Carrageenan induced Paw Edema Modeling & Pharmacodynamics Service
Creative Biolabs offers comprehensive services for evaluating the efficacy of drug candidates in Carrageenan-induced paw edema models, providing both established and customized approaches to ensure robust results.
Introduction
Paw edema is a common experimental model used to study inflammation, primarily involving the induction of swelling in the paw of rodents. This model is employed to investigate acute inflammatory responses, making it relevant for the evaluation of anti-inflammatory drugs and treatments for conditions like rheumatoid arthritis, osteoarthritis, soft tissue injuries, and other inflammatory diseases. The edema is typically induced by injecting an irritant, such as carrageenan, into the paw, causing an inflammatory cascade characterized by vasodilation, increased vascular permeability, and immune cell infiltration. The resulting swelling can be measured and quantified over time, providing a reliable assessment of drug efficacy in reducing inflammation. The paw edema model is widely used due to its simplicity, reproducibility, and the ability to mimic human inflammatory responses, allowing researchers to screen and evaluate a variety of potential anti-inflammatory agents, including NSAIDs, corticosteroids, and novel therapeutic compounds. This model also helps in understanding the underlying molecular pathways of inflammation, offering valuable insights into drug mechanisms of action.
Carrageenan induced Paw Edema Model
The Carrageenan-induced paw edema model involves the subplantar injection of carrageenan into the paw of rodents, leading to a dose-dependent swelling that can be quantified by measuring paw volume or weight. This model is characterized by its simplicity and reproducibility, making it an ideal choice for screening anti-inflammatory agents. Its advantages include rapid induction of edema and ease of measurement. However, it primarily reflects acute inflammation and may not fully capture the chronic inflammatory processes observed in certain human diseases. Its limitations include a lack of involvement of the immune system and the absence of longer-term effects, making it less suitable for evaluating chronic inflammation treatments.
- Simulates: Acute inflammatory response, such as that seen in soft tissue injury, rheumatoid arthritis, and other inflammatory conditions.
- Evaluates Drugs: Anti-inflammatory drugs, analgesics, and corticosteroids, as well as novel compounds aimed at reducing swelling, pain, or inflammation.
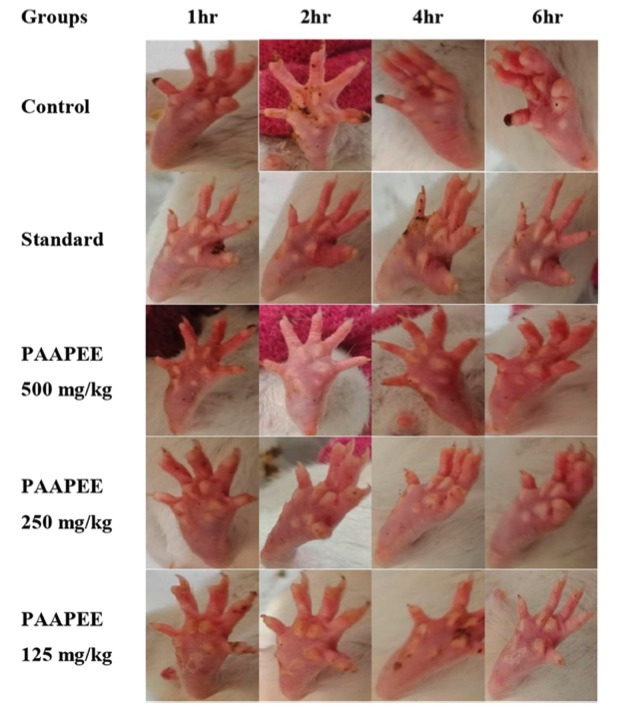 Fig. 1 Macroscopic observation of paws in carrageenan-induced paw edema model.1,3
Fig. 1 Macroscopic observation of paws in carrageenan-induced paw edema model.1,3
Evaluation Platform
- Animals: Mouse, Rat, Hamster, Rabbit, Cat, Dog, NHPs.
-
Measurements
We provide a variety of analytical methods to evaluate the efficacy of drugs in the Carrageenan-induced paw edema model, leveraging advanced technologies, including but not limited to:- General observations: Measurement of paw volume or weight to assess the degree of edema.
- Histopathology: Evaluation of tissue damage, leukocyte infiltration, and inflammatory markers in the affected paw tissues.
- Cytokine profiling (e.g., ELISA):: Detection of inflammatory cytokines such as TNF-α, IL-6, and IL-1β in paw tissue or serum via ELISA.
- Hematology analysis: Examination of changes in blood cell counts because of inflammation.
- Gene/protein expression analysis: RT-qPCR and Western blotting to evaluate the expression of inflammatory mediators at the molecular level.
Our advantages
- Expertise and Experience: With years of experience in preclinical research, our team of scientists is highly skilled in designing and executing animal models tailored to your research objectives.
- Customized Solutions: We offer flexible and customizable models to fit the unique needs of your project, ensuring the most relevant and accurate results for your drug efficacy evaluations.
- State-of-the-Art Technology: Our lab is equipped with cutting-edge technologies and advanced methodologies, including cytokine profiling, histopathology, and gene expression analysis, to deliver comprehensive data for your studies.
- Reliable and Reproducible Results: We pride ourselves on providing high-quality, reproducible results that meet industry standards, helping you advance your research with confidence.
- Comprehensive Support: From experimental design to data interpretation, our scientific team works closely with you to ensure your project runs smoothly and efficiently.
Work with Us
- Summarize the project requirements and fill in the information collection form.
- Sign a CDA from both parties to further communicate information, such as targets.
- Select an animal model, discuss experimental design, and determine assay parameters.
- Project costing and project schedule forecasting.
- We provide a detailed project plan, including the required sample quantities, methods, and protocols.
- Both parties confirm the project details and start the project.
- Confirm the timeline of the project.
- We provide periodic results and information on the animal's condition.
- We will work together to make project adjustments as necessary.
- We provide a comprehensive project report promptly.
- We arrange transportation for the produced samples.
- We provide a discussion of the project results and help to arrange the next steps.
- Data storage and archiving.
FAQs
-
1. What types of drugs can be tested using the Carrageenan-induced paw edema model?
The model is ideal for testing anti-inflammatory agents, analgesics, and other compounds targeting acute inflammation.
-
2. How is the Carrageenan-induced paw edema model performed?
Carrageenan is injected into the paw, and the extent of edema is measured over time to assess the anti-inflammatory effects of the treatment.
-
3. What are the limitations of this model?
While it provides an excellent evaluation of acute inflammation, it may not be suitable for studying chronic inflammatory diseases or immune system involvement.
-
4. How long does it take to see results from this model?
Swelling typically begins within a few hours of injection, with peak edema observed within 3 to 6 hours, making it a rapid and effective model for acute inflammation.
Published Data
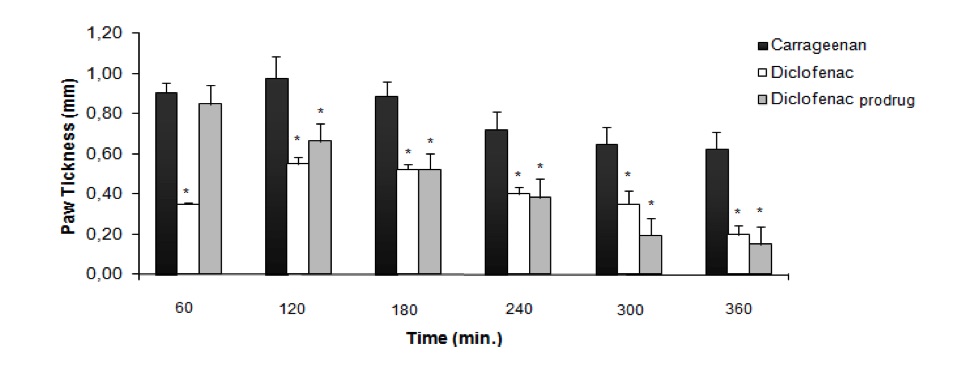 Fig. 2 Anti-inflammatory activity using carrageenan-induced paw edema models in rats.2,3
Fig. 2 Anti-inflammatory activity using carrageenan-induced paw edema models in rats.2,3
This study focuses on the pharmacological evaluation and preliminary pharmacokinetic analysis of a novel diclofenac prodrug designed to eliminate gastric ulceration effects. The anti-inflammatory effects of diclofenac prodrug (1) and diclofenac (2) at a dose of 300 μmol/kg were evaluated in a carrageenan-induced paw edema model in rats, as shown in Figure 2. The results revealed that diclofenac (2) significantly reduced inflammation within 60 minutes of oral administration, whereas the prodrug (1) showed no noticeable activity during this time. However, after 180 minutes, the prodrug (1) exhibited comparable anti-inflammatory effects to diclofenac (2). All observations were statistically significant when compared to the control group, which received an aqueous solution of sodium carboxymethylcellulose (0.5% w/v). Interestingly, at the 300 μmol/kg dosage, the anti-inflammatory activity was found to be equivalent to that observed previously at 100 μmol/kg.
References
- Sarwar, Imtisal et al. "Phytochemical and biological studies of Panicum antidotale aerial parts ethanol extract supported by molecular docking study." Frontiers in pharmacology vol. 14 1243742. 4 Jan. 2024. https://doi.org/10.3389/fphar.2023.1243742
- Santos, Jean Leandro Dos et al. "Pharmacological evaluation and preliminary pharmacokinetics studies of a new diclofenac prodrug without gastric ulceration effect." International journal of molecular sciences vol. 13,11 15305-20. 19 Nov. 2012. https://doi.org/10.3390/ijms131115305
- Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
