Lentiviral Vector Development for Cerebral Adrenoleukodystrophy
Gene therapy using lentiviral vectors (LVs) has emerged as a promising treatment for patients with cerebral adrenoleukodystrophy (cALD), making LVs an attractive potential alternative to allogeneic transplant for those with cALD for multiple reasons. Equipped with world-leading technology platforms and professional scientific staff, Creative Biolabs is dedicated to offering high-quality service of LV development for cALD to meet our clients' development goal in a time-saving manner.
Introduction of cALD
cALD is characterized by demyelination and neurodegeneration. Disease progression, which leads to loss of neurologic function and death, can be halted only with allogeneic hematopoietic stem-cell transplantation. Adrenoleukodystrophy, an X-linked genetic disease, is caused by a defect in the gene ATP-binding cassette subfamily D member 1 (ABCD1). ABCD1 encodes the peroxisomal ABC half-transporter ALD protein and mutations in this gene resulting in the abnormal breakdown of very-long-chain fatty acids. CALD may develop in approximately 35% of affected boys younger than 12 years old. Learning and behavioral manifestations are often observed when affected patients are between 3-15 years-old, and rapidly progressive loss of neurologic function follows.
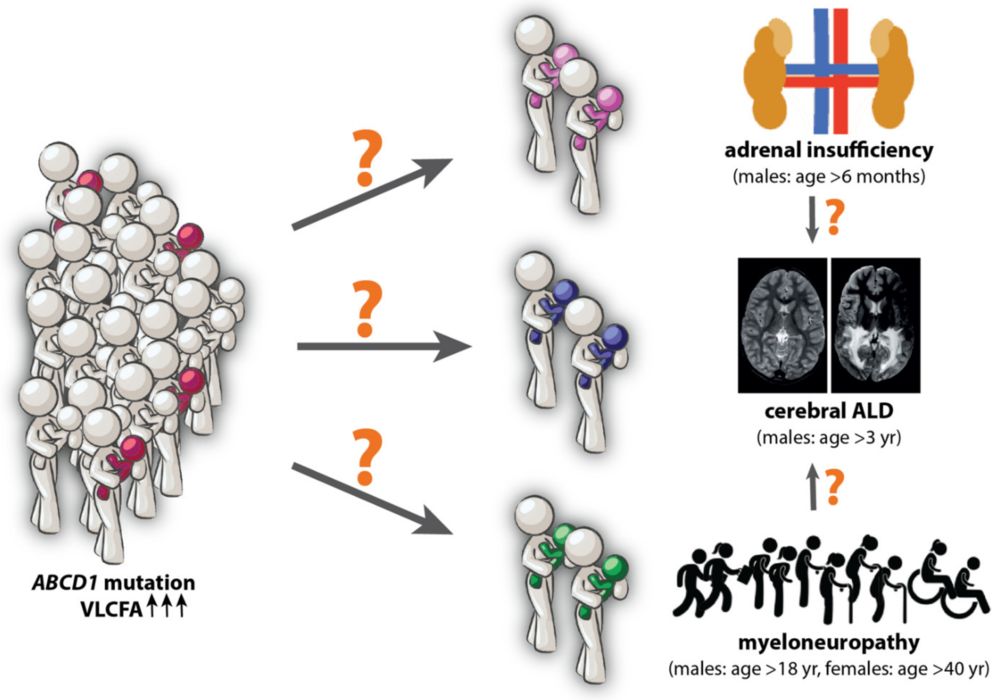 Figure 1. The ALD clinical spectrum. At the molecular level, ALD patients share a genetic defect in the ABCD1 gene and elevated levels of VLCFA.
Figure 1. The ALD clinical spectrum. At the molecular level, ALD patients share a genetic defect in the ABCD1 gene and elevated levels of VLCFA.
Strategic Importance of Lentiviral Gene Therapy for cALD
Lentiviral vectors have become an ideal platform for treating cALD due to their unique biological characteristics.
- Stable Genomic Integration and Long-term Expression: Unlike non-integrating vectors such as adenoviruses, LV can efficiently integrate the functional ABCD1 gene into the genome of host hematopoietic stem cells. This means that as HSCs self-renew and differentiate, the corrected gene can be stably and long-term expressed in all myeloid lineages (including microglia), enabling sustained VLCFA metabolism.
- Efficient Transduction of Hematopoietic Stem Cells: LV can infect both resting and dividing cells, which is crucial for transducing HSCs while maintaining stem cell properties ex vivo. Optimized transduction protocols ensure that a sufficient proportion of stem cells are successfully modified to guarantee clinical efficacy.
Key Challenges in Developing Lentiviral Vectors for cALD
Despite promising prospects, developing safe and effective lentiviral vectors for cALD faces severe challenges. We understand these bottlenecks and address them specifically in our vector design.
| Challenge Area | Impact on Therapy | Our Solutions |
|---|---|---|
| Efficient HSC Transduction | Transduction efficiency directly affects the number of gene-modified HSCs, determining long-term therapeutic efficacy and VLCFA metabolic capacity. | Optimize transduction enhancers and culture conditions, targeting efficient transduction of CD34+ cells, particularly CD90+ HSC subpopulations, to improve gene marking efficiency. |
| Stable, Physiological ABCD1 Expression | Insufficient expression fails to clear VLCFAs; overexpression may cause unknown cytotoxicity or metabolic burden. | Screen myeloid-specific or physiological promoters to achieve optimal ALD protein expression levels in macrophages and microglia. |
| Vector Safety and Insertional Mutagenesis Risk | LV insertion near proto-oncogenes may lead to clonal dominance or even malignant transformation, a core safety concern in gene therapy. | Employ third-generation self-inactivating design, combined with comprehensive integration site analysis to monitor clonal diversity and potential clonal expansion. |
| Large-scale Vector Production and Quality Control | Clinical applications require extremely high-titer, high-purity vectors with strict batch-to-batch consistency. | Establish suspension culture and chromatographic purification processes to ensure high titer and activity during large-scale production, with rigorous process controls. |
| Functional Validation | Detecting gene insertion and protein expression alone is insufficient; must verify whether VLCFA metabolism has been restored. | Establish functional metabolic assay platforms based on patient-derived cells or animal models to directly assess VLCFA level reduction. |
LV Development for cALD at Creative Biolabs
To overcome these challenges, we employ a series of cutting-edge vector engineering technologies to construct next-generation therapeutic lentiviral vectors.
Optimized Vector Backbone
Utilizes third-generation self-inactivating lentiviral systems, deleting enhancer sequences in the U3 region, significantly reducing the risk of activating proto-oncogenes and minimizing interference with internal promoters.
Codon Optimization and Gene Synthesis
Performs codon optimization of the ABCD1 coding sequence, enhancing mRNA stability and translation efficiency without altering the amino acid sequence, achieving higher protein expression.
Advanced Promoter Screening
Offers multiple promoter options, including housekeeping gene promoters (e.g., EF1α), myeloid-specific promoters (e.g., MSCV), and synthetic promoters, to achieve optimal expression strength and lineage specificity for different target cells, particularly HSCs and their myeloid progeny.
Enhanced Packaging Systems
Utilizes optimized versions of REV and GAG/POL helper plasmids, combined with efficient transient transfection systems, to significantly improve viral particle packaging efficiency and infectious titers.
Safety Insulators
Safety Insulators: Incorporates chromatin insulator elements within the vector LTRs or flanking the promoter to further shield internal enhancers from neighbouring genomic regions, providing dual safety assurance.
Workflow of Lentiviral Vector Development for cALD
We follow a rigorous, efficient standardized process to ensure smooth project progression.
-
Step 1: Therapeutic Gene Design
- Analyze ABCD1 gene sequence, perform codon optimization.
- Determine optimal promoter and regulatory elements.
- Finalize design and synthesis of the expression cassette. -
Step 2: Lentiviral Vector Construction and Validation
- Clone optimized expression cassette into SIN lentiviral backbone.
- Perform sequencing validation and restriction enzyme analysis.
- Package virus at small scale, validate expression in cell lines. -
Step 3: Virus Packaging and Purification
- Large-scale transfection of HEK293T cells.
- Harvest supernatant, perform nuclease treatment and depth filtration.
- Purify and concentrate via chromatography and tangential flow filtration. -
Step 4: HSC Transduction and Evaluation
- Isolate CD34+ cells from healthy donors or patients.
- Transduce with lentivirus using optimized protocols.
- Assess transduction efficiency (% positive) and vector copy number by flow cytometry and qPCR. -
Step 5: Functional and Safety Validation
- Evaluate VLCFA metabolism restoration in in vitro differentiation models.
- Perform integration site analysis to assess clonal lineage.
- Complete all necessary QC assays (RCL, sterility, etc.). -
Step 6: Data Delivery and IND Filing Support
- Provide comprehensive experimental reports and data packages.
- Assist in preparing relevant materials required for IND submissions.
Quality Control and Analytical Assays
Rigorous quality control is essential for ensuring the performance and safety of lentiviral vectors.
Our analytical capabilities include:
- Viral titer determination
- Quantitative PCR (qPCR) analysis
- Vector copy number determination
- Integration site analysis
- Replication-competent lentivirus (RCL) testing
- Transgene expression analysis
- Sterility and endotoxin testing
- Functional metabolic assays
These analytical services provide critical data supporting the development of gene therapy vectors.
Applications of Lentiviral Vector Development for cALD
Our lentiviral vector development services support a wide range of research and translational applications, including:
- Gene therapy development for X-linked adrenoleukodystrophy - Design and optimization of therapeutic vectors targeting ABCD1 deficiency.
- Hematopoietic stem cell gene therapy research - Evaluation of lentiviral vector performance in CD34+ cells.
- Preclinical gene therapy studies - Development of vectors for in vitro and in vivo evaluation.
- Neurological disease modeling - Generation of engineered cell models for studying metabolic and neurodegenerative diseases.
- Drug discovery research - Use of lentiviral vectors to investigate metabolic pathways and therapeutic targets.
Key Advantages of Our Service
- Keeping stable and long-term protein expression
- Large-scale production of LVs with features, such as high transduction rates, infection of non-dividing cells and not to expected to elicit immunogenic responses.
- Powered by a proven, first-in-class technology
- Fast turnover time
Customer Reviews

Frequently Asked Questions
Q: What is the specific role of the ABCD1 gene in cALD therapy?
A: The ABCD1 gene encodes a protein that transports VLCFAs into peroxisomes for breakdown. In cALD, mutations in this gene lead to accumulation of VLCFAs (mainly C26:0) in cerebral white matter, triggering demyelination and neuroinflammation. Gene therapy introduces a functional ABCD1 gene, restoring protein function, thereby clearing VLCFAs and halting disease progression.
Q: Why use lentiviral vectors instead of other viral vectors (like AAV) for HSC gene therapy in cALD?
A: Unlike AAV, which primarily persists as episomes, lentiviral vectors stably integrate into the host genome. Because HSCs must continuously divide and self-renew, only integrating LV can ensure that as cells divide, the therapeutic gene is passed to all progeny cells (such as macrophages and microglia), providing sustained therapeutic effect throughout the patient's life.
Q: What vector copy number is required for effective HSC transduction?
A: Clinical data suggest that detectable ALD protein expression in approximately 20% of peripheral blood cells is sufficient to halt disease progression. This typically corresponds to 0.5-2 VCN per cell. However, higher VCN correlates with better clinical outcomes (slower lesion progression), so we aim to optimize transduction protocols to achieve a safe and effective VCN range (typically 0.5-3).
Q: What vector titers are required for stem cell transduction?
A: High-titer viral preparations are typically required to achieve efficient transduction of CD34 stem cells, although the exact requirements vary depending on the experimental system.
Connect with Us Anytime!
Enriched experience, profound expertise and state-of-the-art instruments, all of these can ensure that Creative Biolabs offers high-quality services for LV development for cALD. If you are interested in the development of LV for the research of cALD, please contact us for more information and a detailed quote.
Reference
- Honey M I J, Jaspers Y R J, Engelen M, et al. Molecular biomarkers for adrenoleukodystrophy: an unmet need. Cells, 2021, 10(12): 3427. https://doi.org/10.3390/cells10123427 Distributed under Open Access license CC BY 4.0, without modification.
