Product List Background Trypanosoma cruzi-Laminin Parasite Receptor Aptamer Analysis
Background
Trypanosoma cruzi (T. cruzi), the causative agent of Chagas disease, is an obligate intracellular parasite transmitted to vertebrate hosts via blood-sucking insects. The interaction between T. cruzi and mammalian host cells encompasses various molecules from both organisms. Like other intracellular pathogens, T. cruzi is internalized via multiple endocytic pathways. Among the molecules involved in this invasion process are mucins, major surface glycoproteins of T. cruzi, which act as ligands through their sugar residues. Additional adhesion molecules include trans-sialidases (both active and inactive) and glycoproteins. Host cell molecules such as laminin and integrins serve as receptors, facilitating the binding of T. cruzi to host cells. Aptamers targeting the T. cruzi-laminin parasite receptor may promote the development of drugs that inhibit cell infection by the parasite, offering a novel approach to treating the acute phase of Chagas disease.
Signaling Pathways Activated During T. cruzi Cellular Entry
Several surface molecules on tissue culture-derived trypomastigotes, which have an affinity for the extracellular matrix, play roles in host cell invasion, yet their signal-inducing properties remain underexplored. Among these molecules is a laminin-binding glycoprotein, part of the gp85/trans-sialidase superfamily. Peptides based on FLY bind to CK18 on LLC-MK epithelial cells, promoting CK18 dephosphorylation and reorganization and activating the ERK1/2 signaling pathway, enhancing trypomastigote internalization. In metacyclic forms, gp82-mediated entry triggers PLC, generating DAG and IP3, stimulating PKC and promoting Ca2+ release. PI3K and PTK activation, leading to p175 phosphorylation, also occurs. A Ca2+ agonist from parasite OPB and bradykinin from trypomastigote cruzipain bind to their respective receptors, activating PLC and subsequent IP3-mediated Ca2+ release.
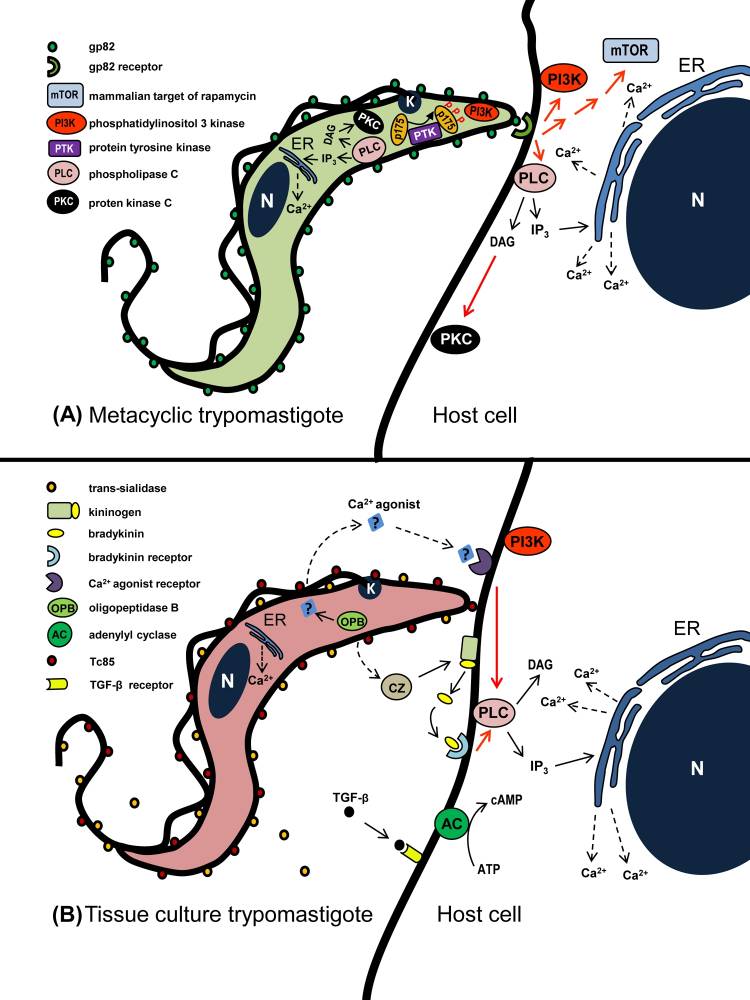 Fig.1 Schematic overview of signaling molecules and pathways activated during T. cruzi entry into nonphagocytic mammalian cells.1,3
Fig.1 Schematic overview of signaling molecules and pathways activated during T. cruzi entry into nonphagocytic mammalian cells.1,3
T. cruzi-Laminin Parasite Receptor Products Utilizations
Chagas disease impacts an estimated 5 million people globally. The causative agent, the intracellular parasite T. cruzi, is diagnosed through microscopy, serology, or PCR-based assays. However, these approaches face inherent challenges in sensitivity and specificity. Thus, there is a critical need for supplementary diagnostic assays to augment existing methodologies. The T. cruzi-laminin parasite receptor is a potentially particular biomarker for T. cruzi infection. Studies have demonstrated that employing a SELEX-based approach enables the identification of parasite-specific aptamers capable of detecting the T. cruzi-laminin parasite receptor in infected mice. These RNA ligands interact with parasite targets as viable tools for detecting biomarkers of Chagas disease.
Trypanosoma cruzi-Laminin Parasite Receptor Aptamer Analysis
Creative Biolabs provides a wide range of Trypanosoma cruzi-laminin parasite receptor related products, including anti-Trypanosoma cruzi-laminin parasite receptor aptamer. These products can effectively help to carry out your experiments and thus play an important role in your research.
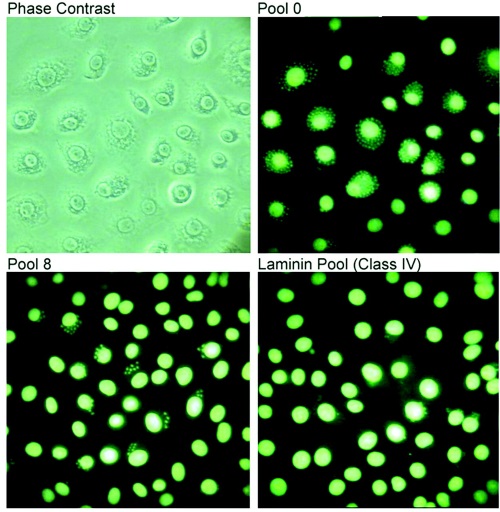 Fig. 2 Selected aptamers inhibit in vitro invasion of LLC-MK2 cells.2,3
Fig. 2 Selected aptamers inhibit in vitro invasion of LLC-MK2 cells.2,3
Henning Ulrich et al. isolated several types of RNA aptamers through eight rounds of SELEX in vitro selection, which have high affinity for parasite receptors such as adhesion protein, fibronectin, thrombospondin, and heparin sulfate on the surface of Trypanosoma cruzi host cells.2,3 In order to verify the inhibitory effect of the selected aptamers on the invasion of Trypanosoma cruzi cells, the invasion of Trypanosoma cruzi on LLC-MK2 cells was tested in vitro. The results showed that the RNA aptamer mixture could not completely prevent cell invasion, which indicates that the invasion of Trypanosoma cruzi on host cells is also affected by other factors unrelated to the ligand.
Creative Biolabs provides extensive products related to the T. cruzi-laminin parasite receptor, including assay kits and aptamers targeting this receptor. Furthermore, we offer customized solutions tailored to the T. cruzi-laminin parasite receptor to meet distinct requirements and specifications.
References
-
Maeda, Fernando Y., Cristian Cortez, and Nobuko Yoshida. "Cell signaling during Trypanosoma cruzi invasion." Frontiers in Immunology 3 (2012): 361.
-
Ulrich, Henning, et al. "In vitro selection of RNA aptamers that bind to cell adhesion receptors of Trypanosoma cruzi and inhibit cell invasion." Journal of Biological Chemistry 277.23 (2002): 20756-20762.
-
Distributed under Open Access license CC BY 4.0, without modification.


 Datasheet
Datasheet Fig.1 Schematic overview of signaling molecules and pathways activated during T. cruzi entry into nonphagocytic mammalian cells.1,3
Fig.1 Schematic overview of signaling molecules and pathways activated during T. cruzi entry into nonphagocytic mammalian cells.1,3
 Fig. 2 Selected aptamers inhibit in vitro invasion of LLC-MK2 cells.2,3
Fig. 2 Selected aptamers inhibit in vitro invasion of LLC-MK2 cells.2,3
