Lentiviral Vector Development for Transfusion-dependent β-thalassemia
Introduction of Transfusion-Dependent β-Thalassemia
β-thalassemia is caused by more than 200 mutations in the HBB globin gene, which encodes the beta subunit of the most common form of adult hemoglobin. These mutations either abolish (β0) or reduce (β+) β-globin synthesis, which leads to intracellular hemichrome precipitation, ineffective erythropoiesis, chronic hemolysis, and profound anemia. Transfusion-dependent β-thalassemia is the most severe clinical form, and patients with this disease require long-term red cell transfusions for survival and the prevention of serious complications. Coinheritance of the genetic variant βE with any β0 mutation is responsible for approximately half of all cases of transfusion-dependent β-thalassemia worldwide.
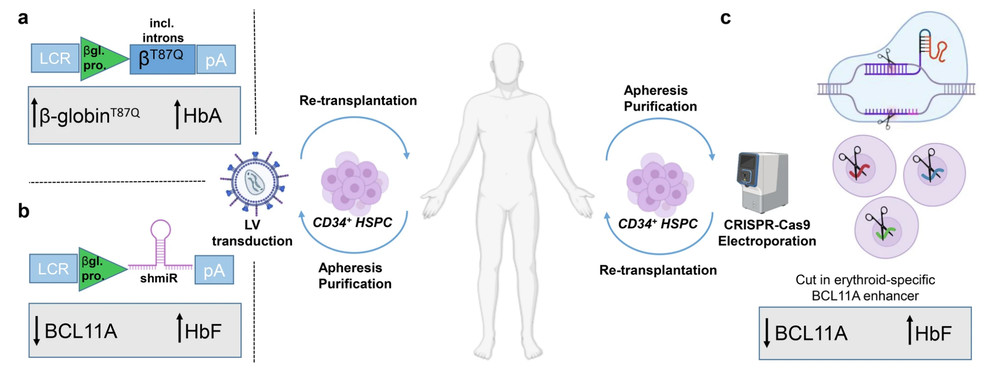 Figure 1. Autologous settings for application of targeted therapies to treat β-thalassemia or sickle cell disease due to β-globin gene mutations.1
Figure 1. Autologous settings for application of targeted therapies to treat β-thalassemia or sickle cell disease due to β-globin gene mutations.1
Lentiviral Gene Therapy for β-Thalassemia
Lentiviral gene therapy aims to restore functional hemoglobin production by introducing a therapeutic β-globin gene into hematopoietic stem cells. The general therapeutic strategy involves the following steps:
- Isolation of CD34+ hematopoietic stem cells from the patient.
- Ex vivo transduction of stem cells using lentiviral vectors carrying a functional β-globin gene.
- Expansion and quality control of modified cells.
- Autologous transplantation back into the patient following conditioning therapy.
- Differentiation of modified stem cells into erythroid cells capable of producing functional hemoglobin.
Because lentiviral vectors can integrate stably into the genome of dividing and non-dividing cells, they enable long-term expression of therapeutic genes in hematopoietic stem cells and their progeny.
Challenges in Developing Lentiviral Vectors for β-Thalassemia
Despite its promise, developing a safe and effective lentiviral vector for TDT presents several formidable technical hurdles. Recognizing these challenges is the first step toward overcoming them.
| Challenge | Impact on Therapy | Description |
|---|---|---|
| High-Level Globin Expression | Insufficient therapeutic hemoglobin | The β-globin gene requires complex regulatory elements for high, erythroid-specific expression. |
| Large Genetic Cargo | Vector packaging limitations | Essential regulatory sequences (e.g., LCR) are large, pushing the limits of lentiviral vector packaging capacity and reducing titer. |
| Efficient HSC Transduction | Poor gene delivery efficiency | Primary human HSCs are notoriously difficult to transduce without inducing differentiation or toxicity. |
| Safety & Genotoxicity | Insertional mutagenesis risk | Vector integration can theoretically activate oncogenes or disrupt tumor suppressor genes. |
| Manufacturing Scalability | Clinical translation difficulty | Producing high-titer, GMP-grade lentivector at a commercial scale is complex and costly. |
Creative Biolabs Lentiviral Vector Development Services
Creative Biolabs offers end-to-end lentiviral vector development services for β-thalassemia research, covering all critical stages of vector engineering and validation.
01 Vector Design and Engineering
Our experts design therapeutic lentiviral vectors specifically optimized for globin gene therapy. Services include:
- Therapeutic HBB gene construct design
- Codon optimization for enhanced gene expression
- Integration of β-globin regulatory elements
- Promoter selection and optimization
- Development of self-inactivating lentiviral vectors
Our design strategies ensure efficient expression of therapeutic globin genes in erythroid cells while maintaining vector safety and stability.
02 Lentiviral Vector Construction
We provide comprehensive vector construction services to generate high-quality lentiviral transfer plasmids.
Key services include:
- Transfer plasmid cloning
- Packaging plasmid system design
- Viral envelope pseudotyping
- Sequence verification and plasmid validation
Our optimized cloning workflows ensure accurate and reliable vector generation.
03 Lentiviral Vector Production
High-titer lentiviral vector production is critical for efficient HSC transduction. Our production services include:
- Small-scale vector production for research studies
- High-titer lentiviral particle generation
- Scalable vector production workflows
- Optimization of production conditions
We utilize advanced lentiviral production systems to achieve high vector titers and reproducible quality.
04 Hematopoietic Stem Cell Transduction Optimization
Efficient gene delivery into hematopoietic stem cells is essential for successful gene therapy.
Creative Biolabs offers:
- CD34+ stem cell isolation and preparation
- MOI optimization for efficient transduction
- Transduction enhancer evaluation
- Optimization of culture conditions
Our protocols are designed to maximize transduction efficiency while preserving stem cell viability and functionality.
05 Functional Validation
We perform comprehensive functional testing to confirm therapeutic gene expression and biological activity.
Validation services include:
- Globin gene expression analysis
- Erythroid differentiation assays
- Hemoglobin production measurement
- Functional characterization of modified cells
These assays help confirm that lentiviral gene transfer restores hemoglobin synthesis.
06 Quality Control and Vector Characterization
Rigorous quality control is essential to ensure vector safety and performance.
Our QC services include:
- Vector titer determination
- Replication-competent lentivirus (RCL) detection
- Integration site analysis
- Vector copy number determination
- Stability testing
Comprehensive QC ensures the reliability and reproducibility of lentiviral vectors used in preclinical studies.
Applications of Our β-Thalassemia Lentiviral Vectors
- Gene Therapy Research: Fundamental studies on HBB gene correction and hemoglobin expression.
- Hematopoietic Stem Cell Engineering: Development of next-generation HSC-based therapies.
- Preclinical Gene Therapy Development: IND-enabling efficacy and toxicology studies in relevant animal models.
- Disease Modeling: Generation of cellular models of β-thalassemia using patient-derived iPSCs or HSCs.
- Gene Editing Support Studies: Lentiviral vectors can also be designed to deliver gene editing components (Cas9, gRNA) to disrupt repressors like BCL11A to reactivate fetal hemoglobin
Advantages of Working with Creative Biolabs
Creative Biolabs has extensive expertise in viral vector engineering and gene therapy development.
Key advantages include:
- Extensive experience in lentiviral vector design and production
- Advanced platforms for hematopoietic stem cell engineering
- Customizable service packages tailored to client needs
- Experienced scientists with deep gene therapy expertise
- Fast turnaround times and reliable technical support
Result Delivery
Upon project completion, Creative Biolabs provides comprehensive deliverables to support further research and development.
| Deliverable | Description |
|---|---|
| Lentiviral plasmid constructs | Validated transfer plasmids |
| High-titer lentiviral particles | Ready-to-use viral vectors |
| Transduction protocols | Optimized experimental procedures |
| Quality control reports | Detailed vector characterization data |
| Experimental data reports | Functional validation results |
These deliverables enable clients to seamlessly integrate our vectors into downstream research programs.
Our Collaborative Process
At Creative Biolabs, we emphasize transparent communication and efficient project management to ensure the success of every gene therapy project. Our collaborative process is designed to streamline lentiviral vector development while maintaining high scientific standards.
01 Project Consultation
We begin with a detailed consultation to understand your research objectives, vector design requirements, and experimental goals. Our experts evaluate project feasibility and recommend optimal strategies for β-thalassemia lentiviral vector development.
02 Customized Project Design
Based on the consultation, we develop a tailored project plan outlining the vector design strategy, production workflow, validation assays, and expected deliverables.
03 Vector Development and Validation
Our scientists perform lentiviral vector engineering, production, and functional validation while maintaining close communication with clients throughout the project.
04 Data Delivery and Technical Support
Upon completion, clients receive comprehensive reports including vector characterization data and experimental results. Our team also provides continued technical support to facilitate downstream research and development.
Customer Reviews

Frequently Asked Questions
Q: What is lentiviral gene therapy for β-thalassemia?
A: It is a treatment where a patient's own hematopoietic stem cells are modified outside the body using a lentiviral vector to add a functional copy of the β-globin gene. These modified cells are then returned to the patient to produce healthy red blood cells, reducing or eliminating the need for blood transfusions .
Q: Why are lentiviral vectors commonly used for globin gene therapy?
A: Lentiviral vectors are uniquely suited because they can efficiently deliver large genetic sequences (like the β-globin gene with its regulatory elements) into non-dividing HSCs and integrate stably into the cell's genome, providing a lifelong source of corrected cells.
Q: What cells are targeted in β-thalassemia gene therapy?
A: The primary target cells are CD34+ hematopoietic stem and progenitor cells (HSPCs) harvested from the patient's bone marrow or mobilized peripheral blood.
Q: How do you ensure the safety of lentiviral vectors?
A: Modern vectors, like the third-generation SIN vectors we use, are designed with multiple safety features. These include separating viral genes across multiple plasmids, deleting key regulatory elements to make the virus self-inactivating, and rigorous quality control testing for replication-competent lentivirus (RCL) and integration site analysis to monitor for clonal outgrowth.
Q: Can your platform support preclinical gene therapy programs?
A: Yes. Our services are designed to generate all the necessary data and materials for successful IND-enabling studies, including efficacy studies in murine models and formal toxicology assessments.
Connect with Us Anytime!
Creative Biolabs provides comprehensive lentiviral vector development services designed to support researchers and biotechnology companies in advancing β-thalassemia gene therapy. From vector design and production to functional validation and quality control, our integrated platform enables efficient development of high-performance lentiviral vectors for gene therapy research. Please contact us for more information and a detailed quote.
Reference
- Morgan M, Schambach A. Successful treatment of transfusion-dependent β-thalassemia: multiple paths to reach potential cure. Signal Transduction and Targeted Therapy, 2025, 10(1): 55.https://doi.org/10.1038/s41392-025-02135-9 Distributed under Open Access license CC BY 4.0, without modification.
