Collagen induced Arthritis (CIA) Modeling & Pharmacodynamics Service
Creative Biolabs offers a variety of well-established models for evaluating the efficacy of drugs targeting arthritis, including both autoimmune and degenerative forms.
Introduction
Arthritis refers to a group of inflammatory joint disorders that cause pain, swelling, stiffness, and reduced mobility. Among the most common forms are osteoarthritis (OA) and rheumatoid arthritis (RA). OA is primarily caused by wear and tear on the joints, leading to cartilage degeneration, while RA is an autoimmune disease in which the immune system attacks healthy joint tissues, causing chronic inflammation. Both conditions lead to significant joint damage, pain, and loss of function, severely affecting quality of life. RA can also cause systemic complications such as cardiovascular disease and osteoporosis. The prevalence of arthritis is increasing globally, with millions of people affected, particularly the elderly and those with a family history of autoimmune diseases. Early diagnosis and effective treatment are crucial to managing the disease and preventing long-term disability. The development of new therapies, including biologics, small molecules, and targeted treatments, requires robust preclinical models that accurately mimic the pathogenesis of arthritis.
Collagen induced Arthritis Model and Applications
The Collagen-Induced Arthritis Model is typically induced by injecting collagen type II (CII) emulsified in Freund’s complete adjuvant into the tail base of rodents, often mice or rats. This triggers an immune response that leads to the development of a chronic, autoimmune inflammatory disease resembling human rheumatoid arthritis. The model exhibits synovial inflammation, cartilage degradation, bone erosion, and systemic effects, particularly in the joints. Arthritis typically develops 10 to 14 days after the initial injection, with peak severity observed around 21 days. One advantage of the CIA model is its reproducibility and ability to mimic human RA in terms of immune cell infiltration, cytokine production, and joint pathology. However, a limitation is that it may not fully replicate the genetic diversity and environmental factors seen in human patients, which may necessitate complementary models for more comprehensive results.
- Simulates: The model simulates autoimmune diseases, specifically rheumatoid arthritis (RA), with inflammation, joint destruction, and immune-mediated damage in multiple joints.
- Evaluates Drugs: It is commonly used to evaluate the efficacy of anti-inflammatory drugs, disease-modifying antirheumatic drugs (DMARDs), biologics targeting specific cytokines, and immunosuppressive agents. It is also used to assess therapies aimed at modulating immune responses and preventing joint damage.
Evaluation Platform
- Animals: Mouse, Rat, Hamster, Rabbit, Cat, Dog, NHPs.
-
Measurements
We offer a variety of measurements for evaluating drug efficacy in the Collagen-Induced Arthritis Model, utilizing advanced technologies, including but not limited to:- General Observations: Joint swelling, paw thickness, body weight changes, and overall health condition of the animals.
- Immunohistochemistry: Analysis of immune cell infiltration (e.g., T-cells, B-cells, macrophages) in inflamed joints and synovial tissue.
- Cytokine Profiling (e.g., ELISA): Quantification of key inflammatory mediators such as TNF-α, IL-1β, IL-6, IL-17, and IL-23 that are involved in the pathogenesis of arthritis via ELISA.
- X-ray Imaging: Assessment of bone erosion and joint destruction.
- Histopathology: Evaluation of joint tissues for inflammation, cartilage degradation, and bone erosion using H&E staining.
- Gene/Protein Expression Profiling: Measuring the expression of inflammatory genes and cytokines through RT-qPCR and Western blot techniques.
Our advantages
- Customizable Experimental Designs: Tailored to your specific research questions, providing flexibility in experimental setup.
- High Reproducibility: Reliable and consistent results across studies, ensuring the validity of data.
- Comprehensive Analytical Platforms: A wide range of advanced technologies for thorough data analysis, from imaging to molecular profiling.
- Expert Consultation: Our scientific team provides support from study design to data interpretation, ensuring the success of your research.
- Validated Models: Well-established and widely recognized models that mirror human disease, offering confidence in your results.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
1. What is the typical timeline for the development of arthritis in this model?
Arthritis usually begins to develop 10-14 days after collagen injection, with peak disease severity observed around day 21.
-
2. Which species are commonly used in the Collagen-Induced Arthritis Model?
Mice are the most used species due to their smaller size and well-established immune response. Rats are also used in some cases for larger joint studies.
-
3. How is joint inflammation measured in this model?
Joint inflammation is typically measured through paw swelling, histopathological analysis, and radiographic imaging to assess joint damage and bone erosion.
-
4. Can this model be used to study chronic arthritis?
Yes, the model can be used to study both acute and chronic phases of arthritis, allowing for long-term assessment of disease progression and therapeutic effects.
-
5. What types of drugs can be tested in this model?
The model is used to test a wide range of drugs, including anti-inflammatory agents, biologics targeting cytokines, DMARDs, and small molecules aimed at modulating the immune response.
Published Data
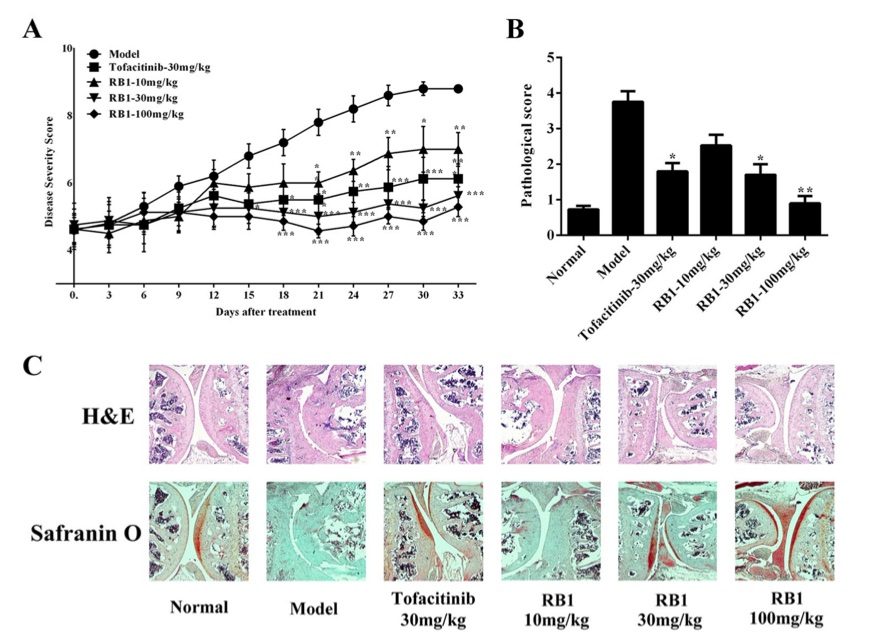 Fig. 1 RB1 is efficacious in treating a collagen-induced arthritis model.1
Fig. 1 RB1 is efficacious in treating a collagen-induced arthritis model.1
The experiment established a collagen-induced arthritis model. Mice treated with RB1 exhibited significantly lower arthritis scores compared to the untreated CIA group (Fig. 1A). The efficacy of RB1 at doses of 10, 30, and 100 mg/kg was found to be comparable to that of Tofacitinib at 30 mg/kg. Representative images of joints and paw swelling showed the therapeutic effect of RB1 in the treated mice. The model group exhibited a mean severity score of 3.8, while the mean histological severity scores in the RB1-treated groups were 0.9, 1.7, and 2.5 for the 100, 30, and 10 mg/kg doses, respectively. The Tofacitinib-treated group had a histological severity score of 1.8 (Fig. 1B). Histological analysis using H&E staining revealed severe synovial inflammation in the model group, characterized by thickening of the synovial lining, infiltration of inflammatory cells, and erosion of both cartilage and subchondral bone. In contrast, RB1-treated mice, particularly at doses of 30 and 100 mg/kg, exhibited significantly reduced inflammation and less cartilage and bone erosion. Cartilage damage was further assessed using safranin O staining, which showed extensive depletion of proteoglycans and loss of chondrocytes in the model group. However, RB1 treatment resulted in a dose-dependent reduction in both inflammation and cartilage degradation (Fig. 1C).
Reference
- Pei, Heying et al. "Discovery of a highly selective JAK3 inhibitor for the treatment of rheumatoid arthritis." Scientific Reports vol. 8,1 5273. 27 Mar. 2018. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1038/s41598-018-23569-y
For Research Use Only.
