Lentiviral Vector Development for ADA-SCID
Adenosine deaminase (ADA) deficiency is caused by a purine metabolic dysfunction, resulting in severe combined immunodeficiency (SCID) and multiple organ damage. Lentiviral vector (LV)-based gene therapy (GT) has recently been used with success in correcting the immune defects. With Ph.D. level scientists in LV development, Creative Biolabs is dedicated to serving every unique need of LV development for ADA-SCID.
The Background of ADA-SCID
ADA-SCID is a severe primary immunodeficiency characterized by impaired T-cell, B-cell, and NK-cell development, and accounts for 15-20% of all cases of SCID. The lack of ADA causes an accumulation of purine metabolites in the plasma, lymphoid tissues, and red blood cells. Suffering from lymphopenia, absent cellular and humoral immunity, patients with ADA-SCID failure to thrive, and suffer recurrent infections. Moreover, hepatic, renal, lung, skeletal and neurologic abnormalities have been observed in some patients, which indicates that the disease should be considered as a systemic metabolic disorder.
Treatment of ADA-SCID
Like other forms of SCID, bone marrow transplantation from a human leukocyte antigen-identical sibling donor is an effective treatment, but transplants from alternative donors are associated with high morbidity and mortality. Enzyme replacement therapy with bovine ADA conjugated to polyethylene glycol provides adequate metabolic detoxification but often with insufficient immune reconstitution, with the risk of developing neutralizing antibodies and autoimmunity.
Recently, the therapeutic efficacy of gene therapy has been investigated in the absence of enzyme replacement therapy. Results of this trial showed that GT with bone marrow CD34+ cells resulted in the correction of both the immune and metabolic defects of ADA-SCID children pretreated with low-intensity conditioning. However, the adverse events occur in one of the SCID-X1 GT trials have raised general concerns on the potential risks of gammaretroviral vectors. Self-inactivating (SIN) LVs based on human immunodeficiency virus (HIV) have an advanced safety profile over non-SIN gammaretroviral vectors and may thus reduce the risk of insertional oncogenesis. Moreover, LVs have been shown to be safer than gammaretroviral vectors in transferring human candidate hematopoietic stem cells and maintaining sustained transgenes expression.
The Evolution of Gene Therapy Vectors: Why Lentiviral Vectors?
Early gene therapy trials for ADA-SCID and the related X-linked SCID (SCID-X1) using γ-retroviral vectors demonstrated proof-of-concept, showing that gene-corrected cells could engraft and restore immunity. However, the success of SCID-X1 trials was shadowed by the development of T-cell leukemia in several patients. This was traced back to a severe adverse event known as insertional oncogenesis, where the strong viral enhancer elements in the vector's long terminal repeats (LTRs) integrated near and activated proto-oncogenes.
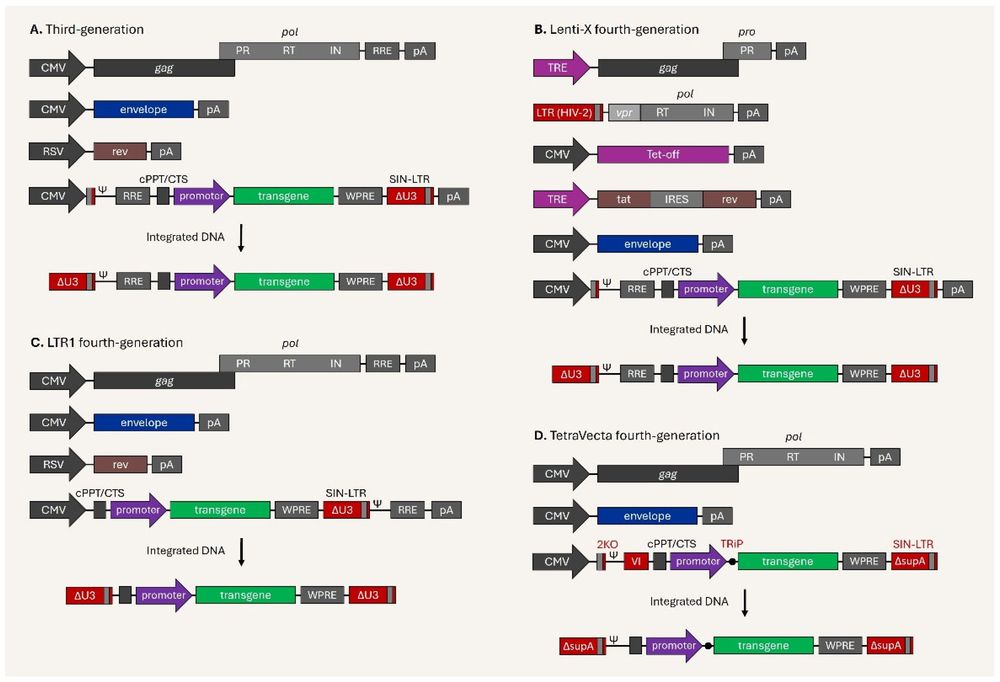 Figure 1. Schematic illustration of third-generation and next-generation lentiviral vectors.1
Figure 1. Schematic illustration of third-generation and next-generation lentiviral vectors.1
This pivotal safety concern drove the development of a new generation of vectors based on lentiviruses, such as HIV-1. Modern lentiviral vectors offer key advantages:
- Enhanced Safety Profile: They are designed as Self-Inactivating (SIN) vectors. A critical deletion in the 3' LTR removes the viral enhancer and promoter sequences upon integration. This "self-inactivation" drastically reduces the risk of activating nearby oncogenes, making them significantly safer than their γ-retroviral predecessors.
- Superior Transduction of Hematopoietic Stem Cells (HSCs): LVs can efficiently transduce quiescent, non-dividing HSCs, which is essential for achieving long-term engraftment and sustained therapeutic effect.
- Stable Gene Expression: LVs mediate stable integration of the therapeutic transgene, ensuring the ADA gene is passed on to all daughter cells of the corrected HSCs, leading to a continuous supply of functional immune cells.
LV Development for ADA-SCID at Creative Biolabs
Creative Biolabs hopes to construct better gene therapy delivery vectors that may correct both the immune system and non-immune system problems. Our scientists have developed an improved LV system carrying a functional ADA gene to correct the genetic defect. This gene encodes for the adenosine deaminase enzyme, which is essential for the proper growth and function of infection-fighting white blood cells called T and B lymphocytes. By collecting an individual's stem cells and modifying them with an LV, the gene-corrected cells can be returned to the patients to help produce normal healthy immune cells.
| Service | Description |
|---|---|
| Vector Design | Construction of ADA expression cassettes with optimized regulatory elements |
| Plasmid Construction | Molecular cloning, sequence verification, and plasmid preparation |
| Lentivirus Production | High-titer lentiviral packaging and purification |
| Vector Optimization | Promoter screening and codon optimization |
| Functional Validation | Transduction assays in relevant cell models |
Why Choose Creative Biolabs
Key Advantages of Partnering with Creative Biolabs
- Deep Scientific Expertise: Our team comprises Ph.D.-level scientists with years of hands-on experience in all aspects of lentiviral vector development, from design to production.
- Proven Technology: Our LV platform is built on robust, validated technology, ensuring high-titer production, efficient gene transfer, and reliable performance.
- Customizable Services: We understand that every research program and preclinical development path is unique. We offer comprehensive, flexible services—from full turn-key solutions to specific a la carte options like vector design, cloning, production, and quality control testing—to meet your precise project needs.
- Commitment to Excellence: We are dedicated to providing the highest quality products and services, supported by rigorous quality control and an unwavering commitment to client success.
- Superior After-Sale Support: Our partnership doesn't end with delivery. Our experts are available to provide ongoing technical support and guidance throughout your research journey.
Result Delivery
Upon completion of the project, clients receive a comprehensive package of deliverables.
| Deliverable | Description |
|---|---|
| Lentiviral plasmid | Sequence-verified vector plasmid |
| Vector map | Annotated plasmid map |
| Lentivirus preparation | High-titer viral particles |
| QC report | Viral titer and safety testing results |
| Experimental protocol | Recommended transduction procedures |
Our Collaborative Development Process
Our lentiviral vector development projects follow a structured workflow designed to ensure scientific rigour, technical reliability, and efficient project execution.
-
Project Consultation and Technical Assessment
Every project begins with a detailed consultation to understand the client's scientific objectives, experimental models, and therapeutic goals. Our experts evaluate key parameters, including target cell type, transgene design, and vector safety considerations, to develop a tailored vector engineering strategy.
-
Vector Architecture Design
Based on the project requirements, our scientists design an optimized lentiviral vector architecture. This stage includes promoter selection, ADA gene cassette design, regulatory element optimization, and safety feature integration to maximize gene expression while maintaining vector stability.
-
Molecular Construction and Sequence Validation
The designed vector is constructed using advanced molecular cloning techniques. All plasmids undergo rigorous sequence verification to ensure accuracy and integrity before proceeding to viral packaging.
-
Lentiviral Packaging and Production
High-titer lentiviral particles are generated using optimized packaging systems. Our production platform enables efficient virus assembly, purification, and concentration, yielding vectors suitable for stem cell transduction experiments.
-
Quality Control and Documentation
Each batch of lentiviral vectors undergoes strict quality control testing, including viral titer measurement, sterility testing, and detection of replication-competent lentivirus. Comprehensive documentation accompanies every delivery.
-
Delivery and Ongoing Support
Clients receive validated vectors along with detailed protocols and experimental recommendations. Our scientific team remains available to provide technical guidance and troubleshooting support throughout the project.
Applications of Our Services
The lentiviral vector engineering services provided by Creative Biolabs support a broad range of research and translational applications in immunology, gene therapy, and stem cell biology.
Hematopoietic Stem Cell Gene Therapy Research
Our lentiviral vectors enable efficient genetic modification of CD34+ hematopoietic stem cells, allowing researchers to investigate strategies for long-term correction of ADA deficiency.
Immune System Reconstitution Studies
By introducing functional ADA genes into stem or immune cells, researchers can explore mechanisms of immune restoration and lymphocyte development.
Genetic Disease Modeling
Lentiviral vectors are valuable tools for generating cellular models of ADA deficiency, helping scientists study disease mechanisms and identify potential therapeutic targets.
Vector Optimization and Technology Development
Our services also support the development and evaluation of next-generation gene delivery technologies, including promoter engineering and transduction optimization strategies.
Customer Reviews

Frequently Asked Questions
Q: What cell types are typically targeted in ADA-SCID gene therapy research?
A: Most ADA-SCID gene therapy strategies focus on CD34+ hematopoietic stem cells, which can be genetically modified ex vivo and reinfused to restore immune function.
Q: Why are lentiviral vectors widely used for stem cell gene therapy?
A: Lentiviral vectors can efficiently transduce both dividing and non-dividing cells and integrate into the host genome, enabling stable and long-term expression of therapeutic genes.
Q: Can lentiviral vectors be customized for specific experimental needs?
A: Yes. Creative Biolabs offers fully customizable vector engineering services, including promoter selection, gene cassette design, and vector optimization based on client requirements.
Q: What viral titers are typically required for hematopoietic stem cell transduction?
A: High-titer lentiviral preparations are generally required for efficient stem cell transduction, often in the range of 108–109 TU/mL, depending on the experimental conditions.
Q: What quality control tests are performed on lentiviral vectors?
A: Our quality control procedures typically include viral titer determination, sterility testing, and replication-competent lentivirus (RCL) detection to ensure vector safety and reliability.
Connect with Us Anytime!
Creative Biolabs is dedicated to supporting researchers with advanced lentiviral vector engineering services for ADA-SCID gene therapy research. By combining expertise in vector design, viral production, and functional validation, we help accelerate the development of innovative gene therapies for inherited immune disorders. Please contact us for more information and a detailed quote.
Reference
- Jargalsaikhan B E, Muto M, Ema M. The era of gene therapy: the advancement of lentiviral vectors and their pseudotyping. Viruses, 2025, 17(8): 1036.https://doi.org/10.3390/v17081036. Distributed under Open Access license CC BY 4.0, without modification.
