What Is Measured in a Total Complement Activity Assay?
Total complement activity is commonly evaluated using hemolytic functional assays. In classical-pathway testing, a CH50 assay measures the capacity of a sample to lyse antibody-sensitized erythrocytes through the coordinated action of the classical pathway and terminal cascade. In alternative-pathway formats such as AH50 or AP50, assay conditions favor alternative-pathway activation and measure corresponding hemolytic capacity. These tests are useful because they integrate the function of multiple complement components instead of isolating a single analyte.
Depending on project goals, our service can be configured to assess:
-
Classical Pathway Total Activity
Measure global functional competence of the classical complement pathway using sensitized red blood cell hemolysis under validated assay conditions.
-
Alternative Pathway Total Activity
Evaluate the integrity of the alternative pathway under magnesium/EGTA or other pathway-selective conditions that suppress classical-pathway initiation while preserving alternative-pathway activity.
-
Comparative CH50/AH50 Profiling
Analyze both classical and alternative pathway activity side by side to reveal pathway-selective changes, sample-specific biases, or mechanism-of-action signatures.
-
Inhibitor Potency and Pharmacology
Quantify the degree to which antibodies, recombinant proteins, peptides, small molecules, aptamers, or serum factors suppress total complement function.
-
Serum Functional Status
Compare normal, disease, treated, or formulation-exposed sera to evaluate complement competence, depletion, consumption, or recovery.
-
Orthogonal Follow-Up
Pair total activity data with C1q binding, C3b deposition, sC5b-9 quantification, or cell-based complement assays to sharpen mechanism interpretation.
Great Minds Choose Creative Biolabs
Our Typical Workflow
At Creative Biolabs, total complement activity tests are respectively carried out by CH50 for the complement classical pathway, AH50 for the complement alternative pathway, and LP50 for the complement lectin pathway. All test procedures are shown as follows, both a one-stop test for all three pathways and a test for any one pathway based on the client's demands are all available.
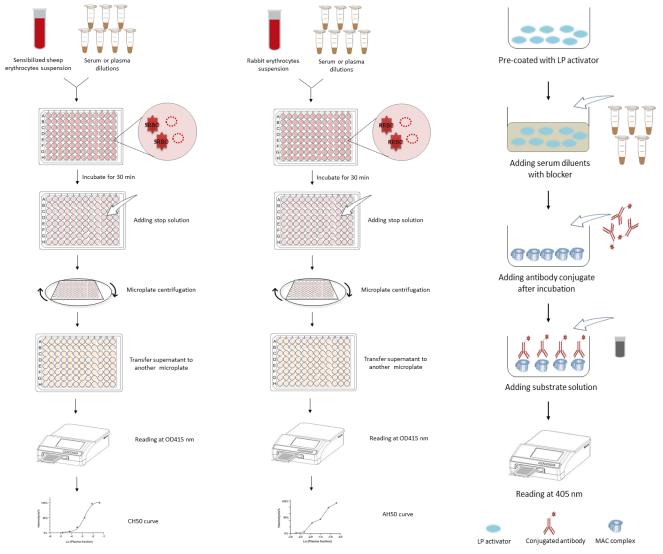 Fig. 1 CH50 test workflow, AH50 test workflow and LP50 test workflow.
Fig. 1 CH50 test workflow, AH50 test workflow and LP50 test workflow.
Design Your Workflow
Add-On Assays That Sharpen Interpretation
A total complement activity result is often most powerful when interpreted in the context of upstream or downstream assays. We routinely design multi-assay packages that connect pathway function to mechanism.
|
Assays
|
Descriptions
|
|
C1q Binding Assay
|
Useful when evaluating classical-pathway initiation potential, especially in antibody and immune complex programs.
|
|
C3b Deposition Assay
|
Helps determine whether functional changes seen in CH50 or AH50 correspond to altered upstream opsonization and convertase activity.
|
|
sC5b-9 Quantification
|
Supports terminal pathway interpretation and can help distinguish upstream suppression from downstream terminal pathway effects.
|
|
Individual Complement Component Activity Testing
|
Recommended when total activity patterns suggest selective pathway defects or target-specific inhibition.
|
|
Cell-Based Complement Assays
|
Helpful when serum hemolytic activity must be translated into a biologically relevant target-cell context.
|
|
Hemolysis Inhibition Assay
|
Useful for inhibitor pharmacology, potency ranking, and mechanism-supporting studies.
|
Design Your Customization
Case Studies
Case 1
Complement classical pathway test and alternative pathway test
The clients sent us 1 patient serum sample for the complement classical pathway test and alternative pathway test. The CH50 curve was drawn after we performed the CH50 test using the sensitized sheep erythrocytes. The AH50 curve was drawn after we performed the AH50 test using the rabbit erythrocytes. The CH50/AH50 value or serum dilution at 50% hemolysis of sheep red blood cells (SRBC) will be calculated using the curve formula.
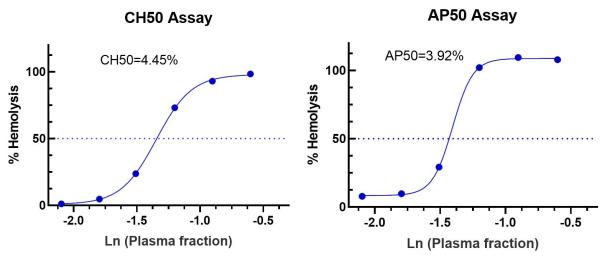 Fig. 2 Complement CH50/AH50 tests.
Fig. 2 Complement CH50/AH50 tests.
Case 2
CH50 and serum C2 levels in SSc patients in comparison to HC
This study aimed to assay complement fractions in SSc patients and to correlate their levels with the clinical course of disease. The results show an increment of CH50 and serum C2 levels in SSc patients in comparison to HC. The researchers maintain that CH50 may represent a biomarker of disease severity and of skin and lung fibrosis in these patients.
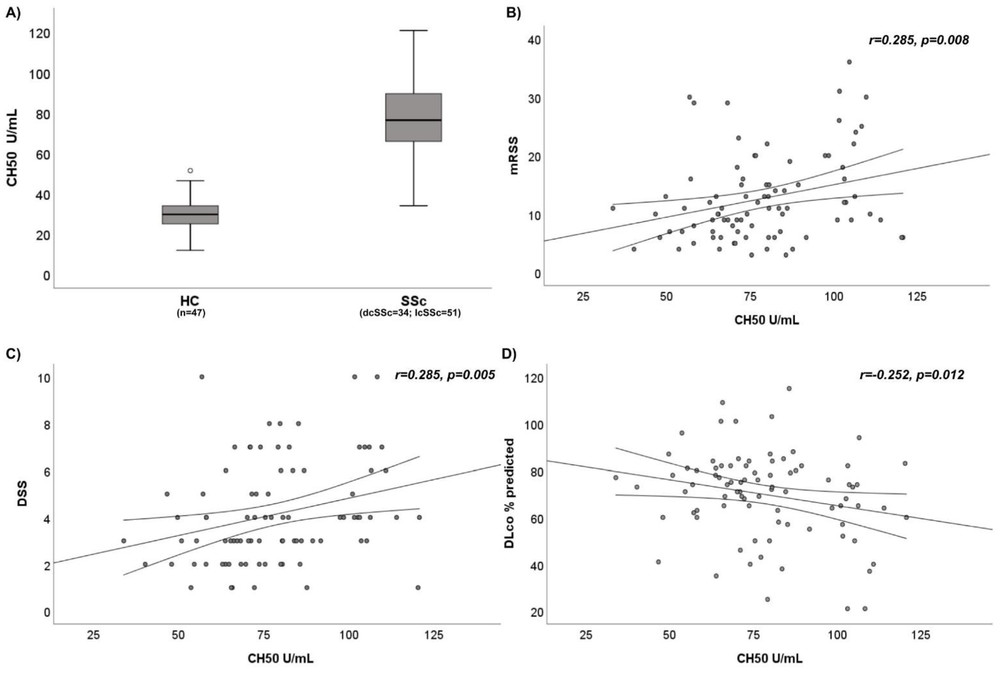 Fig. 3 Comparison of CH50 activity between SSc patients and HC.1,2
Fig. 3 Comparison of CH50 activity between SSc patients and HC.1,2
References
-
Pellicano, Chiara, et al. "Increased complement activation in systemic sclerosis patients with skin and lung fibrosis." Journal of Personalized Medicine 12.2 (2022): 284. https://doi.org/10.3390/jpm12020284
-
Distributed under Open Access license CC BY 4.0, without modification.
Frequently Asked Questions
What is the difference between CH50 and AH50?
CH50 is a classical-pathway hemolytic assay that evaluates the ability of a sample to mediate lysis through the classical pathway and downstream terminal cascade. AH50 is designed to evaluate alternative-pathway functional activity under pathway-selective conditions. When used together, these two assays can help distinguish whether a sample shows global complement impairment or pathway-specific dysfunction.
When is it better to run both CH50 and AH50?
Running both assays is often the most informative choice when mechanism is unclear, when you want a broader functional profile, or when you need to compare pathway effects side by side. A combined CH50/AH50 design is especially valuable for complement therapeutic development, translational sample profiling, deficiency-related research, and studies where pathway-selective interpretation is important.
How should samples be collected and stored?
Because complement is highly sensitive to pre-analytical conditions, proper sample handling is critical. In general, samples should be collected using complement-compatible procedures, processed promptly, aliquoted when appropriate, and stored frozen under conditions that preserve functional integrity. Repeated freeze-thaw cycles should be minimized whenever possible. Specific recommendations may vary depending on the exact workflow.
Can you work with limited sample volume?
In such cases, we can often help design a volume-conscious workflow by optimizing assay layout, replicate strategy, or prioritization plan. Sample limitations should be discussed during project planning so the study can be designed realistically.
Do you provide control recommendations?
Yes. We can recommend appropriate positive controls, negative controls, vehicle controls, pathway-biased controls, and comparator samples based on your specific project. Strong control design is essential for reliable interpretation of total complement activity data.
Can the assay be combined with other complement services?
Yes. In fact, many clients benefit from integrated testing. Total complement activity data can be combined with C3b deposition assays, C1q binding assays, sC5b-9 quantification, hemolysis inhibition studies, individual component analysis, or cell-based complement assays to provide a more complete mechanistic picture.














 Fig. 1 CH50 test workflow, AH50 test workflow and LP50 test workflow.
Fig. 1 CH50 test workflow, AH50 test workflow and LP50 test workflow.




 Fig. 2 Complement CH50/AH50 tests.
Fig. 2 Complement CH50/AH50 tests.
 Fig. 3 Comparison of CH50 activity between SSc patients and HC.1,2
Fig. 3 Comparison of CH50 activity between SSc patients and HC.1,2