AAV Vector Design for Hemophilia A/B
Hemophilia A/B is a group of hemorrhagic diseases with hereditary coagulopathy. Its common feature is the active thromboplastin-producing disorder with prolonged clotting time, and a tendency to post-traumatic hemorrhage in life. Patients often show a lack of the F8 (hemophilia A) or F9 (hemophilia B) genes, based on which, scientists focus on gene therapy to prolong patient's survival time. The most typical case in clinical research is to design adeno-associated virus (AAV) vector, a single-stranded small DNA virus, for the delivery of factor VIII or IX to hepatocytes. Creative Biolabs provides a full range of technical services, including vector preparation, gene loading, gene expression and efficacy validation to satisfy your needs.
Research Progress of Hemophilia
Hemophilia is a clinically recurrent bleeding disease with a high mortality rate. Since the first distinguish of the genes F8 (hemophilia A) or F9 (hemophilia B) in the 1950s, gene therapy of Hemophilia has made great progress. The first clinical treatment for hemophilia A was by intravenous injection of AAV vectors to inject in the omentum during a laparoscopic procedure. The entire process did not achieve long-term results, although it was non-toxic for patients. However, for hemophilia B, doctors transplanted the AAV vectors into the liver of the patient by interventional therapy, with clinical efficacy lasted for several weeks, which lays a solid foundation for subsequent liver transmission.
- Hemophilia A
Gene therapy for hemophilia A is still in the early stages, which needs further research and development by researchers. It has been reported that optimizing codons in the coding region can increase gene F8 expression by up to 40 times, however, this non-wild-type sequence tends to stimulate the antigenicity of the body. Fortunately, the design of AAV vectors for liver-mediated gene therapy has made some progress in hemophilia A dogs.
- Hemophilia B
The size of hemophilia B is about 4.4 kb even for the B-domain-deleted construct, however, due to the structure of AAV itself, AAV vectors cannot insert more than 5 kb genes, which has become a drawback for AAV vectors in the treatment of hemophilia B. Investigators at St Jude Children's Research Hospital and University College London packaged a vector genome into an AAV8 capsid, promoting a significant increase in factor F9 levels, which efficiently relieves the patient's bleeding condition and reduces the pain of the patient. Moreover, scientists attribute the massive replication of the factor F9 to the activation of T cells by the AAV vector, which causes the body to produce strong immune responses. These results fully demonstrate that AAV vectors can be used for gene therapy of hemophilia B and have achieved objective results.
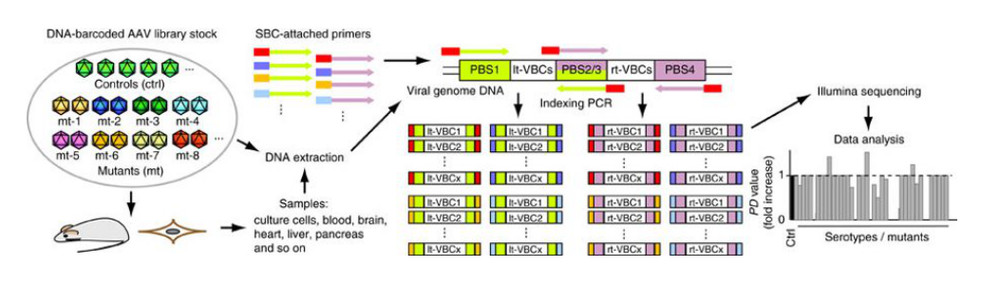 Figure 1. Procedure for the AAV Barcode-Seq analysis.1
Figure 1. Procedure for the AAV Barcode-Seq analysis.1
Why Hemophilia Is an Ideal Target for Gene Therapy
Hemophilia has emerged as one of the most successful and clinically advanced applications of gene therapy due to several intrinsic biological advantages:
1. Monogenic Disease with Well-Defined Targets
Hemophilia A and B are caused by mutations in single genes-F8 and F9-making them ideal candidates for gene replacement strategies.
2. Low Therapeutic Threshold
Even modest increases in clotting factor levels (e.g., 5–10%) can significantly reduce bleeding risk and convert severe disease into a mild phenotype.
3. Liver as a Target Organ
Hepatocytes are natural producers of clotting factors and are highly amenable to gene delivery, particularly via viral vectors.
4. Proven Clinical Feasibility
Multiple AAV-based gene therapies have demonstrated durable expression and reduced bleeding rates in clinical trials, with some achieving near-normal clotting factor levels.
Our Services: Integrated AAV Design Solutions for Hemophilia
As a R&D partner, Creative Biolabs offers a comprehensive suite of customized services. We provide a seamless transition from initial target sequence design to product generation, ensuring every step of your Hemophilia A/B pipeline is executed with precision.
AAV Vector Production & Scale-Up
- Research to Preclinical Scale: Flexible production volumes utilizing adherent/suspension HEK293 triple-transfection or Baculovirus/Sf9 systems.
- Advanced Downstream Purification: State-of-the-art affinity and anion exchange chromatography (AEX) processes designed to maximize "full capsid" enrichment (typically >80-90%).
- Strict Batch Consistency: Robust standard operating procedures (SOPs) ensuring high reproducibility across multiple production runs.
Analytical Testing & Quality Control (QC)
- Genomic Titering: Ultra-precise physical titer quantification utilizing Droplet Digital PCR (ddPCR).
- Empty/Full Ratio Analysis: High-resolution profiling using Analytical Ultracentrifugation (AUC) or SEC-MALS.
- Identity & Purity: Rigorous validation of VP1: VP2: VP3 capsid protein ratios via CE-SDS and SDS-PAGE.
- Safety & Impurities: Comprehensive testing for Host Cell Protein (HCP), residual hcDNA, endotoxins, and mycoplasma.
Vector Engineering for Hemophilia
| Challenge | Our Solution |
|---|---|
| FVIII cDNA too large for AAV | Dual AAV vectors (trans-splicing, overlapping, or hybrid); alternative compact FVIII variants (e.g., B-domain deleted FVIII, FVIII-X10) |
| Poor FIX expression | Optimized liver-specific promoters (LP1, TBG); synthetic introns; WPRE enhancer; codon optimization for human/mouse |
| Anti-capsid antibodies | Capsid engineering (AAV2, AAV5, AAV8, AAV9, AAV-LK03, AAV-Spark100); capsid shuffling; detargeting mutations to evade pre-existing immunity |
State-of-the-Art Gene Editing for Hemophilia
For clients pursuing permanent correction or targeted integration:
- CRISPR-Cas9: Insert FVIII/FIX into the Albumin or AAVS1 safe harbor locus in hepatocytes
- Base editing & Prime editing: Correct point mutations (e.g., FIX-Padua, common FVIII missense mutations) without double-strand breaks
- Non-viral episomal vectors (e.g., Sleeping Beauty, PiggyBac) for sustained expression without AAV limitations
Why Do Global Scientists Choose Creative Biolabs?
When you choose Creative Biolabs, you are not just buying an outsourced service; you are integrating an experienced external R&D think tank.
- Deep Wet-Lab Expertise: Unlike computational biology platforms that focus on virtual simulations, we have decades of hands-on experience in vector construction, cell culture, and animal surgery. We solve real-world problems that happen on the bench, such as plasmid recombination, protein non-expression, and low viral purification yields.
- Unmatched Customizability: We do not offer rigid, one-size-fits-all packages. From promoter screening combinations to the optimization of administration routes, all experimental protocols are co-customized with you by our senior scientists (Ph.D. level experts).
- Time & Cost Efficiency: Leveraging mature platform processes and a massive library of ready-to-use elements, we shorten the traditional AAV R&D cycle by nearly 30%, helping your project gain a head start in the fiercely competitive gene therapy race.
A Glance at Our Success (Case Study)
Case Brief: Optimizing Long-Term Expression of AAV8-BDD-FVIII in a Hemophilia A Mouse Model
Client Pain Point: A startup biotech company found that their proprietary AAV vector design resulted in a rapid decline in FVIII activity within 4 weeks after mouse injection, accompanied by severe liver transaminase elevation.
Our Intervention: Upon taking over the project, the Creative Biolabs scientific team first conducted an off-target effect analysis, discovering insufficient promoter specificity. We redesigned the sequence with a highly specific TTR promoter and employed a dual-purification process, removing the 60% empty capsids present in the previous samples.
Delivered Results: The optimized novel vector was injected into FVIII-KO mice at a low dose of 2 × 10¹² vg/kg. After 12 weeks, plasma FVIII activity stably maintained at approximately 15% of normal levels, with no significant ALT/AST elevation or antibody production observed. The client successfully secured A funding backed.
What Our Clients Say

Frequently Asked Questions (FAQ)
Q: How do you solve the AAV vector packaging limit issue when treating Hemophilia A?
A: Our standard strategy is to use a B-Domain Deleted (BDD) FVIII, which shortens the gene to a size suitable for packaging. Simultaneously, we streamline the promoter and PolyA sequences during design, ensuring the entire Expression Cassette is strictly kept under 4.7 kb. This avoids the generation of truncated viral genomes and guarantees packaging efficiency and purity.
Q: Which AAV serotype do you recommend for Hemophilia B?
A: While AAV8 is currently the most widely used in clinical trials due to its excellent hepatotropism, we can also provide AAV5, AAV9, or proprietary engineered mutant capsids based on your specific needs. These novel capsids offer significant advantages in evading pre-existing neutralizing antibodies.
Q: What Empty/Full ratio can you achieve for the AAV vectors you produce?
A: Through our optimized multi-step chromatographic purification processes (including affinity capture and ion-exchange gradient elution), we guarantee to increase the Full Capsids percentage to over 80% - 90% for most serotypes. This far exceeds the industry average, providing the highest safety margin for your animal experiments.
Q: Why are AAV vectors commonly used in hemophilia gene therapy?
A: AAV vectors are non-pathogenic and capable of delivering genes efficiently to liver cells. They enable long-term expression of clotting factors with a favorable safety profile, making them the preferred delivery system.
Q: Can gene therapy provide a permanent cure for hemophilia?
A: Gene therapy has the potential to provide long-term therapeutic benefit with a single treatment. However, durability varies, and ongoing research aims to achieve stable, lifelong expression.
Contact US
With the rapid development of gene therapy, significant results have been achieved with the use of AAV vectors for hemophilia therapy. Creative Biolabs owns the most scientific AAV vector design and preparation methods. We will offer the best services to support the theoretical basis and scientific guidance in the clinical treatment of hemophilia. For more information, please contact us in time and we will be happy to serve you.
Reference
- Adachi, K.; et al. (2014). Drawing a high-resolution functional map of adeno-associated virus capsid by massively parallel sequencing. Nature Communications. 5: 3075-3099. https://doi.org/10.1038/ncomms4075 Distributed under Open Access license CC BY 4.0, without modification.
