Membrane attack complex (MAC) is an important immune effector of the complement terminal pathway that is composed of complement components C5b, C6, C7, C8, and C9. MAC is the collective come-off of three complement pathways (classical, lectin, or alternative pathway) that penetrates the cell membranes of microbes to form cytotoxic pores. These pores disrupt the phospholipid bilayers of target cell membrane, or form hydrophilic transmembrane channels, leading to cell lysis and death.
Structure and Assembly of MAC
MAC is a multiprotein complex consisting of five terminal complement proteins C5b, C6, C7, C8, and C9. Under electron microscopy, MAC pore measures approximately 10 nm in diameter and is a hollow structure embedded in the surface of pathogen cell membranes. The formation of MAC is initiated by the irreversible proteolytic cleavage of C5 into two components (C5a and C5b) by the C5 convertase. C5b provides a scaffold for C6 binding to form the stable intermediate C5b6, which can spontaneously combine with C7 to form C5b67 (MAC precursor). As a result of this junction, the configuration of C7 is altered, that is beneficial to the exposure the hydrophobic site on C7. Then C7 can insert into the lipid bilayer of cell membranes, where it has a high affinity for C8. Subsequently, C8 binds the membrane-associated C5b67 to form complex C5b678, which has a limited ability to lyse the cells. Finally, the formation of a fully active MAC ring is accomplished by the binding of 12-18 copies of C9.
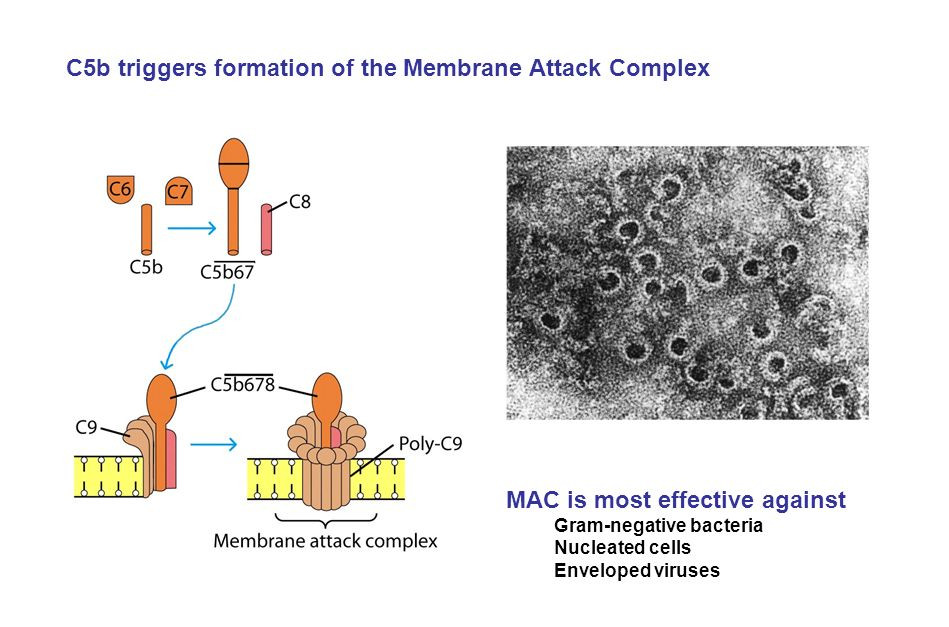
Fig.1 Structural formation and electron imagery of MAC.1, 3
Function of MAC
-
MAC-mediated Cytolysis
The foreign microorganism, viruses, fungi, and damaged erythrocytes or nucleated cells can be lysed by MAC. The cytolytic mechanism is considered to be penetration the cell membrane, leading to uncontrolled influx of ions and water down the osmotic gradient, causing the cell to swell and ultimately burst.
-
Production of Cytokine
The insertion of MAC increases membrane permeability, which is convenient for the ion activity crossing the membrane. Then a lot of intracellular signal pathways are triggered to produce various bioactive substances, including inflammatory mediators (e.g. oxygen free radicals, adipoid) and cytokines. These effects have a positive influence on pathogen clearance and amplification of inflammatory reaction.
-
Adhesion Molecule Expression
The formation of MAC in endothelial cells will promote the expression of p-selectin, which can increase the adhesion of neutrophils, platelets, and monocytes, then accelerate the release of inflammatory mediators.
-
Sublethal Effects
Recent studies suggest that karyocyte can remove MACs deposited on their cell membranes through vesiculation or endocytosis to protect themselves from complement dissolution.
Creative Biolabs provides a full range of therapeutic antibodies, inhibitors, soluble complement regulators, as well as customized services based on the MAC, including
-
Complement Therapeutic Target-C6
-
Complement Therapeutic Target-C7
-
Complement Therapeutic Target-C8
-
Complement Therapeutic Target-C9
Our comprehensive complement platform offers a great number of complement-related products in a rapid and cost-effective manner. If you are interested, please feel free to contact us for more details.
Published Data
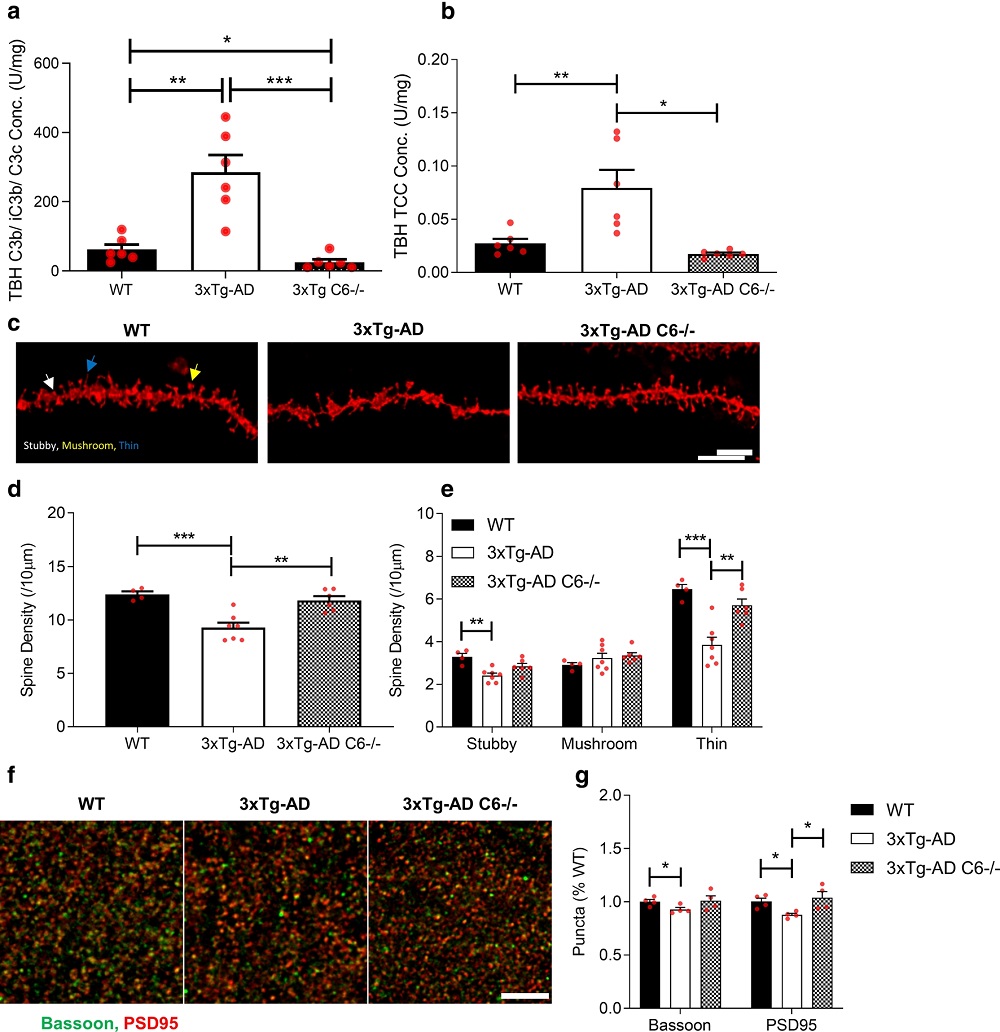 Fig.2 Terminal complement activity is diminished and hippocampal spine density preserved in C6-deficient 3xTg-AD murine models.2, 3
Fig.2 Terminal complement activity is diminished and hippocampal spine density preserved in C6-deficient 3xTg-AD murine models.2, 3
Immune mechanisms in Alzheimer's disease: Dysregulation of complement pathways -including C1q, C3 fragments, and MAC - influences synapse loss in the brains of AppNL-G-F model mice, intensifying with age and disease progression. Synaptoneurosomes and brain homogenates from these mice reveal an uptick in these components compared to controls. Mouse models treated systemically with anti-C7 antibodies or genetically altered to lack C6 exhibit modulated synapse loss, reflected in dendritic spine density near amyloid plaques. Findings underscore MAC's pivotal role in pathological synapse destruction, akin to neuromuscular junction impairment in myasthenia gravis, presenting a potential therapeutic target for Alzheimer's interventions.
References
-
Morgan, B. Paul, Courtney Boyd, and Doryen Bubeck. "Molecular cell biology of complement membrane attack." Seminars in cell & developmental biology. Vol. 72. Academic Press, 2017.
-
Carpanini, Sarah M., et al. "Terminal complement pathway activation drives synaptic loss in Alzheimer's disease models." Acta Neuropathologica Communications 10.1 (2022): 99.
-
Distributed under Open Access license CC BY 4.0, without modification.
Related Product
For Research Use Only.
Related Sections:


 Fig.2 Terminal complement activity is diminished and hippocampal spine density preserved in C6-deficient 3xTg-AD murine models.2, 3
Fig.2 Terminal complement activity is diminished and hippocampal spine density preserved in C6-deficient 3xTg-AD murine models.2, 3