Lentiviral Vector Development for Fanconi Anemia
Through continuous testing and long-term research, Creative Biolabs' experienced team has now launched a lentiviral vector (LV) development platform to offer various LV services for gene therapy for Fanconi anemia (FA). These services not only make breakthroughs in the biotechnology industry, but also complement our platform to meet more customers' requirements. So, no matter what difficulties you have encountered in the relevant research work, you can contact us to get your customized service.
Introduction of Fanconi Anemia
FA is a genetic disease which caused by mechanism defection in repairing damaged DNA. Mutations in at least 15 genes will cause FA, and the proteins encoded by these genes are involved in a cellular process called FA pathway associated with abnormal DNA repair. This DNA repair process is mediated by a combination of eight FA-related proteins to form FA core complex to activate both FANCD2 and FANCI proteins, and therefore allows DNA repair proteins enter the interstrand cross-link (ICL) region to eliminate ICL and continue DNA replication. Topically, 80% to 90% of FA diseases are caused by mutations in three genes, FANCA, FANCC, and FANCG. The proteins encoded by these genes instruct the formation of FA core complexes. When they are unable to function normally, the FA pathway is blocked, and eventually leads to the accumulation of ICL, causing various diseases. Patients may have symptoms of bone marrow failure, physical abnormalities, organ defects, and increased risk of cancer. The most common symptom is bone marrow failure. In general, 90% of bone marrow function is affected, resulting in aplastic anemia with reduced blood cell production.
Gene Therapy for Fanconi Anemia
At present, the treatment for patients with FA is mainly to supplement the missing blood cells of patients by using drugs or blood transfusions. It is feasible to cure FA by transplanting healthy human bone marrow, but this method has relatively high risk and is not suitable for everyone. Bone marrow transplantation itself is also a very dangerous operation.
Therefore, the use of gene therapy to cure FA is currently the best solution. The patient's bone marrow stem cells are corrected by using the modified virus to transfer the normal gene to the FA patient without causing infection. Firstly, patients receiving treatment will take some special growth factors to transfer blood-forming stem cells from the bone marrow to the blood. Adults can also use drugs that increase the production of stem cells in the peripheral blood. Secondly, after collecting enough stem cells, the cells are treated with a lentiviral vector carrying a normal gene in the laboratory to correct the loss-of-function genes. Finally, these repaired cells are periodically introduced into the patient's body to cure these hereditary diseases.
Why Lentiviral Vectors Are Ideal for FA Therapy
Lentiviral vectors have become the preferred delivery system for FA gene therapy because they possess several critical advantages over other viral platforms.
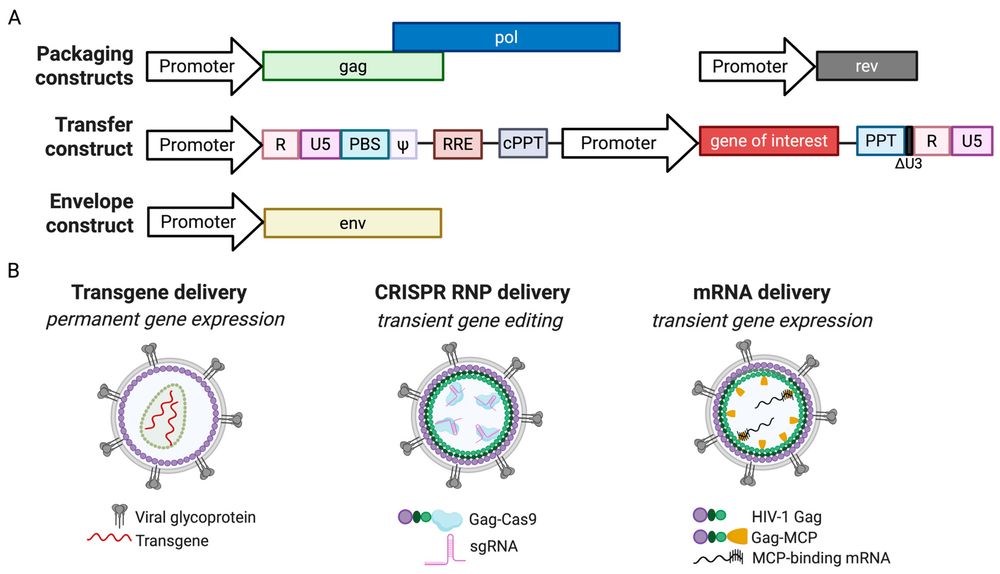 Figure 1. Lentiviral vector system and gene delivery strategies.1
Figure 1. Lentiviral vector system and gene delivery strategies.1
- Stable Genomic Integration
Lentiviral vectors integrate their genetic payload into the host genome, enabling long-term expression of therapeutic genes. This is essential for FA therapy because hematopoietic stem cells must maintain gene expression throughout lifelong hematopoiesis.
- Efficient Transduction of Hematopoietic Stem Cells
Unlike many viral vectors, lentiviruses efficiently transduce quiescent CD34+ hematopoietic stem and progenitor cells, which are the key targets for FA gene correction.
- Reduced Genotoxicity in Modern Vector Designs
Third-generation self-inactivating (SIN) lentiviral vectors incorporate safety modifications such as:
- Deletion of U3 promoter elements
- Internal cellular promoters
- Insulator elements
These modifications significantly reduce the risk of insertional mutagenesis.
Key Challenges in Fanconi Anemia Gene Therapy Development
Despite the promise of lentiviral gene therapy, FA presents unique technical and biological challenges that must be addressed during vector development.
Hypersensitivity of FA Cells to Culture Conditions
FA hematopoietic stem cells are highly sensitive to:
- Oxidative stress
- Cytokine stimulation
- DNA damage during ex vivo culture
Prolonged culture can lead to rapid loss of stem cell function, reducing therapeutic efficacy.
Low Stem Cell Numbers
Patients with FA often have severely reduced hematopoietic stem cell pools, limiting the number of cells available for genetic modification.
Transduction Efficiency Limitations
Achieving efficient gene transfer into fragile FA stem cells requires highly optimized vector design and transduction conditions.
Safety Concerns
Although lentiviral vectors are safer than earlier retroviral vectors, insertional mutagenesis and clonal dominance remain theoretical risks, necessitating careful vector design and integration analysis.
Regulatory Complexity
Gene therapy programs targeting rare diseases must comply with strict regulatory requirements, including vector characterization, safety testing, and manufacturing standards.
Our Solutions
At Creative Biolabs, we have developed advanced strategies to address the specific challenges associated with FA gene therapy development.
- Optimized Vector Design
Our scientists engineer self-inactivating lentiviral vectors with:
- Reduced enhancer activity
- Optimized internal promoters
- FA gene codon optimization
These improvements ensure robust gene expression with enhanced safety profiles.
- Low-Stress Transduction Protocols
We have developed specialized transduction protocols tailored for fragile FA stem cells, minimizing oxidative stress and preserving stemness.
Key strategies include:
- Shortened culture times
- Optimized cytokine cocktails
- Hypoxic culture environments
- High-Titer Lentiviral Production
Creative Biolabs provides ultra-high-titer lentiviral vector production, enabling efficient gene transfer even when limited patient cells are available.
- Comprehensive Safety Evaluation
Our platform includes advanced assays for:
- Vector copy number analysis
- Integration site profiling
- Replication-competent lentivirus (RCL) testing
These assessments ensure maximum safety for preclinical and clinical development programs.
Creative Biolabs Lentiviral Vector Development Platform
Creative Biolabs has established a state-of-the-art lentiviral vector engineering platform supporting gene therapy programs for hematological disorders.
Key platform capabilities include:
Advanced Vector Engineering
- Third-generation self-inactivating vectors
- Codon-optimized FA gene constructs
- Tissue-specific promoter selection
- Safety insulator elements
High-Titer Lentiviral Production
Our optimized packaging systems enable:
- High viral titers
- Stable vector integrity
- Scalable production
Vector Characterization
We perform comprehensive characterization including:
- Vector genome integrity
- Infectious titer quantification
- Transgene expression validation
- Integration site analysis
Workflow of Our FA Lentiviral Vector Development Service
Our streamlined workflow ensures efficient and reliable vector development.
-
Step 1: Project Consultation
Our experts evaluate the client's research goals and design customized vector strategies.
-
Step 2: Vector Construction
Therapeutic gene constructs are designed and cloned into optimized lentiviral backbones.
-
Step 3: Lentiviral Packaging
Vectors are packaged using high-efficiency third-generation systems to achieve high viral titers.
-
Step 4: Transduction Optimization
We test transduction efficiency in hematopoietic stem cells or relevant FA models.
-
Step 5: Functional Validation
Restoration of DNA repair activity and cell survival is evaluated.
-
Step 6: Quality Control and Data Delivery
All vectors undergo rigorous quality control testing before delivery to clients.
Trusted by Global Innovators

Frequently Asked Questions
Q: Why haven't previous FA gene therapy trials worked, and how does your approach differ?
A: Early trials were hampered by inefficient HSC transduction with γ-retroviral vectors and the extreme fragility of FA cells during long ex vivo culture. Our approach leverages the fundamental advantage of LVs in transducing quiescent HSCs and incorporates optimized, short-duration transduction protocols in hypoxic conditions with antioxidants. This minimizes cellular stress and preserves the engraftment potential of these delicate cells, aligning with the methodologies now validated in successful clinical trials.
Q: How do you demonstrate that your vector has functionally corrected the FA defect?
A: We use a multi-tiered approach. The gold standard is the MMC sensitivity assay, where corrected cells should survive and proliferate at MMC concentrations that are lethal to uncorrected cells. For HSCs, we perform colony-forming unit (CFU) assays in MMC-containing media; a significant increase in colony number and size indicates functional correction of hematopoietic progenitors. We can also perform more sophisticated analyses like cell cycle analysis, as FA cells typically arrest in the G2/M phase under DNA damage, a defect that is resolved upon correction.
Q: What FA subtypes can you create vectors for?
A: While FANCA represents the majority of cases, our platform is flexible. We can create lentiviral vectors for any FA subtype, including FANCC, FANCG, and others, by simply cloning the relevant human cDNA into our optimized vector backbones.
Q: What FA genes can your lentiviral vectors deliver?
A: We support lentiviral vector development for multiple FA genes including FANCA, FANCC, FANCG, FANCD2, and other FA pathway components.
The use of lentiviral vectors in FA gene therapy will significantly minimize the potential risks. In the existing cure examples, the patient receiving the treatment is not only in a stable improvement, but also presents no sign of any viral gene insertion. Creative Biolabs is committed to providing you with a lentiviral vector construction service that allows you to get more efficient transfection, and more stable lentiviral vectors. If you have any questions or have any difficulties, you can contact us by email or send us an inquiry to find a complete solution.
Reference
- Arduini A, Katiyar H, Liang C. Progress in pseudotyping lentiviral vectors towards cell-specific gene delivery in vivo. Viruses, 2025, 17(6): 802. https://doi.org/10.3390/v17060802 Distributed under Open Access license CC BY 4.0, without modification.
