Next-IO™ Anti-MSLN × CD3 Therapeutic Bispecific Antibody Program
About This Program
This program aims to develop anti-MSLN × CD3 therapeutic Bispecific Antibody for immuno-oncology.
Rationale for the program:
-
Mesothelin (MSLN), a 40 kDa glycosyl-phosphatidylinositol-linked (GPI) membrane glycoprotein, plays roles in cell survival, proliferation, tumor progression, and compliance. In the norm, MSLN is restrictedly expressed on mesothelial cells lining the peritoneum, pericardium, and pleura.
-
MSLN is overexpressed in several specific cancers, such as mesothelioma, pancreatic cholangiocarcinoma, ovarian cancer, and lung cancer, which are characterized by the invasive phenotype and poor prognosis.
-
Emerging clinical data reveals T cell engagers biotherapeutics can direct T cells to specific tumor cells, resulting in increased potency or tumor selectivity.
Given the above, we propose a novel combination - MSLN / CD3 therapeutic BiTE, which we believe will provide insights into the next-generation cancer treatment.
MSLN 
Although its biological function is not fully understood, the cancer-specific expression of MSLN makes it a potential therapeutic target to be researched in tumor studies. Currently, more research is still needed to verify its role in tumor metastasis.
The schematic on the right depicts MSLN regulation on epithelial-to-mesenchymal transition (EMT) and cancer stem cells:
-
MSLN has a knockdown that can upregulate epithelial cells and adhesion molecules, downregulate mesenchymal and CSC regulatory genes, and alleviate cancer cell self-renewal, proliferation, spread, and metastasis.
-
Overexpression of MSLN in normal cells stimulates anchorage-free growth, migration, and invasion.
MSLN in Cancer Studies
Here are some published data about MSLN × CD3 working as a potential target for cancer immunotherapy.
-
Overexpression of MSLN is associated with decreased survival rate in patients with epithelioid pleural mesothelioma.

Ongoing Clinical Trials
-
Currently, only one anti-MSLN × CD3 therapeutic Bispecific Antibody (called MG1122) is being evaluated in clinical Phase I study. Multiple preclinical data are emerging, demonstrating the potential of MSLN-based immunotherapy.
-
There are sufficient data for us to believe that MSLN × CD3 Bispecific Antibody is a compelling combination to be studied in cancer immunotherapy. In an effort to optimally leverage MSLN-mediated immune response, our next-generation MSLN targeting treatment attempts to explore combination therapy trials by involving other immunomodulatory agents.
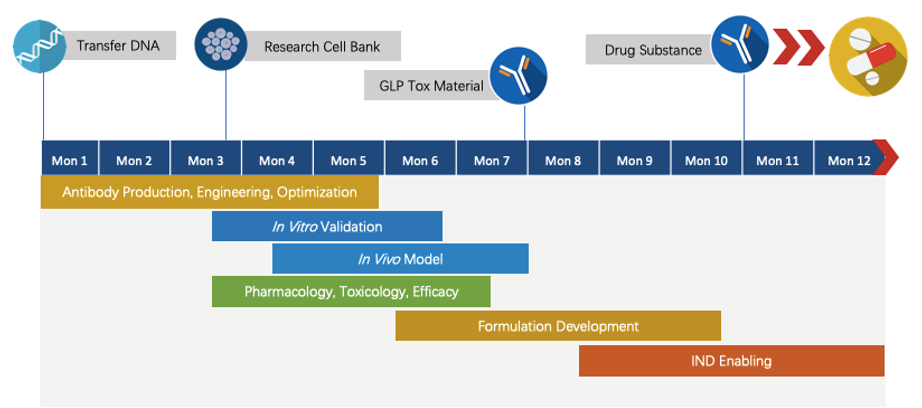
Program Planning and Management
Creative Biolabs has extensive knowledge in end-to-end program development. For each program, we are committed to delivering the final complete program to our clients within 1.5 years before entering the IND stage.
Cooperation
Creative Biolabs is looking for potential partners (include but not limit to major pharma or biotech firms) to develop anti-MSLN × CD3 therapeutic Bispecific Antibody program together. Our scientists are dedicated to bringing years of valuable experience to our partner and achieve a meaningful partnership together. For any partners interested in our Next-IO™ programs, Creative Biolabs welcomes collaboration.
Here are two ways for your choice, and please contact us for more details.
1) Collaborate with us and co-develop the programs from the discovery phase to IND enabling. Costs will be shared.
2) Become a licensed candidate for our programs.
With our quality control protocol and knowledge of global regulatory requirements, we can help our partners advance their programs with more chance to succeed. Look forward to cooperating with you in the near future.
Reference
-
Servais E L., et al. Mesothelin Overexpression Promotes Mesothelioma Cell Invasion and MMP-9 Secretion in an Orthotopic Mouse Model and in Epithelioid Pleural Mesothelioma Patients[J]. Clinical Cancer Research, 2012, 18(9): 2478-2489.
For Research Use Only | Not For Clinical Use


