Lentiviral Vector Development for Wiskott-Aldrich Syndrome
Wiskott-Aldrich syndrome (WAS) is a life-threatening immunodeficiency caused by mutations within the WAS gene. Lentiviral vector (LV)-based gene therapy of WAS is a promising alternative. With Ph.D. level scientists and extensive experience in LV development, Creative Biolabs is dedicated to serving every unique need of our clients in LV development for WAS by providing customized services.
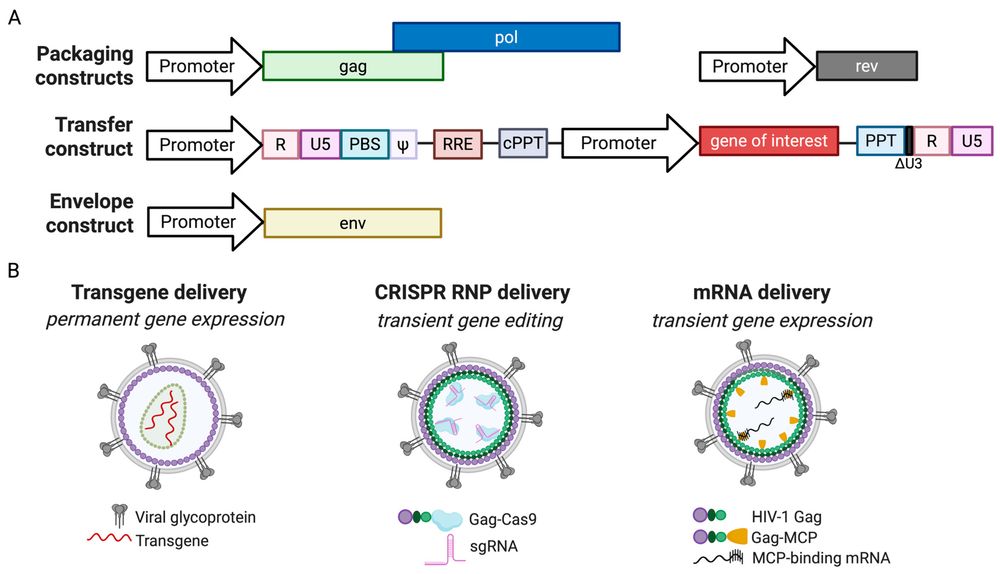 Figure 1. Lentiviral vector system and gene delivery strategies.1
Figure 1. Lentiviral vector system and gene delivery strategies.1
The Background of WAS
WAS is a rare X-linked primary immunodeficiency that results from the loss of WAS protein (WASp). WASp involves in signal transduction pathways that activate the actin cytoskeleton downstream of multiple cell surface receptors, including the B and T cell antigen receptors. And WAS is characterized by micro-thrombocytopenia, recurrent infections, eczema. This complex disease is also associated with a high incidence of autoimmunity and lymphoid malignancies. More and more attentions are paid to WAS not only because it highlights the rich cellular-biology and systems-biology but also because it is a candidate for hematopoietic stem cell gene therapy indication.
Gene Therapy of WAS
Although the phenotype of WAS can be alleviated by hematopoietic stem cell transplantation (HSCT), the success rate of this therapy is variable, which depends on the patient's age, donor compatibility, conditioning regimen, and the extent of reconstitution. Since the phenotype of WAS deficiency impacts only hematopoietic cells, gene therapy is a possible alternative. The gene therapy of WAS represents a new challenge compared to other immune deficiencies. WASp can be stably integrated into the chromatin of autologous hematopoietic stem cells (HSCs) using viral-based gene delivery. The use of self-inactivating LVs for gene transfer is one critical improvement, combining a safer integration profile with the ability to select internal promoters that optimize transgene expression and safety.
Why Lentiviral Vectors for WAS?
Lentiviral vectors are the vehicle of choice for WAS gene therapy due to several critical biological advantages:
Stable Integration
LVs integrate the functional WAS gene into the host genome, ensuring long-term expression across all hematopoietic lineages (T cells, B cells, NK cells, and platelets).
HSC Transduction Efficiency
Unlike AAV, LVs efficiently transduce non-dividing hematopoietic stem cells (CD34+), the primary target for a durable cure.
Safety Profile
Our use of third-generation Self-Inactivating (SIN) designs eliminates the risk of replication-competent lentivirus (RCL) and minimizes the potential for insertional oncogenesis.
Large Payload Capacity
Ample space to include the full-length WAS cDNA along with complex endogenous regulatory elements.
Our Value Proposition: Your Partner in WAS Gene Therapy
- Targeted Expertise: Optimized vector design for efficient and stable WAS gene expression in hematopoietic stem/progenitor cells (HSPCs).
- End-to-End Solutions: Integrated services covering the entire development chain, from vector design to clinical-grade production.
- Quality & Compliance: Stringent quality control and the capability to produce materials under guidelines to support regulatory submissions.
- Proven Track Record: Experienced teams dedicated to overcoming the unique challenges of rare disease gene therapy development.
LV Development for WAS at Creative Biolabs
Scientists at Creative Biolabs have developed an LV that combines a chromatin insulator upstream of a viral promoter to drive WASp expression. Used as a gene therapeutic, this efficient vector resulted in stable WASp+ cells in all hematopoietic lineages and rescue of T and B cell defects with a low number of viral integrations and without insertional mutagenesis. Our results demonstrate that this promoter safely and efficiently reconstitutes WASp expression and can be potentially used for future WAS therapy.
| Service Module | Key Offerings & Technical Highlights | Deliverables & Outcomes |
|---|---|---|
| Vector Design & Construction |
Promoter Optimization: Screening of proprietary and ubiquitous promoters (e.g., MND, EF1α) for optimal WASp expression in HSPCs. Transgene Optimization: Codon optimization of the WAS cDNA to enhance translation efficiency; removal of inhibitory sequences (e.g., CpG islands). Safety by Design: Utilization of 3rd generation self-inactivating (SIN) vector systems to minimize insertional mutagenesis risk. |
Sequence-verified, high-purity transfer plasmids and packaging plasmids; detailed design and optimization report. |
| Virus Packaging & Production |
Flexible Production Systems: Adherent HEK293T transient transfection for early R&D; suspension-adapted stable producer cell lines for scaled-up manufacturing. Scalability: Linear scalability from research-grade (milligram) to clinical-grade (gram) lots. High Titer Assurance: Optimized transfection and harvest protocols to achieve high-titer viral stocks (e.g., >1e8 TU/mL). |
High-titer, high-potency lentiviral vector pre-purification bulk, available at various scales. |
| Purification & Process Development |
Multi-Step Purification Strategy: Employing TFF (Tangential Flow Filtration) for concentration, followed by ion-exchange (IEX) and affinity chromatography for efficient removal of empty capsids, host cell proteins (HCP), and host cell DNA (HCD). Formulation Optimization: Development of customized formulations to ensure vector stability during storage and administration. |
Highly purified, concentrated LVV with defined residual impurity profiles; optimized formulation buffer. |
| Quality Control & Analytics |
Comprehensive Release Testing: • Identity & Purity: qPCR (titer), ddPCR, SDS-PAGE/CE-SDS. • Safety: Replication-competent lentivirus (RCL) assay, sterility, mycoplasma, endotoxin. • Impurities: Residual HCP, HCD. Potency Assay Development: Functional titer determination via transduction of WAS patient-derived cells (e.g., Jurkat T-cells or CD34+ cells) and measurement of WASp protein expression (by flow cytometry/Western blot) and functional correction (e.g., F-actin polymerization assay). |
Certificate of Analysis (CoA) with full release testing data; validated potency assay methods; comprehensive stability study reports. |
Tailored Functional Validation for WAS
We understand that successful gene therapy relies on proof-of-concept. Our services extend beyond vector production to include critical functional validation studies to support your preclinical development.
Ex Vivo Functional Studies
We can perform transduction optimization and efficacy testing using your provided or commercially sourced WAS patient CD34+ cells. Our assays are designed to confirm:
- Efficient Gene Transfer: Transduction efficiency in HSPCs measured by flow cytometry.
- Protein Restoration: Quantification of WASp protein expression by intracellular flow cytometry and Western blot.
- Functional Correction: Assessment of downstream T-cell function, including proliferation capacity and IL-2 production upon stimulation, or correction of cytoskeletal defects (e.g., F-actin content in platelets or T-cells).
In Vivo Proof-of-Concept Support
We can coordinate with your team or our trusted partners to design mouse model studies (e.g., using NSG mice) to demonstrate the long-term engraftment, multilineage reconstitution, and phenotypic correction of gene-modified HSPCs in vivo.
Why Choose Our WAS-Targeted Platform?
01. Deep Disease Biology Insight
Our team has extensive experience in rare primary immunodeficiencies. We understand the critical role of precise WAS gene regulation and the importance of achieving therapeutic expression levels without toxicity. Our vector designs are guided by this biological understanding.
02. A Technology Platform Built for Scalability & Quality
- Advanced Systems: We utilize state-of-the-art production systems, including serum-free suspension culture for robust and reproducible manufacturing.
- Analytical Excellence: Our QC suite is equipped with cutting-edge technologies to perform all required assays, including a highly sensitive RCL detection method.
Our Collaborative Partnership Model
We believe in transparent and efficient collaboration. Our process is designed to keep you informed and in control.
- Inquiry & Consultation: Share your project goals, target product profile, and specific requirements.
- Proposal & Design: Our scientists will work with you to design an optimal vector and propose a tailored service plan with clear timelines and deliverables.
- Agreement & Initiation: Formalize the partnership and initiate the project with a dedicated project manager assigned.
- Execution & Reporting: Regular progress reports and open communication channels ensure complete transparency.
- Delivery & Support: Receive your final deliverables, including full documentation and post-delivery technical support.
Frequently Asked Questions
Q: What makes the third-generation SIN lentiviral system safer for WAS gene therapy?
A: The third-generation Self-Inactivating (SIN) system features a deletion in the U3 region of the 3' Long Terminal Repeat (LTR). During reverse transcription, this deletion is transferred to the 5' LTR, effectively abolishing the transcriptional activity of the viral promoter. This significantly reduces the risk of insertional mutagenesis—the activation of nearby oncogenes—which is a primary safety concern in hematopoietic stem cell (HSC) gene therapy.
Q: Why is the choice of promoter critical for Wiskott-Aldrich Syndrome?
A: Over-expression of the WASP protein can be as detrimental as its deficiency, potentially leading to cellular toxicity or abnormal immune signaling. We recommend using endogenous human WAS promoters (e.g., the 1.6 kb fragment) or specific hybrid promoters that mimic natural expression patterns. This ensures that WASP is expressed at physiological levels and specifically within the hematopoietic lineage, enhancing both safety and efficacy.
Q: How do you ensure high transduction efficiency in CD34+ Hematopoietic Stem Cells?
A: CD34+ cells are notoriously difficult to transduce. We optimize the process through:
- Vector Pseudo-typing: Typically using VSV-G for broad tropism.
- Transduction Enhancers: Utilizing proprietary or clinically-validated enhancers (like Vectofusin-1 or Retronectin) to overcome electrostatic repulsion between the virus and the cell membrane.
- Optimized MOI: Conducting a matrix of Multiplicity of Infection (MOI) studies to find the "sweet spot" where transduction is maximized without compromising cell viability.
Q: Can you provide animal models for WAS efficacy studies?
A: Yes. We offer validated WASP-knockout (WASP-/-) mouse models. These models replicate key human symptoms, such as microthrombocytopenia and defective T-cell activation. We can perform longitudinal studies to measure platelet count recovery, T-cell cytokine production (e.g., IL-2, IFN-γ), and the restoration of secondary lymphoid organ architecture.
Q: How do you handle Replication-Competent Lentivirus (RCL) testing?
A: Safety is our top priority. We perform RCL testing using validated qPCR-based assays to detect the VSV-G envelope gene or the gag-pol sequence in the final product. For projects heading toward clinical trials, we also offer cell-based amplification assays to ensure the absolute absence of any replicative particles.
Connect with Us Anytime!
With years of experience in safety design and high-titer production of LV, Creative Biolabs has gained significant knowledge in LV development for WAS. We are more than happy to share our experience and help our customers with this important step in LV-based gene therapy for WAS. Please contact us for more information and a detailed quote.
Reference
- Arduini A, Katiyar H, Liang C. Progress in pseudotyping lentiviral vectors towards cell-specific gene delivery in vivo. Viruses, 2025, 17(6): 802. https://doi.org/10.3390/v17060802 Distributed under Open Access license CC BY 4.0, without modification.
