Complement Activation Fragments Assay
Complement activation fragments provide some of the most informative readouts for understanding how a therapeutic candidate, biologic surface, delivery system, or pathological substrate interacts with the complement cascade. For drug developers, translational researchers, and platform teams working in immunology, inflammation, gene delivery, nanomedicine, and antibody engineering, these measurements deliver the mechanistic clarity needed to make confident decisions.
At Creative Biolabs, our complement activation fragments assay services are designed to quantify and interpret key deposited complement products across customized experimental settings. We help clients measure fragment deposition on cells, immune complexes, coated antigens, particles, biomaterials, and engineered delivery systems under physiologically relevant serum conditions.
Our service portfolio currently includes quantification of C3b deposition service and C5b-9 deposition measurement service. These services can be ordered independently or integrated into a broader complement characterization workflow together with C1q binding, CH50/AH50 testing, hemolysis inhibition, CDC evaluation, and complement inhibitor validation. By combining upstream, central, and terminal pathway readouts, we help generate a coherent and decision-ready complement profile for your candidate or model system.
Great Minds Choose Creative Biolabs
Assay Formats
To match diverse target types and project objectives, we offer multiple assay formats.

Microplate-Based Deposition Assays

Cell-Based Deposition Assays

Flow Cytometry-Based Opsonization and Terminal Deposition

Customized Surface and Biomaterial Assays
Design Your Workflow
Add-On Assays That Sharpen Interpretation
Complement activation fragments provide powerful information on their own, but many programs benefit from adjacent assays that add functional or mechanistic depth.
|
Assays
|
Descriptions
|
|
C1q Binding Assay
|
Useful for understanding classical pathway initiation potential, especially in antibody programs.
|
|
C4d or Early Classical/Lectin Markers
|
Helpful for mapping activation further upstream when central or terminal deposition is observed.
|
|
Soluble Terminal Complement Quantification
|
Useful when clients want both deposited and soluble terminal pathway perspectives.
|
|
CH50 / AH50
|
Provides pathway activity context and can complement fragment deposition results in broader cascade studies.
|
|
CDC Assay
|
When terminal deposition is biologically meaningful, cell lysis studies can help determine whether complement engagement translates into functional cytotoxicity.
|
|
Hemolysis Inhibition Assay
|
A practical functional add-on when inhibitor programs or complement-regulating strategies are under evaluation.
|
Design Your Customization
Published Data
Case 1
Ex vivo test for measuring complement attack on endothelial cells
These tests consist of the quantification of complement activation products (C3 activation fragments and C5b-9) deposits on EC by immunofluorescence (IF) measured by confocal microscopy or flow cytometry (fluorescence-activated cell sorting, FACS) after incubation with a serum sample of interest.
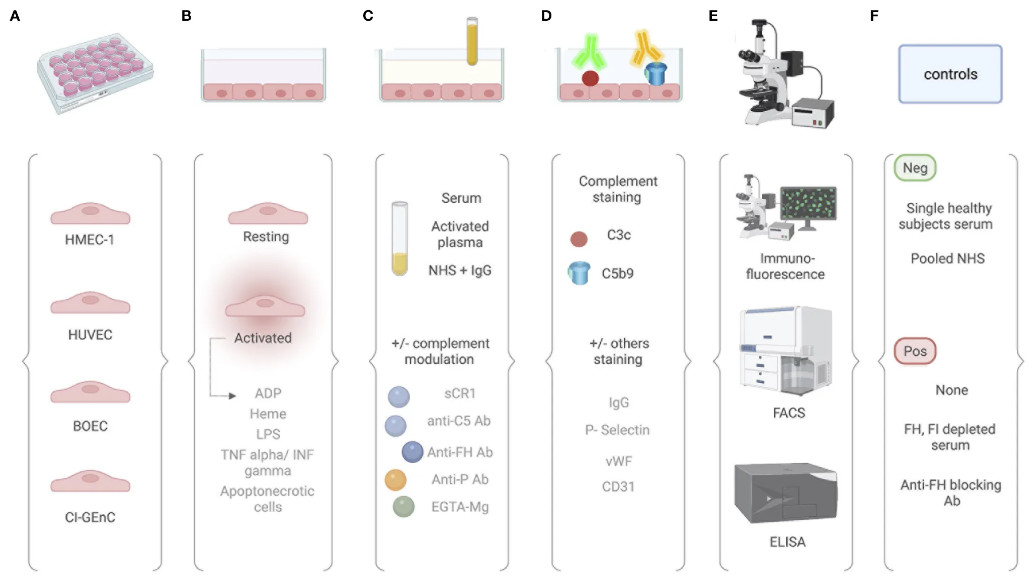 Fig. 1 Comparative analysis of different protocols used for the ex vivo complement activation test with endothelial cells.1,2
Fig. 1 Comparative analysis of different protocols used for the ex vivo complement activation test with endothelial cells.1,2
References
-
Meuleman, Marie-Sophie, et al. "Ex vivo test for measuring complement attack on endothelial cells: from research to bedside."Frontiers in immunology 13 (2022): 860689. https://doi.org/10.3389/fimmu.2022.860689
-
Distributed under Open Access license CC BY 4.0, without modification.
Frequently Asked Questions
Should I choose C3b deposition analysis, C5b-9 analysis, or both?
The answer depends on your scientific objective. If you want to understand early-to-central complement engagement, compare opsonization levels, or assess pathway amplification, C3b deposition is often the right starting point. If you are more concerned with terminal pathway activity, membrane attack complex formation, or downstream injury potential, C5b-9 may be more informative. In many projects, combining both gives the most complete picture.
Can you evaluate complement inhibitors using this service?
Yes. These assays are well suited for complement inhibitor development because they can directly show whether a test article reduces deposition of central or terminal complement products. Depending on assay design, they may help determine whether an inhibitor acts upstream, centrally, or closer to the terminal pathway.
What sample types can be tested?
We can support a range of sample and target types depending on project design. These may include purified antibodies, recombinant proteins, immune complexes, cultured cells, primary cells, nanoparticles, extracellular vesicles, viral vectors, beads, coated antigens, and selected biomaterial surfaces. If your material is unconventional, our team can evaluate whether a customized workflow is appropriate.
Do you offer pathway-specific assay design?
Yes. When scientifically appropriate, we can design studies to investigate the relative involvement of the classical, lectin, or alternative pathway. This may include specific serum conditions, depletion or reconstitution strategies, and carefully selected controls that help distinguish pathway contributions.
Which species are supported?
Human serum is commonly used and often preferred for translational relevance, but other species may be supported depending on the project. Available options can include mouse, rat, and non-human primate matrices, subject to experimental suitability and study goals.
How much sample is required?
Sample requirements vary based on assay format, number of conditions, replicate strategy, and whether optimization is needed. During project planning, we provide sample guidance tailored to your study design. If sample amount is limited, we can often suggest a phased or prioritized approach.



















 Fig. 1 Comparative analysis of different protocols used for the ex vivo complement activation test with endothelial cells.1,2
Fig. 1 Comparative analysis of different protocols used for the ex vivo complement activation test with endothelial cells.1,2