Lentiviral Vector Development for X-SCID
To address the efficacy and safety of gene therapy for X-linked severe combined immunodeficiency (X-SCID), Creative Biolabs has developed an LV carrying a codon-optimized human IL2RG cDNA, driven by a short promoter. This vector can be potentially used for gene therapy for X-SCID. We are fully competent and dedicated to serving as your one-stop-shop for LV development for X-SCID.
The Background of X-SCID
X-SCID is caused by loss-of-function mutations in the γc gene (IL2RG), which encodes the common γ-chain (γc) subunit for the IL2, IL4, IL7, IL9, IL15, and IL21 receptors and is required for signal transduction with these cytokines. Human X-SCID is characterized by lack of T, natural killer (NK) cells, and nonfunctional B cells and is fatal early in life from progressive infections if left untreated.
Gene Therapy of X-SCID
Allogeneic stem cell transplantation is the current standard of care for X-SCID and can be curative, particularly when a matched sibling donor is available. However, most patients lack a matched sibling donor. Most experts agree that better treatment is desirable for X-SCID patients who lack a matched sibling donor. Gene therapy is one of several approaches and is being developed as an alternative to haploidentical allogeneic transplantation. Transplantation of hematopoietic stem/progenitor cells (HSPCs) genetically corrected with early murine leukemia retrovirus (MLV)-derived gammaretroviral vectors have shown restoration of T cell immunity in patients, but it results in vector-induced insertional oncogenesis.
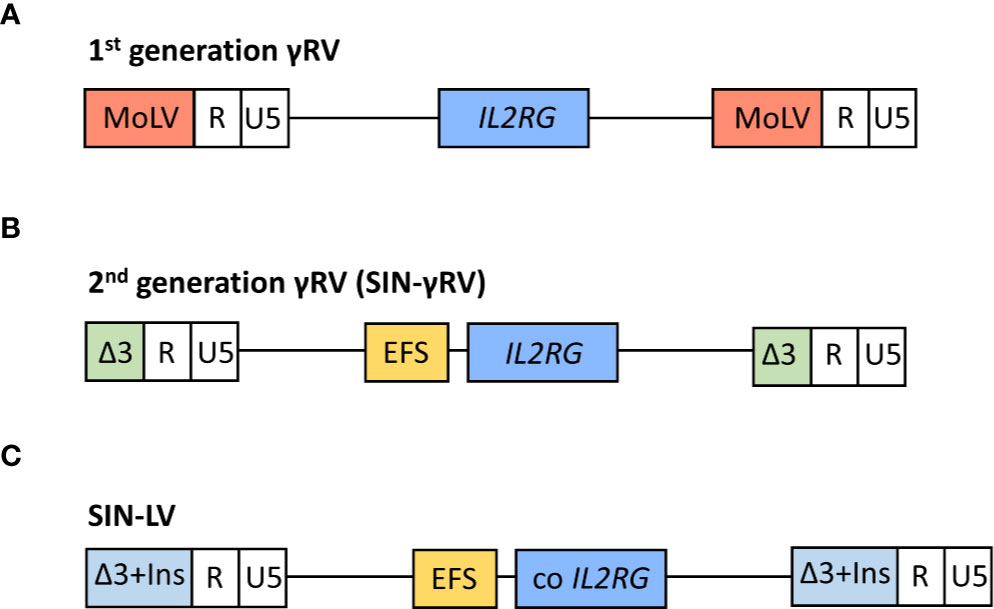 Figure 1 Schematic representation of viral vectors used in the different clinical trials for the treatment of SCID-X1 patients.1
Figure 1 Schematic representation of viral vectors used in the different clinical trials for the treatment of SCID-X1 patients.1
To overcome the risk of insertional oncogenesis, a new generation of vectors using the innovative integration pattern of LVs is inherently safer than that of MLV vectors. LVs preferentially integrate throughout the transcribed portion of active genes, targeting at high-frequency genes located in the outer portion of the nucleus in proximity to the nuclear pore, with no preference for regulatory elements or specific gene categories. Early data indicate the potential of LVs as a method of gene therapy for X-SCID.
Strategic Importance of Lentiviral Gene Therapy for X-SCID
Lentiviral vectors have become the preferred platform for many gene therapy applications, particularly those targeting hematopoietic stem cells. Compared with earlier γ-retroviral vectors, modern lentiviral systems provide improved safety profiles and greater control over gene expression.
Several characteristics make lentiviral vectors particularly suitable for the correction of IL2RG mutations:
01 Stable genomic integration
Lentiviral vectors integrate into the host genome, allowing long-term expression of therapeutic genes in hematopoietic stem cells and their progeny.
02 Efficient transduction of hematopoietic stem cells
Unlike many viral systems, lentiviruses can effectively transduce quiescent and slowly dividing cells, which is critical for modifying HSC populations.
03 Improved safety profiles
Self-inactivating (SIN) lentiviral vectors significantly reduce the risk of insertional oncogenesis compared with earlier vector generations.
04 Durable immune reconstitution
Successful lentiviral gene therapy can lead to long-term immune restoration by enabling corrected HSCs to generate functional immune cell lineages.
Common Challenges in Lentiviral Vector Development for X-SCID
Despite the significant progress achieved in lentiviral gene therapy, several technical challenges must be carefully addressed during vector development and optimization.
| Technical Challenge | Impact on Research or Therapy |
|---|---|
| Low transduction efficiency in hematopoietic stem cells | Reduced therapeutic gene delivery and limited immune reconstitution |
| Insertional mutagenesis risks | Potential activation of oncogenes due to integration events |
| Vector instability | Reduced viral titers and inconsistent transduction results |
| Limited scalability | Difficulty producing sufficient vector quantities for translational research |
| Transgene expression variability | Inconsistent therapeutic gene expression levels |
Our Solutions for X-SCID Lentiviral Vector Development
Creative Biolabs has developed a comprehensive set of technologies and strategies to overcome the technical challenges associated with lentiviral vector engineering.
Our Solutions
- Optimized pseudotyping strategies and transduction protocols
- Advanced self-inactivating (SIN) vector systems
- Promoter and regulatory element optimization
- High-titer lentiviral packaging platforms
Our Lentiviral Vector Development Capabilities for X-SCID
Creative Biolabs provides end-to-end lentiviral vector development services to support gene therapy research and preclinical studies focused on X-SCID.
| Module | Description |
|---|---|
| Vector design | Engineering of lentiviral transfer plasmids carrying optimized IL2RG expression cassettes |
| Gene synthesis | Codon optimization and sequence verification |
| Lentivirus production | High-titer lentiviral packaging and purification |
| Functional validation | In vitro transduction testing in relevant cell models |
| Biosafety testing | Replication-competent lentivirus detection and quality control |
Our integrated development platform allows researchers to rapidly design, optimize, and validate lentiviral vectors tailored to specific experimental goals.
Quality Control and Release Testing
Strict quality control is essential to ensure the safety and performance of lentiviral vectors.
| QC Test | Purpose |
|---|---|
| Functional titer measurement | Determine transduction efficiency |
| p24 ELISA | Quantify viral particle concentration |
| Sterility testing | Detect microbial contamination |
| Mycoplasma detection | Ensure culture purity |
| RCL assay | Detect replication-competent lentivirus |
Lentiviral Vector Development Workflow
Creative Biolabs follows a structured development process to ensure efficient project execution and reliable results.
-
Step 1 – Project consultation
Our experts work with clients to define research objectives, vector requirements, and experimental design.
-
Step 2 – Vector construction
Transfer plasmids containing the therapeutic gene cassette are designed and constructed.
-
Step 3 – Lentivirus packaging
High-titer lentiviral particles are produced using optimized packaging systems.
-
Step 4 – Viral purification and concentration
Purification methods such as ultracentrifugation or chromatography are used to improve vector quality.
-
Step 5 – Functional validation
Vectors are tested for transduction efficiency and transgene expression.
-
Step 6 – Quality control and delivery
Comprehensive QC testing ensures that vectors meet required specifications before delivery.
Applications of X-SCID Lentiviral Vectors
- Hematopoietic stem cell gene therapy: Ex vivo transduction of autologous CD34+ cells followed by reinfusion into patients after conditioning. This is the primary clinical application.
- Immunology research: Use of IL2RG-transduced HSCs to reconstitute a functional human immune system in immunodeficient mice (humanized mice) for studying human immune responses, infectious diseases, and cancer immunology.
- Preclinical efficacy & toxicology: IND-enabling studies in relevant animal models (e.g., X-SCID mice, non-human primates) to assess biodistribution, persistence, and safety.
- Disease modeling: Generation of IL2RG-corrected iPSCs or primary cells from X-SCID patients for studying disease mechanisms or drug screening.
- CAR-T and TCR-T cell therapy: IL2RG gene addition may be combined with chimeric antigen receptor (CAR) or T-cell receptor (TCR) vectors to enhance T-cell function in immunotherapy applications.
Why Choose Creative Biolabs
Creative Biolabs is committed to supporting innovative gene therapy research through advanced viral vector technologies.
Our advantages include:
-
Extensive experience in viral vector engineering
Our scientists have extensive expertise in lentiviral vector design and optimization. -
High-titer vector production platforms
We utilize optimized packaging systems to generate vectors with high functional titers. -
Customized development solutions
Each project is tailored to meet specific experimental goals and research requirements. -
Comprehensive quality control
Our QC framework ensures that vectors meet strict quality standards. -
Dedicated scientific support
Clients receive continuous technical support from experienced scientists throughout the project lifecycle.
Customer Reviews

Frequently Asked Questions
Q: Why are lentiviral vectors particularly suitable for X-SCID gene therapy?
A: Lentiviral vectors stably integrate into the genome of HSCs, ensuring that all daughter cells carry the functional IL2RG gene. They transcribe efficiently in non-dividing HSCs and provide long-term expression, essential for lifelong immune reconstitution. Their SIN design also reduces genotoxicity compared to earlier vectors.
Q: Can you customize promoters for immune-cell-specific expression?
A: Yes, we routinely use promoters like MND, EF1α, PGK, and also offer lineage-restricted (e.g., CD2 promoter for T cells, CD68 for myeloid) or synthetic promoters upon request. Inducible systems are also available.
Q: What viral titers can be achieved for X-SCID vectors?
A: Our standard high-titer preps range from 1×10⁸ to 5×10⁹ TU/mL (unconcentrated), and we can provide concentrated lots up to 1×10¹⁰ TU/mL for in vivo or large-animal studies.
Q: What QC tests are included as standard?
A: Standard QC includes functional titer (TU/mL), p24 ELISA, sterility, mycoplasma, endotoxin, and RCL assay. Additional assays (vector copy number, sequencing, Western blot, residual host cell DNA/protein) can be added as needed.
Connect with Us Anytime!
As a long-term expert in the field of LV development, Creative Biolabs owns lots of scientists who are proficient in LV development for X-SCID. We are confident in providing clients with the best service at the most competitive cost. Please contact us for more information and a detailed quote.
Reference
- Blanco E, Izotova N, Booth C, et al. Immune reconstitution after gene therapy approaches in patients with X-linked severe combined immunodeficiency disease. Frontiers in immunology, 2020, 11: 608653. https://doi.org/10.3389/fimmu.2020.608653 Distributed under Open Access license CC BY 4.0, without modification.
