Why Cell-Based Complement Activity Evaluation Matters
While purified-protein assays are useful for dissecting individual mechanisms, they cannot fully recapitulate the complexity of complement activation on a living cell surface. Cell-based systems, by contrast, allow investigators to measure complement engagement where it actually matters: at the interface of target membrane composition, antibody binding geometry, serum components, and endogenous complement regulators.
Our cell-based complement activity platform can be configured to assess classical-, lectin-, and alternative-pathway contributions, quantify surface deposition of complement fragments, evaluate terminal pathway progression, and connect upstream activation signals with downstream biological outcomes such as cell lysis, opsonization, and protection by inhibitors. By integrating assay design, optimized controls, pathway-resolved conditions, and robust analytics, Creative Biolabs delivers a clear picture of complement function in your model system.
Great Minds Choose Creative Biolabs
Cell Models We Commonly Support
We work with a broad range of cell types and can adapt protocols based on cell fragility, antigen level, serum compatibility, and the required endpoint.
-
Tumor Cell Lines - Useful for oncology and CDC-focused antibody programs, especially where target density and complement regulator expression influence outcome.
-
Primary Immune Cells - Suitable for depletion strategies, immune modulation studies, and donor-specific analyses.
-
Stem Cell-Derived and Engineered Cells - Applicable to regenerative medicine, cell therapy, and translational compatibility studies.
-
Epithelial and Endothelial Models - Relevant in inflammatory, renal, vascular, pulmonary, and barrier-related complement research.
-
Erythrocyte, Platelet, or Blood-Associated Models - Valuable for hemolysis-linked biology, transfusion research, and hematologic complement studies.
-
Infected, Stressed, or Cytokine-Primed Cells - Designed to mimic disease-associated surfaces that alter complement susceptibility or activation profile.
-
Client-Supplied Custom Models - We welcome client-specific cell lines, engineered targets, transfectants, and specialized biological systems when accompanied by handling guidance and project goals.
Design Your Protocol
Core Readouts Available in Our Cell-Based Platform
Our cell-based complement activity evaluation service can be structured around one or several complementary endpoints.
|
Readouts
|
Descriptions
|
|
Surface Complement Fragment Deposition
|
We quantify the deposition of key complement fragments such as C3b, iC3b, C4b, C4d, C3d, or terminal complex-associated markers on the cell surface. These measurements are especially valuable for understanding whether activation has been initiated, amplified, or stabilized on the target.
|
|
Terminal Pathway Progression
|
When the research goal requires insight into lytic competence, we assess progression into terminal pathway activity. This can include membrane attack complex-related deposition and correlations with functional cell injury.
|
|
Complement-Dependent Cytotoxicity
|
For cell-killing programs, we measure lysis or viability change following complement activation. Depending on the model, this may involve dye release, viability staining, metabolic readouts, or flow-cytometric discrimination of dead and live populations.
|
|
Opsonization and Phagocytic Relevance
|
In some programs, complement function is valuable not because it lyses the cell directly, but because it marks the target for immune recognition. In those cases, deposition-focused assays help estimate opsonization strength and support follow-on studies involving phagocytic or myeloid effector systems.
|
|
Inhibitor Potency and Mechanism in Complex Matrices
|
We compare inhibitor candidates in the presence of intact serum and membrane-bound targets to determine how robustly they suppress complement activation under near-physiological conditions. This is especially informative when serum proteins, competing ligands, or cell-surface regulators are expected to influence performance.
|
|
Pathway Bias and Mechanistic Resolution
|
By changing serum conditions, depletion/reconstitution formats, activator context, or blocking reagents, we can help determine whether observed activity is primarily classical, lectin, alternative, or mixed-pathway in origin.
|
Design Your Customization
Case Studies
Case 1
Modeling complement activation on human glomerular microvascular endothelial cells
The glomerular microvascular endothelial cells (GMVECs) are the primary target of AP activation in aHUS, but the exact mechanisms behind this high susceptibility of GMVECs to complement deposition remains unknown. The aim of this study was to model complement activation in aHUS patients ex vivo, by measuring complement deposition on human conditionally immortalized GMVECs (ciGMVECs). Moreover, by having the unique possibility to create a ciGMVEC line derived from an aHUS patient, the researchers could model the complement activation in samples of an aHUS patient on patient's own endothelial cells.
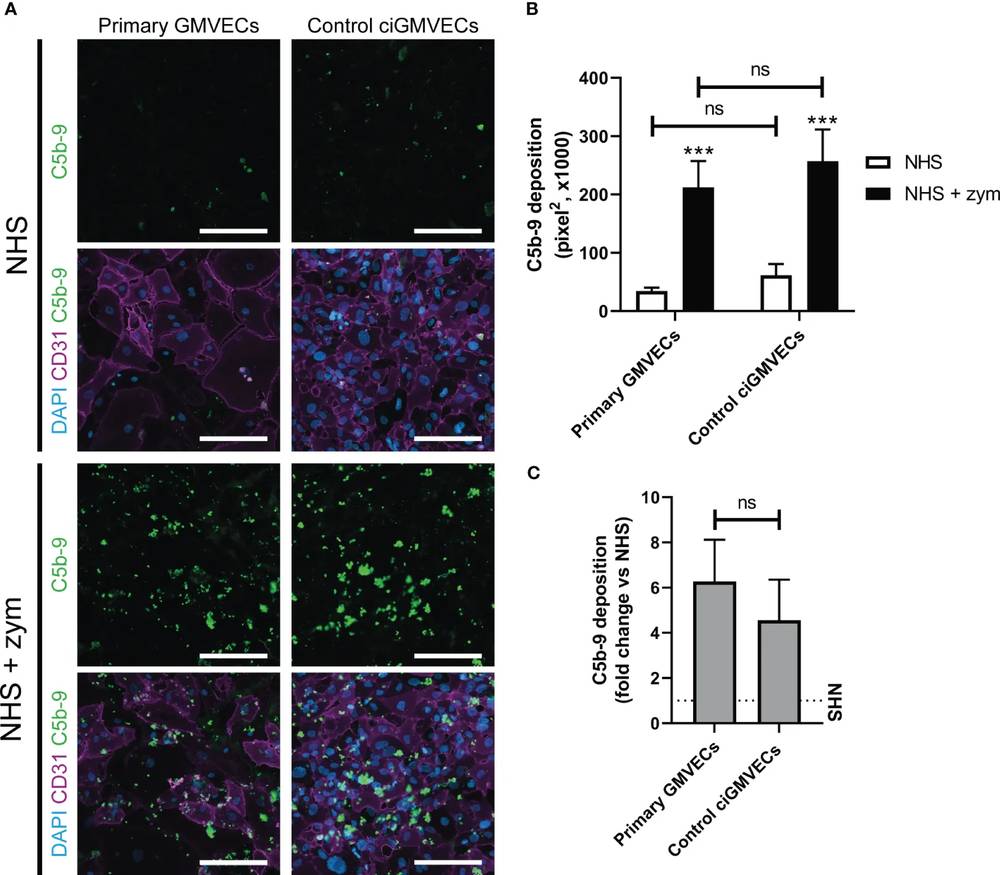 Fig. 1 Serum-induced C5b-9 deposition on primary glomerular microvascular endothelial cells (GMVECs) and conditionally immortalized GMVECs (ciGMVECs).1,2
Fig. 1 Serum-induced C5b-9 deposition on primary glomerular microvascular endothelial cells (GMVECs) and conditionally immortalized GMVECs (ciGMVECs).1,2
References
-
Stevens, Kes H., et al. "Modeling complement activation on human glomerular microvascular endothelial cells." Frontiers in immunology 14 (2023): 1206409. https://doi.org/10.3389/fimmu.2023.1206409
-
Distributed under Open Access license CC BY 4.0, without modification.
Frequently Asked Questions
How is a cell-based complement assay different from a purified complement assay?
Purified or plate-based assays are excellent for mechanistic reductionism and target-specific questions, but they do not fully capture the biological behavior of complement on living cell surfaces. Cell-based assays reflect membrane composition, antigen density, regulator expression, and cell-state effects, all of which can significantly alter complement outcomes.
Which endpoint should I choose first: deposition or lysis?
That depends on your biological question. If you need to know whether complement is engaging the target at all, deposition is often the most informative starting point. If your goal is direct effector function, such as CDC, a lysis readout should be included. Many programs benefit from measuring both, because strong upstream activity does not always lead to terminal pathway progression.
What if my cells express complement regulatory proteins?
That is common and often biologically important. Cell-surface regulators can significantly reduce terminal pathway progression even when upstream deposition is strong. We can incorporate study elements that clarify whether regulator expression is limiting your endpoint and whether a candidate can overcome or bypass that protection.
What sample types work best?
Serum is generally preferred for complement activation studies, and viable target cells are critical for cell-based work. Antibodies, inhibitors, proteins, and custom reagents can also be incorporated. We provide project-specific guidance before study initiation.
Can you combine this with CDC, C1q, or terminal pathway assays?
Absolutely. Many clients request integrated packages that connect upstream recruitment, intermediate deposition, and downstream function. This often produces a far clearer development story than any single endpoint alone.
What does the final report include?
Reports typically include methods, conditions, controls, figures, processed results, and interpretation. We aim to present not only what happened in the assay, but what the data mean for your program and which next steps may be most informative.









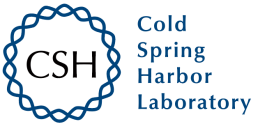








 Fig. 1 Serum-induced C5b-9 deposition on primary glomerular microvascular endothelial cells (GMVECs) and conditionally immortalized GMVECs (ciGMVECs).1,2
Fig. 1 Serum-induced C5b-9 deposition on primary glomerular microvascular endothelial cells (GMVECs) and conditionally immortalized GMVECs (ciGMVECs).1,2