgenes constitutively in all transduced cells, increasing off-target effects and immunogenicity risks.
Q2: How do I choose the optimal regulatory element for my research?
A: Our expert team will guide you based on:
- Target cell/tissue type (e.g., AFP promoter for liver cancer, Synapsin promoter for neurons)
- Desired expression kinetics (e.g., Tet-On for temporal control, hypoxia-responsive for conditional activation)
- Application (e.g., oncolytic vectors require replication-regulating elements; vaccines prioritize high immunogenicity)
Q3: What is the maximum packaging capacity of your adenoviral vectors?
A: Our third-generation E1/E3-deleted vectors can accommodate up to 8 kb of exogenous DNA. For larger inserts, we offer helper-dependent adenoviral vectors (HDAd) with a packaging capacity of up to 36 kb (custom service).
Q4: Can the delivered virus be directly used for animal experiments?
A: Yes. All vectors undergo strict endotoxin testing (/mL) and sterility validation, meeting the standards for in vivo use. We also provide custom formulations (e.g., PBS with 10% glycerol) for optimal stability during animal administration.
Partner with Us
Due to the basic progress in elucidating molecular mechanisms of human diseases and the arrival of post-genomic time, a number of cellular targets and therapeutic genes are available for gene therapy in clinical trials. As a famous biological company in the world, Creative Biolabs is committed to developing gene delivery vectors with high efficiency through target cell selectivity, who has already established the most widely used and regulated adenoviral vector systems. Based on our high reputation and professional services, a growing base of clients come back to us for more adenoviral vector cloning and packaging projects. For more information, please feel free to contact us.
Reference
- Mendonça S A, Lorincz R, Boucher P, et al. Adenoviral vector vaccine platforms in the SARS-CoV-2 pandemic. npj Vaccines, 2021, 6(1): 97.https://doi.org/10.1038/s41541-021-00356-x Distributed under Open Access license CC BY 4.0, without modification.
