Lentiviral Vector Development for Hematopoietic Stem Cell-based Gene Therapy
Based on the outstanding expertise and rich experience, Creative Biolabs offers our clients lentiviral vector design service in the most high-quality and cost-effective way. Particularly, hematopoietic stem cells (HSCs)-based gene therapy development is our featured service which is reliable and economical.
Hematopoietic Stem Cell-based Gene Therapy
HSCs are multipotent, self-renewing progenitor cells that develop from mesodermal hemangioblast cells. HSCs can be found in adult bone marrow, peripheral blood, and umbilical cord blood. Currently, the hematopoietic system has been treated as an ideal target for gene therapy because of the ease with which HSCs can be accessed for ex vivo gene manipulation, effective gene modification, and re-administration as an intravenous infusion. The engrafted HSCs ensure a steady supply of genetically engineered progeny potentially for the recipient's lifetime. Mature cells of different lineages may then reverse pathological conditions such as inherited immune deficiencies, blood and storage disorders, infections, and cancer.
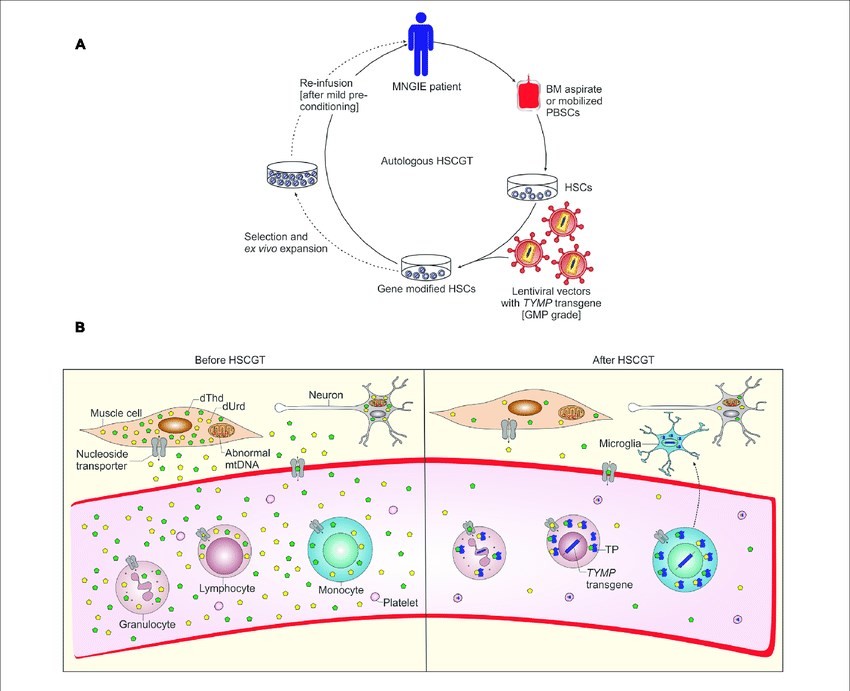 Figure 1. Schematic representation of autologous hematopoietic stem cell-based gene therapy for MNGIE and possible mechanism of biochemical correction by gene-modified HSCs.1
Figure 1. Schematic representation of autologous hematopoietic stem cell-based gene therapy for MNGIE and possible mechanism of biochemical correction by gene-modified HSCs.1
Application of HSCs-based Gene Therapy
Successful HSC gene therapy requires inserting the therapeutic gene into the cellular chromosomal DNA, or editing a selected nucleotide sequence, to ensure the maintenance of gene correction as the cell replicates its genome and transmits it to the progeny, whether during self-renewal or in the output of differentiating cell lineages. Recently, HSC gene therapy is becoming a powerful and versatile strategy to treat a growing number of human diseases. It has been reported that HSC gene therapy is very effective for the malignant hematologic disease but genetic diseases of the blood including immunodeficiencies, disorders of red cell production, or red cell function can also be corrected. Furthermore, HSC gene therapy can also correct some genetic diseases where a secreted enzyme, normally expressed in hematopoietic lineages can be taken up by surrounding affected cells.
Challenges in Lentiviral Vector Engineering for HSC Applications
Although lentiviral vectors are widely used for HSC gene therapy, several technical challenges remain in achieving optimal gene transfer efficiency and clinical safety.
1. Efficient Transduction of Quiescent Stem Cells
HSCs are often maintained in a relatively quiescent state, which can reduce susceptibility to viral transduction. Efficient gene delivery requires optimized viral envelope pseudotyping and transduction conditions.
2. Stable and Controlled Gene Expression
Long-term expression of therapeutic genes is essential for sustained clinical benefit. However, promoter selection and vector architecture can strongly influence expression stability in hematopoietic lineages.
3. Minimizing Genotoxicity
Integration of viral vectors into the host genome carries the risk of insertional mutagenesis. Careful vector design is required to minimize disruption of endogenous genes and oncogenic activation.
4. Maintaining Stem Cell Function
Genetic modification should not impair the self-renewal capacity or differentiation potential of HSCs.
5. Scalability and Regulatory Compliance
For clinical translation, vector production must be scalable and compatible with regulatory standards for gene therapy development.
Advanced Lentiviral Vector Design Platform
Creative Biolabs has developed a comprehensive lentiviral vector engineering platform optimized for hematopoietic stem cell gene therapy. Our solutions address the critical challenges associated with HSC transduction and therapeutic gene delivery.
- Safety-Optimized Vector Architecture
We employ third-generation self-inactivating (SIN) lentiviral vectors that minimize the risk of insertional mutagenesis while maintaining high transduction efficiency. These vectors contain only essential viral elements required for packaging and integration, thereby improving biosafety.
- Promoter Engineering for Hematopoietic Expression
To ensure robust and lineage-appropriate expression of therapeutic genes, we provide a wide selection of promoters commonly used in HSC gene therapy, including:
- EF1α promoter for stable expression across differentiation stages
- PGK promoter for moderate, consistent expression
- UBC promoter for ubiquitous transcriptional activity
- Lineage-specific promoters for targeted gene expression
Promoter choice plays a critical role in determining transcriptional activity and stability of gene expression in stem cells and differentiated progeny.
- Envelope Pseudotyping and Targeting
Our platform supports pseudotyping with a range of viral envelopes to enhance tropism and transduction efficiency in hematopoietic cells. Pseudotyping strategies can significantly improve vector entry and gene transfer efficiency across different cell types.
- Vector Payload Optimization
Our scientists carefully optimize vector payload design, including:
- Therapeutic gene sequences
- Codon optimization
- Regulatory elements
- Reporter genes or selection markers
- Multi-gene expression systems
Comprehensive Service Overview
Creative Biolabs provides a full suite of services covering every stage of lentiviral vector design and development for HSC-based gene therapy programs.
| Service Category | Description |
|---|---|
| Vector Design | Custom design of lentiviral constructs optimized for HSC gene delivery |
| Gene Synthesis | Codon optimization and synthesis of therapeutic gene sequences |
| Plasmid Construction | Generation of high-quality transfer plasmids |
| Lentiviral Production | High-titer lentivirus generation using optimized packaging systems |
| Vector Optimization | Promoter selection, regulatory element engineering, and vector configuration refinement |
| Functional Validation | Assessment of transduction efficiency and gene expression in HSC models |
| Safety Evaluation | Replication-competent lentivirus testing and vector integration analysis |
| Scale-Up Production | Preparation of research-grade or preclinical-grade viral vectors |
Comprehensive Analytical Services & Quality Control
Safety and quality are paramount. We offer a full suite of analytical assays to characterize your vector and ensure it meets the highest standards.
- Identity & Purity: ddPCR/qPCR for titer determination, HPLC/SDS-PAGE for purity analysis.
- Safety Testing: Rigorous testing for Replication-Competent Lentivirus (RCL), sterility, mycoplasma, and endotoxins.
- Potency: Transduction efficiency assays on relevant HSC lines or primary cells, followed by flow cytometry or qPCR to verify functional gene transfer and expression.
- Integration Site Analysis: Utilizing advanced techniques like targeted next-generation sequencing to analyze vector insertion profiles, assess clonality, and evaluate genotoxicity risk.
Deliverables
Clients partnering with Creative Biolabs receive comprehensive deliverables that support both research and translational development.
Typical deliverables include:
- Fully characterized lentiviral transfer plasmids
- High-titer lentiviral vector preparations
- Vector sequence verification data
- Viral titer reports
- Transduction efficiency analysis
- Functional assay results
- Detailed experimental protocols
- Comprehensive project reports
These deliverables ensure that clients can rapidly integrate vector reagents into downstream research workflows.
Our Development Workflow
To ensure efficient project execution and high-quality deliverables, Creative Biolabs follows a structured and transparent development workflow.
-
Step 1: Project Consultation and Experimental Design
Our experts collaborate with clients to understand project goals, target genes, disease models, and desired vector features. Based on these requirements, we develop a tailored vector design strategy.
-
Step 2: Vector Design and Plasmid Construction
We design and construct lentiviral transfer plasmids incorporating optimized promoters, regulatory elements, and therapeutic genes.
-
Step 3: Lentiviral Packaging and Production
Vectors are packaged into replication-defective lentiviral particles using advanced packaging systems to achieve high viral titers.
-
Step 4: Vector Purification and Titration
Produced vectors undergo purification and rigorous titer determination to ensure consistent performance.
-
Step 5: Functional Testing
We evaluate vector performance in relevant cell models to assess transduction efficiency and gene expression.
-
Step 6: Quality Control and Data Delivery
Clients receive comprehensive analytical reports, including vector characterization, QC metrics, and experimental data.
Applications of HSC-Based Lentiviral Gene Therapy
Lentiviral vectors engineered for hematopoietic stem cells are widely used across multiple biomedical research and therapeutic fields.
Genetic Blood Disorders
HSC gene therapy is being developed to correct genetic mutations responsible for diseases such as:
- β-thalassemia
- Sickle cell disease
- Fanconi anemia
Primary Immunodeficiencies
Genetic modification of HSCs enables restoration of immune function in patients with severe immune disorders.
Metabolic and Lysosomal Storage Diseases
Because hematopoietic cells can secrete therapeutic enzymes that are taken up by other tissues, HSC gene therapy can treat systemic metabolic disorders.
Cancer Immunotherapy
Engineered hematopoietic cells can be used to generate immune cells with enhanced tumor-targeting capabilities.
Gene Editing Applications
Lentiviral vectors can deliver gene-editing systems such as CRISPR/Cas9 into hematopoietic stem cells to enable precise genome modification.
Why Choose Creative Biolabs?
Creative Biolabs has established itself as a trusted partner for viral vector engineering and gene therapy development.
Extensive Vector Engineering Expertise
Our team possesses extensive experience in lentiviral vector design for a wide range of gene therapy applications.
End-to-End Service Platform
From vector design to functional validation and scale-up production, we provide a fully integrated development pipeline.
Rigorous Quality Control
Every vector undergoes comprehensive quality testing to ensure safety, consistency, and performance.
Flexible and Customized Solutions
We tailor each project according to client requirements, ensuring optimal outcomes for diverse research programs.
Frequently Asked Questions (FAQ)
Q: Why are lentiviral vectors widely used for hematopoietic stem cell (HSC) gene therapy?
A: Lentiviral vectors are currently one of the most reliable gene delivery systems for HSC-based gene therapy because they can efficiently transduce both dividing and non-dividing cells and integrate stably into the host genome. This allows long-term expression of therapeutic genes in hematopoietic stem cells and their differentiated progeny. Compared with earlier retroviral systems, modern third-generation lentiviral vectors also feature improved biosafety through self-inactivating (SIN) designs that reduce the risk of insertional mutagenesis. As a result, lentiviral vectors have become the platform of choice in many clinical gene therapy programs targeting hematological disorders.
Q: What factors influence lentiviral transduction efficiency in hematopoietic stem cells?
A: Several key factors determine the success of lentiviral gene transfer into HSCs, including vector design, viral titer, envelope pseudotyping, transduction conditions, and cell activation state. For example, the choice of promoter can significantly affect transgene expression stability in hematopoietic lineages, while the use of optimized viral envelopes can enhance vector entry into stem cells. In addition, culture conditions, cytokine stimulation, and multiplicity of infection (MOI) must be carefully optimized to maximize transduction efficiency without compromising stem cell viability or functionality.
Q: How do you minimize safety risks associated with lentiviral vector integration?
A: Safety is a critical consideration in HSC gene therapy development. At Creative Biolabs, we implement multiple strategies to reduce the risks associated with vector integration. These include the use of third-generation self-inactivating lentiviral systems, removal of unnecessary viral sequences, and incorporation of regulatory elements that limit unintended transcriptional activation. In addition, our quality control procedures include replication-competent lentivirus (RCL) testing and vector characterization to ensure biosafety and compliance with gene therapy research standards.
Q: Can lentiviral vectors be customized for specific disease targets or therapeutic genes?
A: Yes. Lentiviral vectors can be extensively customized to meet the requirements of different gene therapy projects. Our scientists can design vectors containing therapeutic genes, reporter genes, or gene-editing components such as CRISPR/Cas systems. We also provide promoter engineering, codon optimization, and regulatory element integration to achieve optimal gene expression profiles in hematopoietic cells. This flexibility allows the vectors to be tailored for a wide range of applications, including correction of genetic blood disorders, immune system engineering, and metabolic disease therapy.
Q: What information should clients provide when initiating an HSC lentiviral vector design project?
A: To ensure efficient project initiation, clients are encouraged to provide key details such as the target gene or sequence of interest, desired promoter or expression system, intended cell type or disease model, and downstream experimental goals. Additional information about vector size constraints, reporter markers, or selection systems may also be helpful. Based on these inputs, our team will design a customized lentiviral vector strategy and propose an optimized development workflow tailored to the client's research objectives.
Working in the field of gene therapy for many years, Creative Biolabs is committed to offering high-quality lentiviral vector design service to promote the development of HSC-based gene therapy. Our professional scientists are happy to help you construct the most appropriate lentiviral vector targeting numerous diseases. For more detailed information, please feel free to contact us or directly send us a quote.
Reference
- Yadak, R.; et al. (2017). Mitochondrial neurogastrointestinal encephalomyopathy caused by thymidine phosphorylase enzyme deficiency: from pathogenesis to emerging therapeutic options. Frontiers in cellular neuroscience. p.31. Distributed under Open Access license CC BY 4.0, without modification.
